Argentina In Vitro Diagnostics Market Analysis by Mordor Intelligence
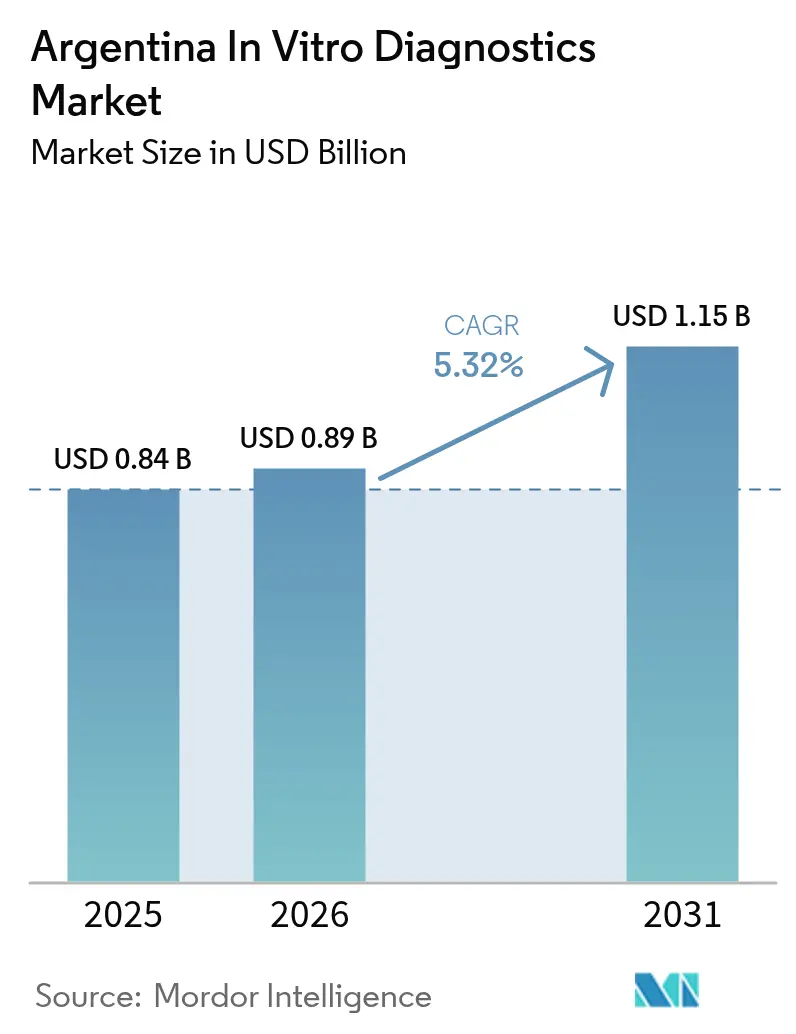
Argentina in vitro diagnostics market size in 2026 is estimated at USD 0.89 billion, growing from 2025 value of USD 0.84 billion with 2031 projections showing USD 1.15 billion, growing at 5.32% CAGR over 2026-2031. Rising chronic disease prevalence, wider private insurance uptake and public-sector lab upgrades are the primary engines of growth. Currency volatility, however, continues to raise the landed cost of imported analyzers and consumables, prompting hospitals to explore locally made reagents. Precision-medicine programs are stimulating demand for molecular assays, while mandatory e-prescriptions effective January 2025 link test data directly to clinical workflows. Opportunities also stem from point-of-care platforms that extend diagnostics into remote provinces.
Key Report Takeaways
- By test type, Clinical Chemistry led with 27.62% of Argentina in vitro diagnostics market share in 2025, while Molecular Diagnostics is projected to grow at a 8.92% CAGR through 2031.
- By product category, Reagents & Consumables accounted for 66.45% share of the Argentina in vitro diagnostics market size in 2025; Software & Services is anticipated to expand at a 10.42% CAGR to 2031.
- By technology, ELISA/CLIA methods held 36.55% Argentina in vitro diagnostics market share in 2025, whereas NGS Panels are forecast to rise at an 11.23% CAGR over the same period.
- By end user, Diagnostic Reference Laboratories commanded 50.35% share of the Argentina in vitro diagnostics market size in 2025; Point-of-Care settings exhibit the fastest growth at 9.86% CAGR to 2031.
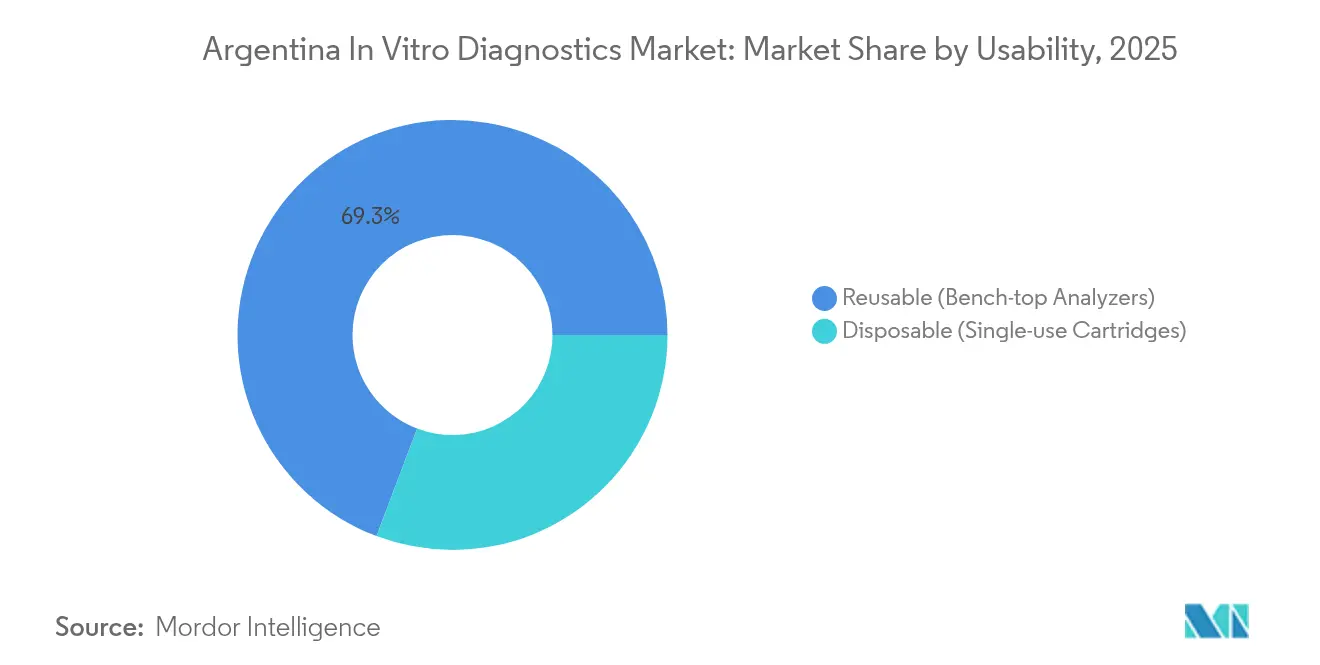
- By usability, Reusable Analyzers led with 69.25% of Argentina in vitro diagnostics market share in 2025; Disposable Cartridges are projected to expand at a 10.08% CAGR through 2031.
- By application, Infectious Diseases accounted for 32.60% share of the Argentina in vitro diagnostics market size in 2025, while Oncology/Cancer Biomarkers are advancing at a 9.05% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Argentina In Vitro Diagnostics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Burden of chronic & infectious diseases | +1.8% | Buenos Aires, Córdoba, Mendoza | Long term (≥ 4 years) |
| Government modernization of public laboratories & reimbursement | +1.2% | Urban centers | Medium term (2-4 years) |
| Expansion of private health insurance | +0.9% | Buenos Aires Metropolitan Area | Medium term (2-4 years) |
| Local reagent manufacturing initiatives | +0.7% | Industrial hubs nationwide | Long term (≥ 4 years) |
| Shift toward point-of-care testing in underserved provinces | +0.5% | Northwestern & northeastern provinces | Short term (≤ 2 years) |
| Post-COVID Acceleration of Molecular & Immunoassay Platform Adoption | +0.8% | National, with concentration in major cities and teaching hospitals | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Burden of Chronic & Infectious Diseases Boosting Diagnostic Demand
Non-communicable diseases account for 78% of Argentina’s disease burden, and cardiovascular ailments alone cause 30% of annual deaths[1]Thomas Gaziano, “Project Details – NIH RePORTER,” National Institutes of Health, nih.gov. High mortality from chronic kidney disease, with an age-standardized rate of 9.2 per 100,000, underscores the need for earlier testing. Hospitals are therefore investing in biomarker and molecular panels that shorten therapeutic decision times. Expanded HIV and STI screening programs further lift reagent volumes. Together these factors underpin steady unit growth across the Argentina in vitro diagnostics market.
Government Modernization of Public Laboratories & Test Reimbursement
The National Digital Health Strategy promotes interoperable lab information systems, while Programa Sumar ties provincial funding to performance indicators[2]Transform Health Coalition, “Country-Specific Fact Sheet: Argentina,” transformhealthcoalition.org. SENASA’s expanded laboratory network improves nationwide sample logistics. These actions stimulate procurement of analyzers compatible with electronic prescription workflows, accelerating data-driven diagnostics in the Argentina in vitro diagnostics market.
Expansion of Private Health Insurance
Private insurers now cover a widening range of preventive screens, creating a discerning customer base keen on high-specificity molecular tests. Regulatory price caps linked to a “health cost index” push payers toward value-based purchasing, favoring platforms with demonstrable clinical impact. Uptake is strongest in Buenos Aires, where higher household incomes support premium policies that reimburse next-generation sequencing.
Shift Toward Point-of-Care Testing in Underserved Provinces
mHealth-enabled POCT initiatives improve cardiovascular screening adherence in Jujuy, Misiones and Salta provinces through the PRIMECare trial. Portable lateral-flow assays allow same-visit treatment decisions where central labs are distant. This decentralization accelerates result turnaround and elevates test volumes for manufacturers with rugged cartridge platforms.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Currency volatility elevating import costs | -1.5% | Nationwide | Medium term (2-4 years) |
| Fragmented provincial procurement hindering volume-based pricing | -0.8% | Smaller provinces | Long term (≥ 4 years) |
| Shortage of Skilled Laboratory Workforce Outside Major Urban Centers | -0.6% | Rural provinces and secondary cities | Long term (≥ 4 years) |
| Extended ANMAT Approval Cycles Delaying Market Entry | -0.7% | National, with heightened impact on innovative technologies | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Currency Volatility Elevating Import Costs for Instruments & Consumables
Multiple exchange rates and capital controls lift the peso cost of imported analyzers, forcing providers to delay upgrades. Even as the federal budget posted a primary surplus in 2024, recurrent devaluation inflated reagent invoices by double digits[3]OECD, “OECD Economic Outlook 2024 Issue 2: Argentina,” oecd.org. Vendors often denominate quotes in USD, adding hedging surcharges that compress hospital margins and slow high-end segment adoption inside the Argentina in vitro diagnostics market.
Fragmented Provincial Procurement Hindering Volume-based Pricing
Argentina’s 24 provinces run independent tender cycles that prevent pooled purchasing power. Studies in Mendoza show 19.7% of adults reporting unmet care needs attributable to administrative inefficiencies. Smaller jurisdictions thus pay higher unit prices for assays, widening access gaps between urban centers and remote districts. Limited batch sizes also deter suppliers from holding local inventory, elongating lead times.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Test Type: Molecular Diagnostics Gaining Speed
Clinical Chemistry generated 27.62% of Argentina in vitro diagnostics market share in 2025 on the back of high-volume lipid, renal and liver panels used for chronic disease monitoring. Routine deployment across public and private labs ensures predictable reagent pull-through. Yet reimbursement ceilings cap price escalation, prompting vendors to focus on reagent stability to protect margins. Immunodiagnostics maintains traction in HIV and hepatitis screening, where algorithmic confirmatory testing drives repeat orders.
Molecular Diagnostics is forecast to post a 8.92% CAGR, the swiftest within the Argentina in vitro diagnostics market. COVID era capacity remains in place, repurposed toward oncogenic mutation detection and multi-pathogen respiratory panels. Laboratories in Buenos Aires and Córdoba are validating liquid biopsy workflows despite staffing gaps that slow nationwide rollout. The segment’s rise signals a long-term tilt toward precision medicine, though test affordability remains a hurdle away from major metros.


By Product: Software & Services Accelerating Connectivity
Reagents & Consumables delivered 66.45% of Argentina in vitro diagnostics market size in 2025 due to the consumable nature of chemistry, immunoassay and PCR workflows. Currency swings have made single-source contracts desirable, pushing health systems to cultivate local suppliers. The emergent domestic reagent consortium, backed by academic know-how, is shortening lead times and stabilizing kit pricing for provincial hospitals.
Software & Services is projected to grow 10.42% per year as e-prescription mandates drive adoption of lab information systems. Middleware that consolidates analyzer outputs into clinical records elevates data integrity and meets audit requirements. Vendors offering turnkey integration with hospital EMR suites enjoy an early-mover edge in the Argentina in vitro diagnostics market, particularly where cloud connectivity is feasible.
By Technology: NGS Panels Driving Genomic Profiling
ELISA/CLIA procedures still accounted for 36.55% of revenue in 2025, owing to versatility in hormone, autoimmune and pathogen testing. Advances such as magnetic-bead separation and nanostructured surfaces have raised assay sensitivity, extending platform relevance. Real-time PCR retains dominance in viral load monitoring given its lower capital threshold versus sequencing.
NGS Panels are set to register an 11.23% CAGR and will expand Argentina in vitro diagnostics market size through broader oncology panels reimbursed by private insurers. Laboratories are piloting hereditary cancer panels that consolidate multiple genes into one run, reducing per-sample cost. However, skill shortages outside Buenos Aires hinder uniform adoption, and reimbursement codes are still evolving.
By Usability: Disposable Cartridges Extending Reach
Reusable analyzers comprised 69.25% of 2025 instrument placements, leveraging throughput economies in central labs. Their upgrade cycle, typically seven to 10 years, shields suppliers from short-term shocks. Preventive maintenance packages are bundled to keep uptime above 95%, a contractual metric demanded by tertiary hospitals.
Disposable cartridge systems are forecast to grow 10.08% annually, supported by public-health projects delivering rapid tests at primary-care posts. Cardiovascular and renal panels packaged into sealed microfluidic chips simplify training, vital in regions lacking specialist technologists. Field studies with hantavirus antibody strips further demonstrate cartridge versatility.

By Application: Oncology Biomarkers Outpacing Growth
Infectious disease assays held 32.60% revenue in 2025 as Argentina sustained HIV, dengue and Chagas surveillance programs. Ministries procure combined ELISA and rapid tests to widen screening breadth, reinforcing steady reagent demand. Diabetes testing adds a sizable share, mirrored by escalating type 2 prevalence among adults.
Oncology biomarkers are projected to expand at a 9.05% CAGR, the fastest within the Argentina in vitro diagnostics industry. Precision-oncology clinics order comprehensive genomic profiles to tailor targeted therapies, with private payers reimbursing companion diagnostics. Public coverage remains limited, pushing manufacturers to craft tiered panel offerings to suit diverse budgets.
By End User: Point-of-Care Settings Gathering Momentum
Diagnostic reference laboratories captured 50.35% of Argentina in vitro diagnostics market size during 2025 principally through consolidated sample volumes and specialized test menus. Their long-term agreements with managed-care organizations guarantee reagent throughput, though staff attrition in secondary cities threatens turnaround-time targets.
Point-of-care environments are poised for 9.86% CAGR growth as handheld analyzers reach pharmacies, ambulatory clinics and mobile units. The PRIMECare intervention couples Bluetooth-enabled cholesterol meters with teleconsults, illustrating how integrated workflows can raise screening adherence in remote towns.
Geography Analysis
Buenos Aires Metropolitan Area accounted for roughly 39.45% of national test volumes thanks to high insurance coverage and a dense network of tertiary hospitals. Reference labs here run 24/7 sequencing hubs, making the region the early adopter of NGS oncology panels despite elevated import costs. Provincial governments depend on capital-city logistics corridors that expedite reagent distribution.
Central provinces of Córdoba, Santa Fe and Mendoza form the second cluster of demand. Regional nephrology clinics rely heavily on chemistry and immunoassay panels to monitor chronic kidney disease, an area with a 9.2 per 100,000 mortality footprint. Digital-health pilots using cloud LIS platforms demonstrate how the Argentina in vitro diagnostics market can address distance barriers between secondary cities and rural catchments.
Northwestern and northeastern provinces remain under-served. Limited centrifuge infrastructure and shortages of laboratory technologists constrain routine testing capacity. Pilot programs distribute portable lateral-flow kits for dengue and leptospirosis, bridging gaps until fixed labs are upgraded. Conditional transfers under Programa Sumar incentivize these provinces to expand basic chemistry services, yet procurement fragmentation keeps analyzer prices elevated.
Competitive Landscape
The Argentina in vitro diagnostics market is moderately concentrated. Multinationals such as Abbott, Roche and Siemens Healthineers command the bulk of high-end instrument placements, leveraging nationwide distributor footprints and service depots. Their strategy centers on reagent-rental contracts that preserve long-term customer lock-in despite peso volatility.
Local biotechnology firms are scaling reagent production, with 340 companies active by 2023 and revenues of USD 3.75 billion. Partnerships between public labs and private developers accelerate tech-transfer of ELISA kits for endemic diseases. BD’s decision to spin off its Biosciences and Diagnostic Solutions business marks a global move toward more focused portfolios, hinting at potential distribution realignments in Argentina.
Competitive advantage is tilting toward integrated ecosystems that link analyzers, middleware and decision-support modules. Vendors embedding regulatory reporting templates for ANMAT’s updated import fee structure position themselves as compliance partners rather than mere suppliers, an increasingly important distinction amid evolving trade rules.
Argentina In Vitro Diagnostics Industry Leaders
Danaher Corporation
Bio-Rad Laboratories Inc.
BioMérieux
F. Hoffmann-La Roche Ltd.
Thermo Fisher Scientific Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2025: Argentina’s biotech sector reported 340 companies generating USD 3.75 billion in product revenue, including molecular diagnostic kits and ELISA assays
- December 2024: The Chan Zuckerberg Initiative granted USD 300,000 to bolster Argentina’s Reagent Collaboration Network, promoting local reagent production.
- April 2024: ANMAT updated import fee schedules for medical products, including IVDs, introducing a progressive structure aligned with technological complexity.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the Argentina in-vitro diagnostics (IVD) market as all reagents, consumables, analyzers, software, and services that enable clinical testing of human biological samples across laboratory, hospital, point-of-care, and home settings. According to Mordor Intelligence, the market is valued at USD 0.84 billion in 2025 and is projected to reach USD 1.09 billion by 2030, reflecting the full spectrum of test technologies from clinical chemistry to next-generation sequencing.
Scope Exclusion: Veterinary diagnostics and in-house research-use assays remain outside this definition.
Segmentation Overview
- By Test Type
- Clinical Chemistry
- Immunodiagnostics
- Molecular Diagnostics
- Hematology
- Coagulation & Hemostasis
- Microbiology
- Point-of-Care Lateral Flow
- Other Test Types
- By Product
- Instruments / Analyzers
- Reagents & Consumables
- Software & Services
- By Technology
- ELISA / CLIA
- Real-time PCR & Isothermal NAAT
- NGS Panels
- Microarrays & Lab-on-Chip
- Lateral-Flow Immunoassay
- Flow Cytometry
- By Usability
- Disposable (Single-use Cartridges)
- Reusable (Bench-top Analyzers)
- By Application
- Infectious Diseases
- Diabetes
- Cancer / Oncology Biomarkers
- Cardiology
- Autoimmune & Inflammatory Disorders
- Nephrology
- Blood Screening & Transfusion Safety
- Other Applications
- By End User
- Diagnostic Reference Laboratories
- Hospitals & Clinics
- Point-of-Care Settings (Polyclinics, Ambulances)
- Home & Self-Testing Channels
Detailed Research Methodology and Data Validation
Primary Research
Our team interviewed laboratory managers in Buenos Aires and Córdoba, procurement heads at private hospital chains, public tender officials, and regional distributors. These conversations clarified test-mix shifts, average selling prices, reagent replenishment cycles, and import-delay buffers, which helped us reconcile secondary gaps and fine-tune model coefficients.
Desk Research
We began with structured desk work that harvested public datasets from credible bodies such as the National Ministry of Health, INDEC trade statistics, the Pan American Health Organization, and peer-reviewed journals indexed on PubMed. Industry-specific associations, including the Argentine Chamber of Diagnostic Products, complemented company 10-Ks and investor decks to size reagent flows and instrument installed bases. To enrich hard-to-find shipment splits, Mordor analysts tapped premium repositories, D&B Hoovers for company revenues, Dow Jones Factiva for price movements, and Questel for patent velocity tracking. This list is illustrative; multiple other sources were reviewed to ground every assumption.
Market-Sizing & Forecasting
The baseline derives from a top-down construct that rebuilds national demand from patient encounters and test-per-case ratios, layered with import-export tallies and public reimbursement volumes. Supplier roll-ups and sampled price×volume checks supply a selective bottom-up lens for validation. Key variables like diabetes prevalence, oncology biopsy counts, public-sector procurement budgets, reagent duty rates, and point-of-care cartridge penetration feed a multivariate regression whose parameters are stress-tested through scenario analysis before final locking. Forecasts reflect weighted consensus from our primary expert panel on currency depreciation and technology adoption curves. Gaps in sub-segment reporting were bridged using analog benchmarks from neighboring Brazil, adjusted by per-capita healthcare spend differentials.
Data Validation & Update Cycle
Outputs pass two analyst reviews, automated variance screening, and a senior sign-off. We refresh every twelve months, with interim revisions triggered by regulatory or macro shocks, ensuring clients always access the most current view.
Why Mordor's Argentina In Vitro Diagnostics Baseline Commands Confidence
Published numbers often differ because firms choose dissimilar product baskets, currency bases, and refresh cadences.
Key gap drivers we notice include narrower scope limited to reagents, undisclosed currency rebasing, and one-time survey extrapolations that skip laboratory import volatility. Our disciplined scoping, annual refresh, and dual-source validation give decision-makers a steadier anchor.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 0.84 B (2025) | Mordor Intelligence | - |
| USD 0.42 B (2024) | Regional Consultancy A | Excludes software and home testing; converts sales at fixed 2023 FX rate. |
| USD 0.59 B (2024) | Global Consultancy B | Covers only instruments and reagents; relies on three-year-old hospital survey without import adjustments. |
The comparison shows that, while others provide useful snapshots, Mordor's broader scope, live FX tracking, and annual field validation yield the most balanced, transparent baseline for planning and investment decisions.


Key Questions Answered in the Report
What is the current value of the Argentina in vitro diagnostics market?
The market is valued at USD 0.89 billion in 2026 and is expected to reach USD 1.15 billion by 2031.
Which test type is growing the fastest?
Molecular Diagnostics is projected to record a 8.92% CAGR through 2031 as oncology and infectious-disease uses expand.
How is government policy influencing laboratory demand?
Programmes such as the National Digital Health Strategy and Programa Sumar are driving lab modernization and reimbursement, boosting demand for interoperable analyzers and reagents.
Why is local reagent production important?
Domestic manufacturing reduces currency-related import cost spikes and improves supply security, strengthening market resilience.
Which regions show the greatest unmet diagnostic need?
Northwestern and northeastern provinces face limited lab capacity and benefit most from point-of-care testing initiatives.
What technology is advancing precision medicine in Argentina?
Next-generation sequencing panels are rapidly gaining ground, enabling comprehensive genomic profiling for targeted cancer therapy decisions.
Page last updated on: