Skin Cancer Therapeutics Market Size and Share
Market Overview
| Study Period | 2022 - 2031 |
|---|---|
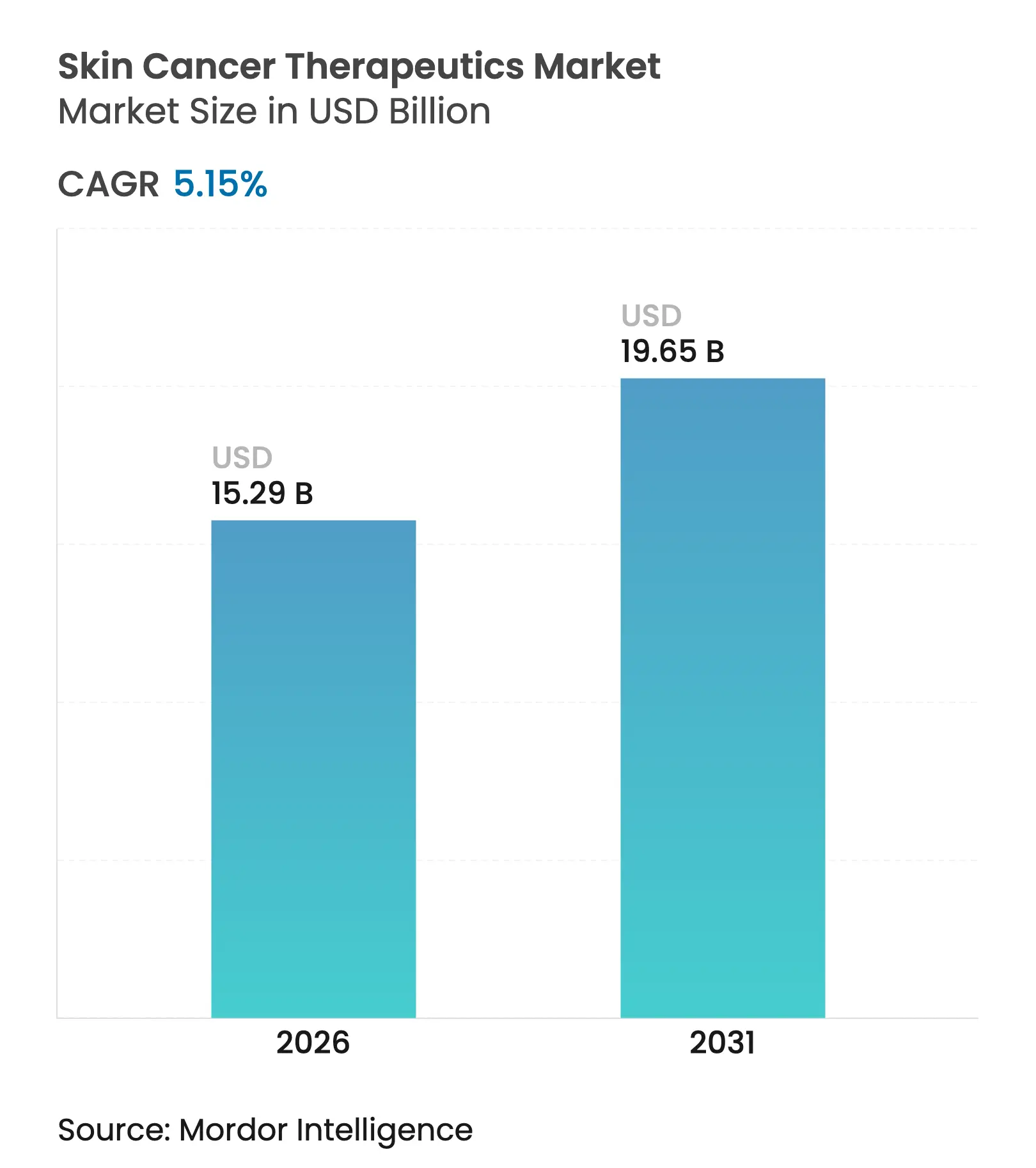
| Market Size (2026) | USD 15.29 Billion |
| Market Size (2031) | USD 19.65 Billion |
| Growth Rate (2026 - 2031) | 5.15 % CAGR |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
| Market Concentration | Medium |
Major Players *Disclaimer: Major Players sorted in no particular order. Image © Mordor Intelligence. Reuse requires attribution under CC BY 4.0. |
Skin Cancer Therapeutics Market Analysis by Mordor Intelligence
The skin cancer therapeutics market size in 2026 is estimated at USD 15.29 billion, growing from 2025 value of USD 14.54 billion with 2031 projections showing USD 19.65 billion, growing at 5.15% CAGR over 2026-2031. The adoption of precision medicine, real-time molecular tests, and artificial-intelligence (AI) decision support is moving care away from stand-alone surgery and toward data-guided therapy selection. AI diagnostic platforms now match specialist performance, posting 96% sensitivity, while mRNA-based vaccines have cut melanoma recurrence rates by 49% in late-stage studies.[1]American Cancer Society, “Cancer Facts & Figures 2025,” cancer.org Regulators are fast-tracking novel agents, payers are broadening reimbursement for tele-dermatology, and investors are funding outpatient delivery models that lower procedure costs. Supply-side dynamics are equally important: pharmaceutical alliances combining checkpoint inhibitors with personalized vaccines, manufacturers embedding AI into therapeutics, and practice consolidations backed by private equity are together reshaping competitive positioning.
Key Report Takeaways
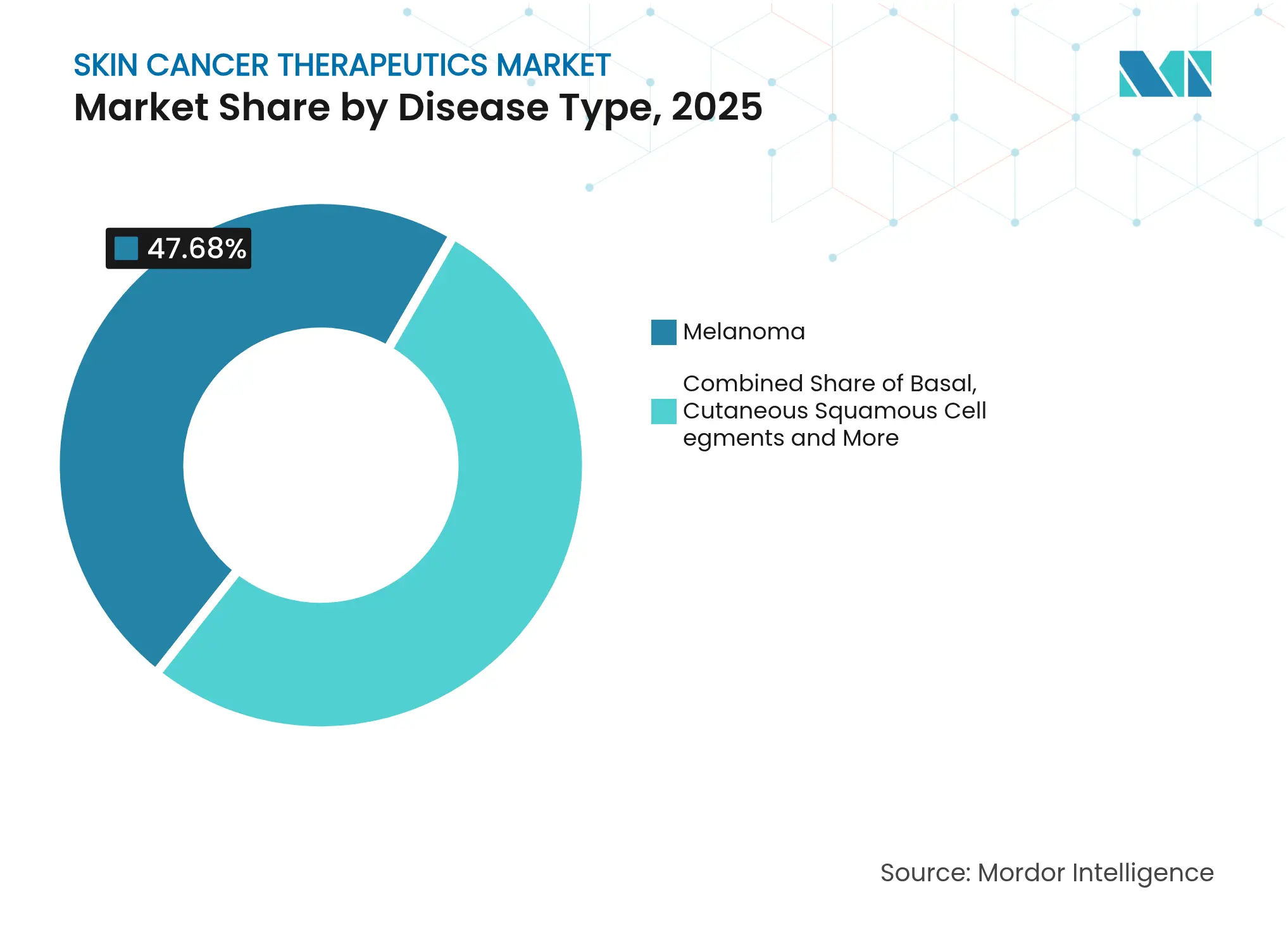
- By disease type, melanoma held 47.68% of the skin cancer therapeutics market share in 2025, while Merkel cell and other rare cancers are projected to expand at a 13.02% CAGR through 2031.
- By treatment modality, immunotherapy led with a 41.92% revenue share in 2025; mRNA-immunotherapy combinations are forecast to grow at a 21.2% CAGR to 2031.
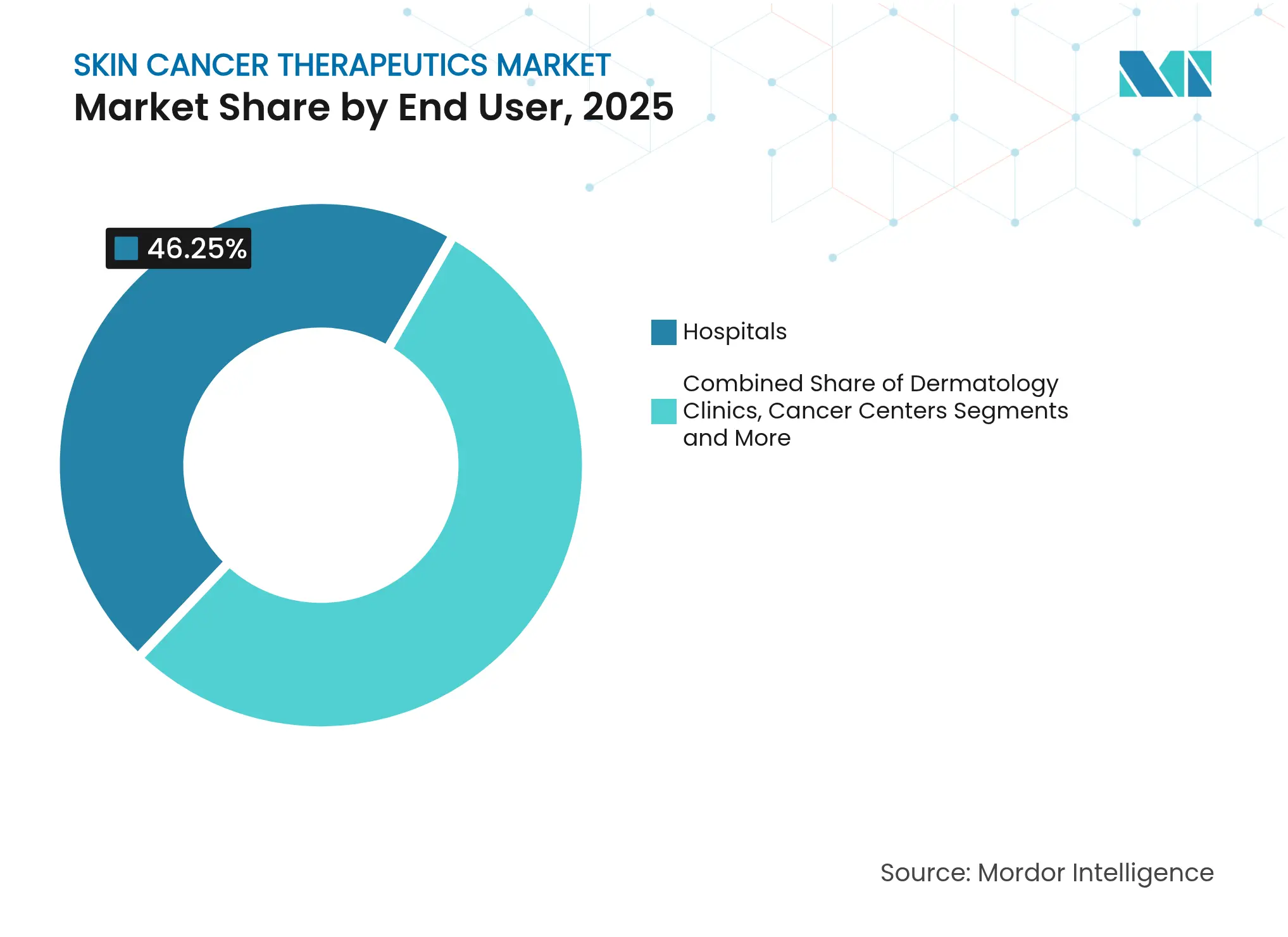
- By end-user, hospitals accounted for 46.25% share of the skin cancer therapeutics market size in 2025; ambulatory surgical centers are advancing at an 11.05% CAGR through 2031.
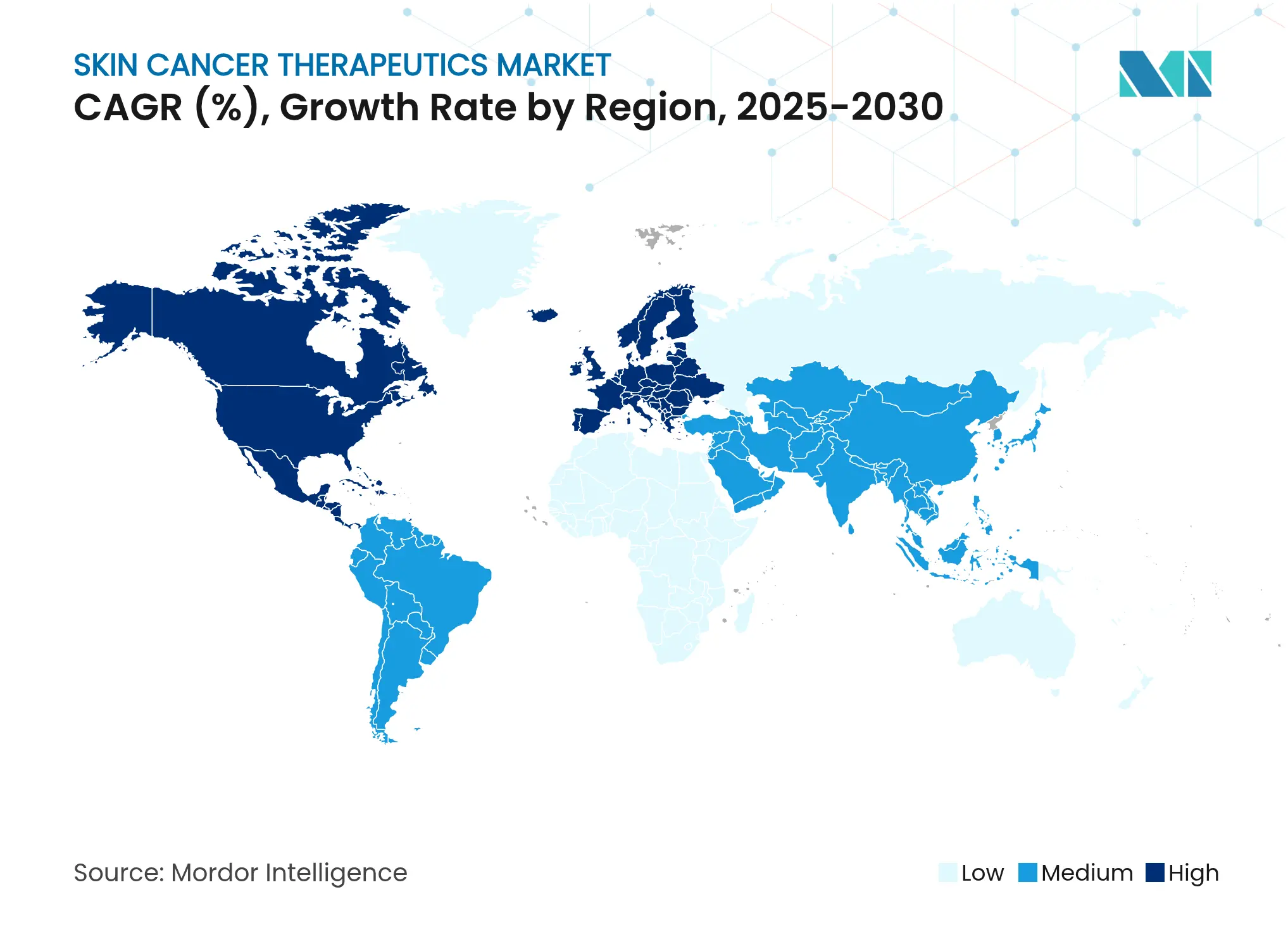
- By geography, North America commanded a 39.72% share in 2025, while Asia Pacific is set to climb at a 10.1% CAGR to 2031.
Global Skin Cancer Therapeutics Market Trends and Insights
*Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline | |||
|---|---|---|---|---|---|---|
Increasing Incidence of Skin Cancer Increasing Incidence of Skin Cancer | +1.20% | Global; highest influence in Australia and North America | Long term (≥ 4 years) | (~) % Impact on CAGR Forecast:+1.20% | Geographic Relevance:Global; highest influence in Australia and North America | Impact Timeline:Long term (≥ 4 years) |
Growing Approvals of Immune-Oncology Drugs Growing Approvals of Immune-Oncology Drugs | +2.90% | North America and EU; extending to Asia Pacific | Medium term (2-4 years) | |||
Expansion of Targeted BRAF/MEK Inhibitor Therapies Expansion of Targeted BRAF/MEK Inhibitor Therapies | +1.10% | North America, EU, and Asia Pacific precision-medicine hubs | Medium term (2-4 years) | |||
Rising Adoption of Hedgehog Pathway Inhibitors in Advanced BCC Rising Adoption of Hedgehog Pathway Inhibitors in Advanced BCC | +1.20% | North America and EU; uptake beginning in high-income APAC | Short term (≤ 2 years) | |||
Emergence of Personalized Neoantigen Cell Therapies Emergence of Personalized Neoantigen Cell Therapies | +1.40% | United States and Western Europe; pilot programs in Japan | Medium term (2-4 years) | |||
Breakthrough Therapy Designations for Topical Oncolytic Patches Breakthrough Therapy Designations for Topical Oncolytic Patches | +1.60% | North America and EU dermatology centers | Short term (≤ 2 years) | |||
| Source: Mordor Intelligence | ||||||
Increasing Incidence of Skin Cancer
Melanoma cases rose 42% from 2015 to 2025, confirming that aging populations, lifestyle shifts, and environmental pollutants jointly heighten disease burden.[2]U.S. Food and Drug Administration, “Subcutaneous Nivolumab Approved for Solid Tumors,” fda.gov Urban pollution hotspots show unexpectedly high incidence, hinting that airborne carcinogens add to ultraviolet exposure risks. Earlier detection through high-resolution imaging increases case counts yet improves survival, sustaining demand for both diagnostics and therapy. Emerging economies now mirror Western sun-exposure patterns, assuring that the skin cancer therapeutics market will keep expanding even as prevention campaigns intensify.
Growing Approvals of Immune-Oncology Drugs
Regulators accelerate access to novel agents. In 2024, the FDA cleared subcutaneous nivolumab for all solid tumors, the first PD-1 inhibitor delivered outside an infusion suite.[3]U.S. Food and Drug Administration, “Subcutaneous Nivolumab Approved for Solid Tumors,” fda.gov China’s nod for toripalimab as frontline melanoma therapy illustrates regulatory harmonization with Western standards. The ten-year survival of 43% for the nivolumab–ipilimumab doublet sustains premium pricing. Broader indications and simplified dosing support continued uptake across the skin cancer therapeutics market.
Rapid Uptake of Combination mRNA–IO Vaccines
Merck and Moderna’s mRNA-4157, combined with pembrolizumab, cut recurrence risk by 49% and distant metastasis by 62% at nearly three-year follow-up. BioNTech’s BNT111 produced meaningful responses in PD-1-refractory patients, extending benefit to previously untreatable populations. Manufacturing personalization requires robust cold chains and rapid sequencing, but FDA breakthrough designation speeds U.S. review. Platform versatility hints at multi-cancer applications, positioning mRNA technology as a future growth engine for the skin cancer therapeutics market.
Emergence of Personalized Neoantigen Cell Therapies
Personalized neoantigen cell therapies bring precision medicine and immunotherapy together in a single, patient-specific treatment for advanced skin cancer. In February 2024, the FDA cleared Amtagvi (lifileucel), the first tumor-infiltrating lymphocyte therapy for a solid tumor, after it delivered a 31.5% objective response rate in heavily pre-treated melanoma patients. The approach involves harvesting a patient’s own immune cells, expanding the cells in a laboratory, then reinfusing them so they can target that individual’s cancer mutations. Building these “living drugs” demands sophisticated facilities and highly trained staff, which both limit new market entrants and support premium pricing that currently exceeds USD 100,000 per course of therapy.
*Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline | |||
|---|---|---|---|---|---|---|
High Therapy & Device Capital Cost High Therapy & Device Capital Cost | -0.70% | Global; acute in emerging markets | Long term (≥ 4 years) | (~) % Impact on CAGR Forecast:-0.70% | Geographic Relevance:Global; acute in emerging markets | Impact Timeline:Long term (≥ 4 years) |
Severe Immune-Related Adverse Events Severe Immune-Related Adverse Events | -0.50% | Global; regulatory focus in developed markets | Medium term (2-4 years) | |||
Global Shortage of Trained Dermatologists Global Shortage of Trained Dermatologists | -0.40% | Worldwide; most acute in rural regions | Long term (≥ 4 years) | |||
AI-Algorithm Racial Bias & Data-Privacy Gaps AI-Algorithm Racial Bias & Data-Privacy Gaps | -0.30% | Global; under strict scrutiny in the United States & EU | Short term (≤ 2 years) | |||
| Source: Mordor Intelligence | ||||||
High Therapy & Device Capital Cost
Checkpoint inhibitors exceed USD 23,000 per dose, while tumor-infiltrating lymphocyte therapy can run USD 100,000 per course. MOH's surgery costs climbed even as U.S. Medicare reimbursement fell 46% in real terms from 2007 to 2024. In low-income nations, these prices dwarf annual health budgets, limiting adoption and dragging on the skin cancer therapeutics market. Practice consolidation creates operational savings but can raise patient fees as investors seek returns.
Severe Immune-Related Adverse Events
Combination regimens trigger grade-3/4 toxicities in nearly half of treated patients, including pneumonitis and colitis that demand expensive management. Community oncologists need new training, and hospitals must staff rapid-response teams. Biomarker-based selection mitigates risk but amplifies diagnostic costs. Progress on predictive tests is essential to balance efficacy with safety in the skin cancer therapeutics market.
Segment Analysis
By Disease Type: Melanoma Drives Premium Value Creation
Melanoma controlled 47.68% of the skin cancer therapeutics market in 2025, a commanding position given that the tumor accounts for only 1% of overall cases. Premium drug prices and multidisciplinary management underpin that share. Combination nivolumab–ipilimumab costs exceed USD 100,000 per patient year, reinforcing revenue concentration. Rare entities such as Merkel cell carcinoma are forecast to rise to 13.02% CAGR as diagnostic awareness improves and targeted agents enter the pipeline. Basal cell carcinoma maintains growth through sheer volume, while cutaneous squamous cell carcinoma gains from newly approved antibodies that post 47.4% response rates.
Precision-biopsy tests shrink unnecessary surgery. Castle Biosciences’ DecisionDx-Melanoma guides sentinel-node decisions, cutting morbidity without impairing control. Neoadjuvant therapy trials and mRNA vaccines widen the treatable population to earlier stages. Collectively, these advances protect melanoma’s revenue importance even as other subtypes accelerate.
By Treatment Modality: Immunotherapy Dominance Faces mRNA Disruption
Immunotherapy contributed 41.92% of 2025 revenue, yet the fastest-growing modality, like mRNA-immunotherapy combinations, will expand at 21.2% CAGR to 2031. Surgery retains a core role, now enhanced by AI-guided imaging that sharpens margin control. Near-infrared photo-immunotherapy blends optical precision with immune activation, lessening collateral tissue damage. Targeted therapy uptake grows as biomarker panels refine patient eligibility, while chemotherapy use retreats.
Algorithm-driven treatment sequencing matches agent choice to evolving tumor biology, cutting unnecessary toxicity. This dynamic keeps the skin cancer therapeutics market fluid, with each modality’s competitive weight shifting as supporting evidence matures.
By End-user: Hospitals Lead While ASCs Accelerate
Hospitals generated 46.25% revenue in 2025, fueled by advanced infusion suites and intensive-care backup. Yet ambulatory surgical centers (ASCs) are growing quickly at an 11.05% CAGR, propelled by minimally invasive devices and payer incentives for lower-cost sites. Dermatology clinics extend their reach with teledermatology triage, while cancer centers focus on high-complexity immunotherapy.
ASCs leverage lean staffing and rapid turnover to lower per-procedure cost, drawing routine excisions and even some Mohs surgeries out of hospitals. Therapeutics in primary care feed ASCs with confirmed cases, making outpatient settings an increasingly important node in the skin cancer therapeutics market.
Geography Analysis
North America retained a 39.72% share in 2025 through early technology adoption, dense specialist networks, and favorable reimbursement. Europe follows with harmonized rules and universal-care funding mechanisms.
Asia Pacific records the fastest 10.1% CAGR: China’s NMPA accepted toripalimab for first-line melanoma, and Japan is pioneering boron neutron capture therapy, treating more than 500 patients to date, nature.com.
The Middle East and Africa rely on medical-tourism corridors, while South America benefits from rising public-health budgets. Supply chains are localizing: Chinese contract manufacturers now produce PD-1 antibodies, and Indian device firms supply dermoscopy units. These shifts highlight regional self-sufficiency trends within the skin cancer therapeutics market, even as global companies drive technology transfer alliances.
Competitive Landscape
Market Concentration
Market structure is moderately fragmented but tilting toward consolidation. Bristol Myers Squibb, Merck, and Pfizer remain central, anchored by broad immunotherapy portfolios and globally distributed trial networks.
Partnerships redefine boundaries: Moderna and Merck co-develop personalized mRNA vaccines, while Sanofi placed EUR 300 million with Orano Med to access alpha-emitting radioligands. Private-equity funds now back 10-15% of dermatology practices, integrating diagnostics, surgery, and infusion under a single operating umbrella.
Disruptors focus on precision and automation. Castle Biosciences posted 51% revenue growth on molecular tests that address a USD 540 million U.S. opportunity. AI platform vendors secure FDA clearances yet face commercialization risk, as shown by DermTech’s 2024 Chapter 11 filing despite differentiated technology. Competitive advantage increasingly hinges on digital pathology, robotics, and genome-informed therapy design. These assets will decide future share in the skin cancer therapeutics market.
Skin Cancer Therapeutics Industry Leaders

*Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- June 2025: Newcastle University scientists reported that adding the anti-CD30 agent brentuximab vedotin to anti-PD-1 therapy lifted median overall survival by 24% in patients with metastatic melanoma who had failed earlier immunotherapy, offering a hopeful option for a hard-to-treat population.
- May 2025: Dubai-based start-up Medicus secured approval from the UAE Department of Health to launch a basal-cell-carcinoma study, highlighting the Middle East’s growing role in cutting-edge skin-cancer research.
- April 2025: The FDA cleared Sanofi and Regeneron’s Dupixent for a broader range of skin disorders, demonstrating how a blockbuster biologic can extend its reach well beyond atopic dermatitis.
- December 2024: : The FDA authorized subcutaneous nivolumab for all of its current solid-tumour indications, the first time a PD-1 inhibitor can be given outside the infusion chair—an advance expected to ease pressure on oncology clinics while preserving a 24.2% response rate.
Table of Contents for Skin Cancer Therapeutics Industry Report
1. Introduction
- 1.1Study Assumptions & Market Definition
- 1.2Scope of the Study
2. Research Methodology
3. Executive Summary
4. Market Landscape
- 4.1Market Overview
- 4.2Market Drivers
- 4.2.1Increasing Incidence of Skin Cancer
- 4.2.2Growing Approvals of Immune-Oncology Drugs
- 4.2.3Expansion of Targeted BRAF/MEK Inhibitor Therapies
- 4.2.4Rising Adoption of Hedgehog Pathway Inhibitors in Advanced BCC
- 4.2.5Emergence of Personalized Neoantigen Cell Therapies
- 4.2.6Breakthrough Therapy Designations For Topical Oncolytic Patches
- 4.3Market Restraints
- 4.3.1High Therapy & Device Capital Cost
- 4.3.2Severe Immune-Related Adverse Events
- 4.3.3Global Shortage Of Trained Dermatologists
- 4.3.4AI-Algorithm Racial Bias & Data-Privacy Gaps
- 4.4Supply-Chain Analysis
- 4.5Regulatory Landscape
- 4.6Technological Outlook
- 4.7Porter's Five Forces Analysis
- 4.7.1Threat of New Entrants
- 4.7.2Bargaining Power of Buyers
- 4.7.3Bargaining Power of Suppliers
- 4.7.4Threat of Substitutes
- 4.7.5Competitive Rivalry
5. Market Size & Growth Forecasts (Value, USD)
- 5.1By Disease Type
- 5.1.1Melanoma
- 5.1.2Basal Cell Carcinoma
- 5.1.3Cutaneous Squamous Cell Carcinoma
- 5.1.4Other Rare Skin Cancers
- 5.2By Treatment Modality
- 5.2.1Surgery
- 5.2.2Chemotherapy
- 5.2.3Immunotherapy
- 5.2.4Targeted Therapy
- 5.2.5Photodynamic Therapy
- 5.2.6Others
- 5.3By End-user
- 5.3.1Hospitals
- 5.3.2Dermatology Clinics
- 5.3.3Cancer Centers
- 5.3.4Ambulatory Surgical Centers
- 5.3.5Research & Academic Institutes
- 5.4By Geography
- 5.4.1North America
- 5.4.1.1United States
- 5.4.1.2Canada
- 5.4.1.3Mexico
- 5.4.2Europe
- 5.4.2.1Germany
- 5.4.2.2United Kingdom
- 5.4.2.3France
- 5.4.2.4Italy
- 5.4.2.5Spain
- 5.4.2.6Rest of Europe
- 5.4.3Asia-Pacific
- 5.4.3.1China
- 5.4.3.2Japan
- 5.4.3.3India
- 5.4.3.4South Korea
- 5.4.3.5Australia
- 5.4.3.6Rest of Asia-Pacific
- 5.4.4Middle East and Africa
- 5.4.4.1GCC
- 5.4.4.2South Africa
- 5.4.4.3Rest of Middle East and Africa
- 5.4.5South America
- 5.4.5.1Brazil
- 5.4.5.2Argentina
- 5.4.5.3Rest of South America
6. Competitive Landscape
- 6.1Market Concentration
- 6.2Market Share Analysis
- 6.3Company Profiles (includes Global level Overview, Market level overview, Core Segments, Financials as available, Strategic Information, Market Rank/Share for key companies, Products & Services, and Recent Developments)
- 6.3.1Amgen
- 6.3.2Pfizer
- 6.3.3Bristol-Myers Squibb
- 6.3.4Iovance Biotherapeutics
- 6.3.5Sanofi
- 6.3.6Merck & Co.
- 6.3.7Novartis
- 6.3.8Regeneron
- 6.3.9Sun Pharma
- 6.3.10Evaxion Biotech
- 6.3.11InxMed
- 6.3.12MediWound
- 6.3.13Roche
- 6.3.14AstraZeneca
- 6.3.15Moderna
- 6.3.16Castle Biosciences
- 6.3.17DermTech
- 6.3.18Squaremind
- 6.3.19Medicus Pharma
- 6.3.20Canfield Scientific
- 6.3.21Leica Microsystems
7. Market Opportunities & Future Outlook
- 7.1White-space & Unmet-Need Assessment
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Segmentation Overview
- By Disease Type
- Melanoma
- Basal Cell Carcinoma
- Cutaneous Squamous Cell Carcinoma
- Other Rare Skin Cancers
- Melanoma
- By Treatment Modality
- Surgery
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Photodynamic Therapy
- Others
- Surgery
- By End-user
- Hospitals
- Dermatology Clinics
- Cancer Centers
- Ambulatory Surgical Centers
- Research & Academic Institutes
- Hospitals
- By Geography
- North America
- United States
- Canada
- Mexico
- United States
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Germany
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia-Pacific
- China
- Middle East and Africa
- GCC
- South Africa
- Rest of Middle East and Africa
- GCC
- South America
- Brazil
- Argentina
- Rest of South America
- Brazil
- North America
Detailed Research Methodology and Data Validation
Primary Research
Desk Research
Market-Sizing & Forecasting
Data Validation & Update Cycle
Why Mordor's Skin Cancer Therapeutics Baseline Commands Reliability
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver | ||
|---|---|---|---|---|
USD 14.54 Bn (2025) | Mordor Intelligence | Anonymized source:Mordor Intelligence | Primary gap driver: | |
USD 10.30 Bn (2025) | Global Consultancy A | counts only small-molecule drugs and omits biologics that dominate first-line therapy | ||
USD 11.10 Bn (2024) | Trade Journal B | rolls forward historic sales without adjusting for accelerating immunotherapy uptake | ||
USD 10.61 Bn (2024) | Regional Consultancy C | excludes emerging Asia-Pacific markets and applies fixed 2023 exchange rates |