Asia-Pacific Cardiac Monitoring Market Analysis by Mordor Intelligence
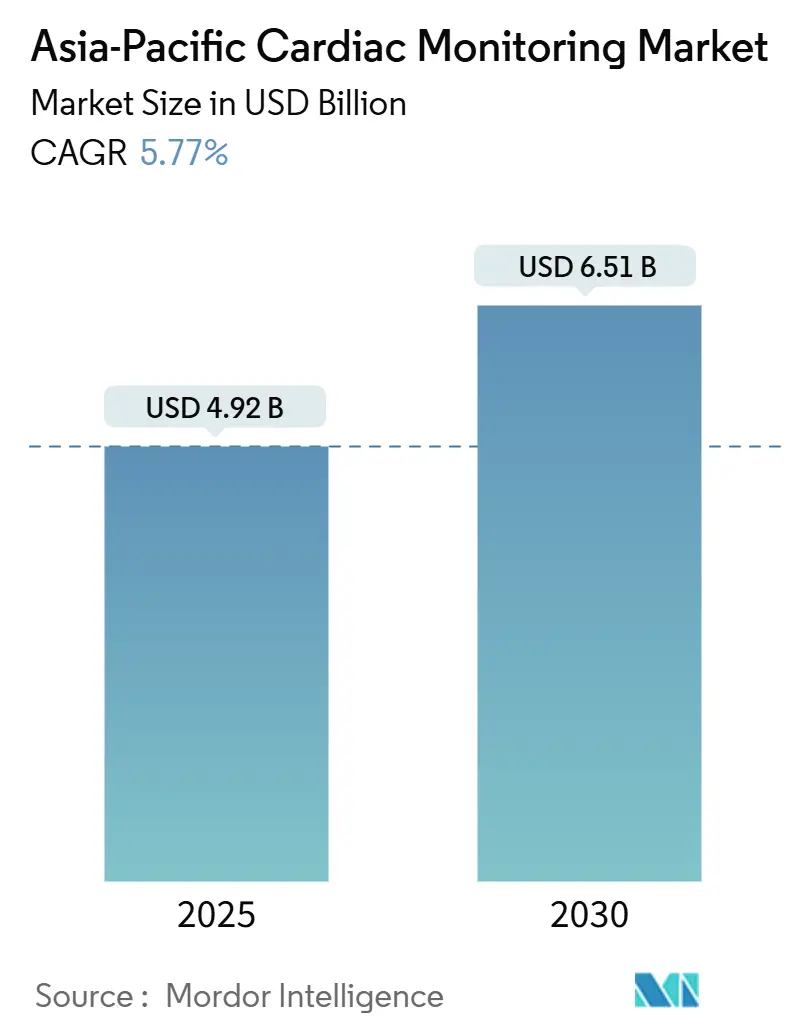
The Asia-Pacific Cardiac Monitoring Market size is estimated at USD 4.92 billion in 2025, and is expected to reach USD 6.51 billion by 2030, at a CAGR of 5.77% during the forecast period (2025-2030).
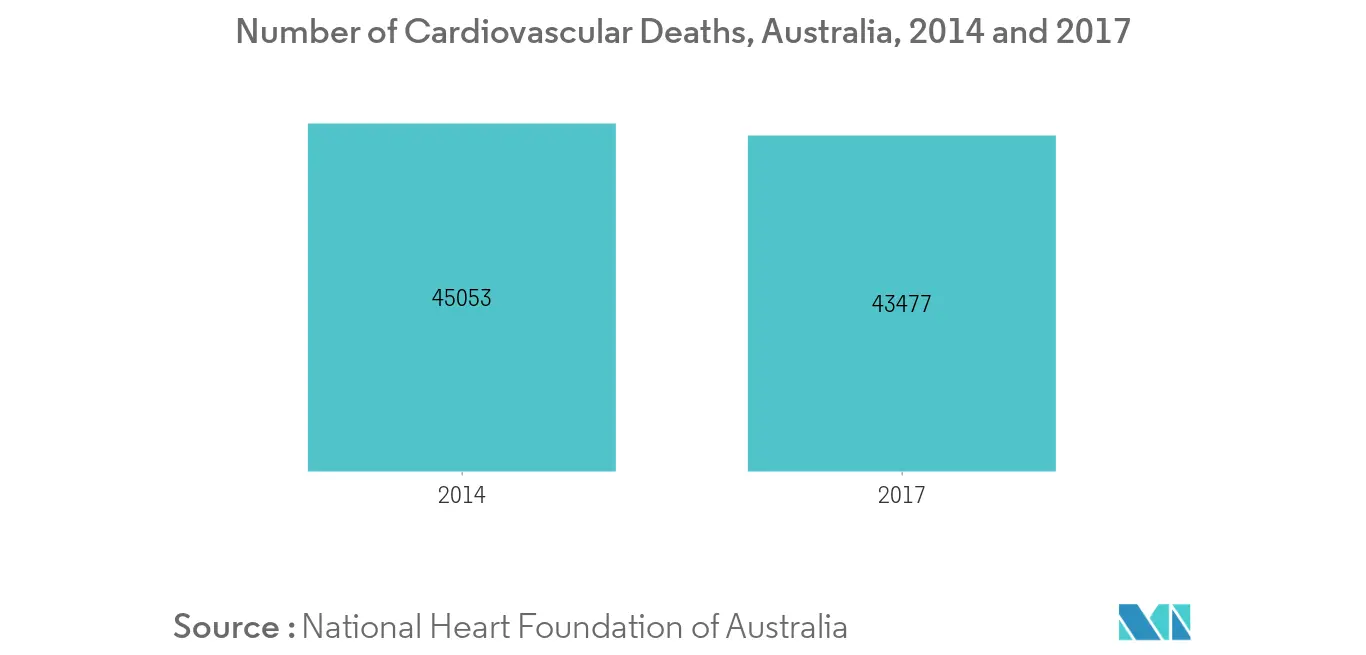
Cardiac monitoring devices are used to monitor and diagnose various abnormalities or disorders of the heart. The cardiac monitoring market is driven by rising burden of cardiovascular diseases, a growing ageing population who are very susceptible to coronary heart diseases, technological developments in cardiac monitoring devices that minimise the chances of infection, and improved efficiency of devices. Smoking is one of the major underlying causes of lung cancer and cardiac diseases. According to World Health Organisation (WHO), smoking causes around 1 million cardiovascular deaths in Asia-Pacific region each year. More than 1 in 5 cardiovascular diseases, such as heart attack and stroke, result from smoking. The Heart Foundation states that CVD is a major cause of death in Australia, with 43,477 deaths attributed to CVD in 2017. The rate of diagnosis of cardiovascular diseases is increasing over time due to rising burden of cardiac diseases and the need to control mortality rate by early diagnosis, and treatment for CVD with the help of cardiac monitoring devices. The other factors, such as technological advancements and the demand for home-based, point of care and remote monitoring cardiac systems are also anticipated to drive the market in the forecast period.
Asia-Pacific Cardiac Monitoring Market Trends and Insights
ECG Monitor Segment is Expected to Show Better Growth in the Forecast Years
Based on Device type, the market is segmented into ECG Monitor, Event Recorder, Implantable Cardiac Loop Recorder, Pacemaker, Defibrillator, CRT Devices, Smart Wearable and others. The ECG devices can be used for the continuous monitoring of patients suffering from CVD and for the detection of problems when they are usually easier to treat, while avoiding life-threatening and expensive emergency admissions.Thedevelopment of commercially advanced devices,with enabled Bluetooth sensor technology and mobile app technology is expected to drive the popularity of ECG Monitors. In 2018, Qardio, Inc., received the approval from Australia's Therapeutic Goods Administration (TGA) for QardioCore, its revolutionary ECG monitor.Furthermore, technological advancements and product innovations taking place in the development of ECG devices are likely to propel the market growth.
Competitive Landscape
The major players into the Asia-Pacific cardiac monitoring market are Abbott Laboratories, Biotronik, Boston Scientific Corporation, GE Healthcare, Hill-Rom Holdings, Inc., Medtronic PLC, MicroPort Scientific, Schiller AG.
Asia-Pacific Cardiac Monitoring Industry Leaders
Abbott Laboratories
Biotronik
Boston Scientific Corporation
Medtronic PLC
MicroPort Scientific
- *Disclaimer: Major Players sorted in no particular order


Asia-Pacific Cardiac Monitoring Market Report Scope
Continuous cardiac monitoring is an important tool in the clinical assessment of patients with a variety of conditions. It allows the detection of changes in heart rate and rhythm and is essential in the detection of life-threatening arrhythmia's.
| ECG Monitor |
| Event Recorder |
| Implantable Cardiac Loop Recorder |
| Pacemaker |
| Defibrillator |
| Cardiac Resynchronization Therapy (CRT) Devices |
| Smart Wearable |
| Other Device Types |
| Hospitals and Clinics |
| Home Care Settings |
| Ambulatory Surgical Centers |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific |
| Device Type | ECG Monitor | |
| Event Recorder | ||
| Implantable Cardiac Loop Recorder | ||
| Pacemaker | ||
| Defibrillator | ||
| Cardiac Resynchronization Therapy (CRT) Devices | ||
| Smart Wearable | ||
| Other Device Types | ||
| End User | Hospitals and Clinics | |
| Home Care Settings | ||
| Ambulatory Surgical Centers | ||
| Geography | Asia-Pacific | China |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||


Key Questions Answered in the Report
How big is the Asia-Pacific Cardiac Monitoring Market?
The Asia-Pacific Cardiac Monitoring Market size is expected to reach USD 4.92 billion in 2025 and grow at a CAGR of 5.77% to reach USD 6.51 billion by 2030.
What is the current Asia-Pacific Cardiac Monitoring Market size?
In 2025, the Asia-Pacific Cardiac Monitoring Market size is expected to reach USD 4.92 billion.
Who are the key players in Asia-Pacific Cardiac Monitoring Market?
Abbott Laboratories, Biotronik, Boston Scientific Corporation, Medtronic PLC and MicroPort Scientific are the major companies operating in the Asia-Pacific Cardiac Monitoring Market.
What years does this Asia-Pacific Cardiac Monitoring Market cover, and what was the market size in 2024?
In 2024, the Asia-Pacific Cardiac Monitoring Market size was estimated at USD 4.64 billion. The report covers the Asia-Pacific Cardiac Monitoring Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Asia-Pacific Cardiac Monitoring Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:








