Single-use Bioreactor Market Size and Share
Single-use Bioreactor Market Analysis by Mordor Intelligence
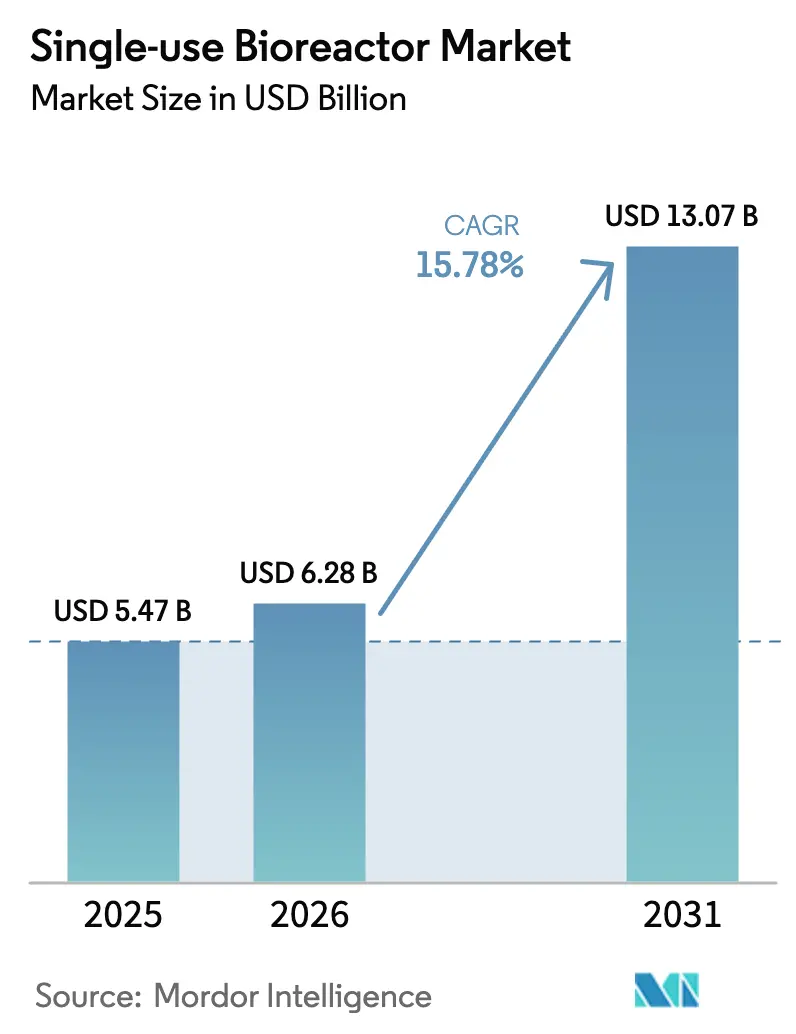
The Single-use Bioreactor Market size is projected to be USD 5.47 billion in 2025, USD 6.28 billion in 2026, and reach USD 13.07 billion by 2031, growing at a CAGR of 15.78% from 2026 to 2031.
Momentum rests on the shift from stainless-steel suites toward disposable platforms, the maturation of cell and gene therapies, and national vaccine-preparedness mandates. Mid-scale stirred-tank systems dominate installed capacity, yet rapid growth is visible in orbitally-shaken and wave-induced formats that support flexible, small-batch runs for autologous treatments.[1]Danaher Corporation, “2024 Annual Report,” Danaher Investor Relations, danaher.com Contract development and manufacturing organizations (CDMOs) are accelerating purchases as they lock in multi-year consumable revenue streams, while sovereign manufacturing initiatives in China, South Korea, and India favor modular, capital-light facilities. PFAS scrutiny and leachables compliance add cost and timeline risk, but expanded gamma-irradiation capacity and regulatory support for advanced manufacturing temper these headwinds.[2]U.S. Food and Drug Administration, “Advanced Manufacturing Technologies Designation Program,” U.S. Food and Drug Administration, fda.gov
Key Report Takeaways
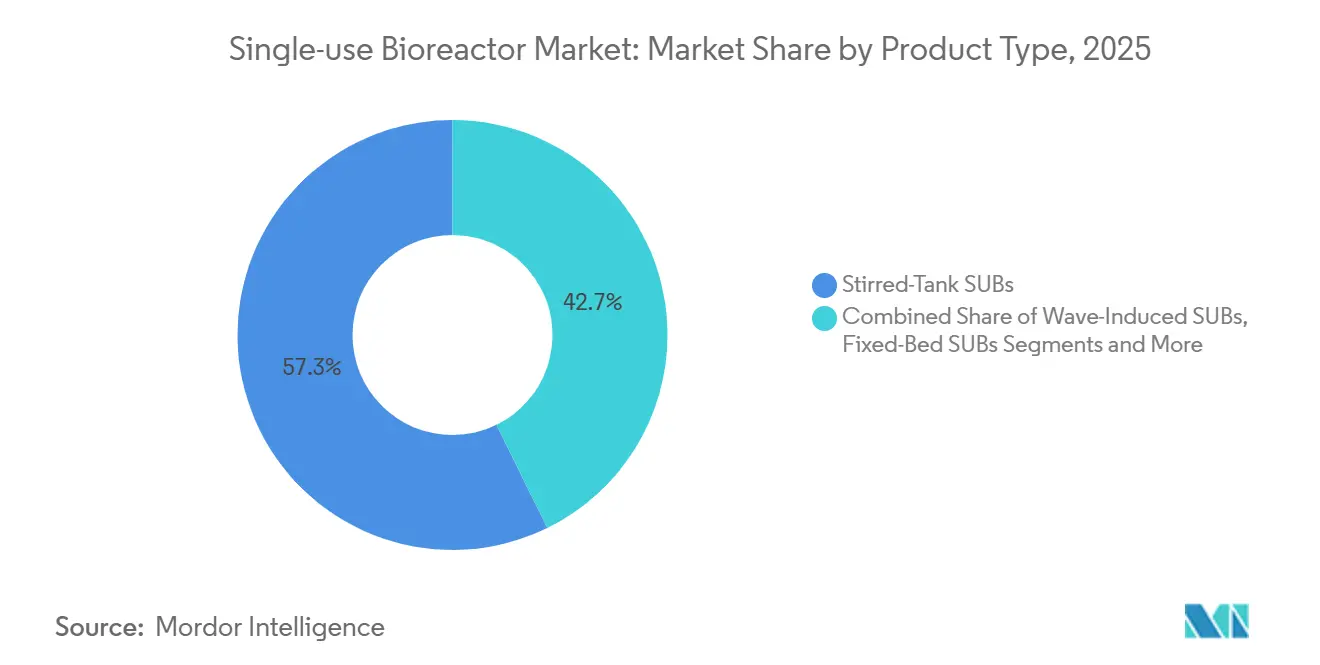
- By product type, stirred-tank systems held 57.26% of 2025 revenue, while orbitally-shaken and other niche formats are forecast to advance at an 18.68% CAGR through 2031.
- By volume range, 500–2,000 L vessels accounted for 43.72% of the single-use bioreactor market size in 2025, whereas sub-100 L units are projected to expand at a 19.42% CAGR over 2026-2031.
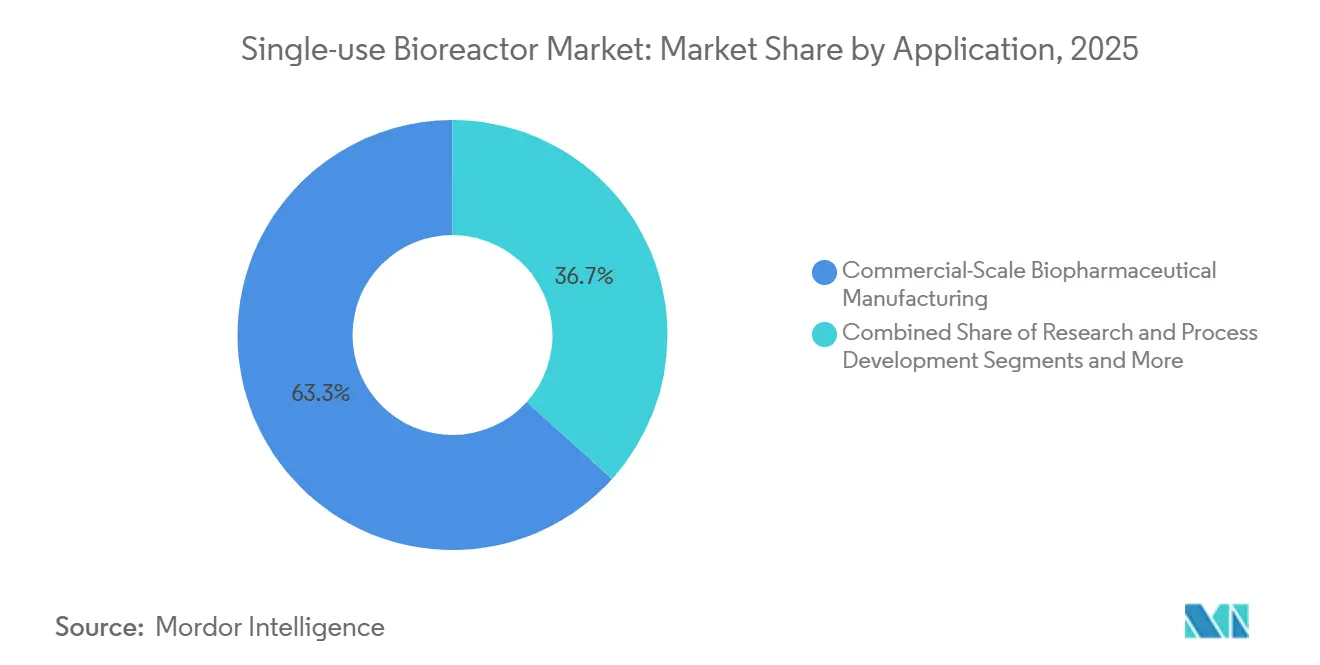
- By application, commercial-scale manufacturing commanded 63.33% of the 2025 total, while clinical-scale production is expected to post an 18.34% CAGR to 2031.
- By end-user, pharmaceutical and biopharmaceutical companies captured 69.21% of 2025 demand, whereas CDMOs/CMOs are anticipated to grow at a 17.53% CAGR during the forecast window.
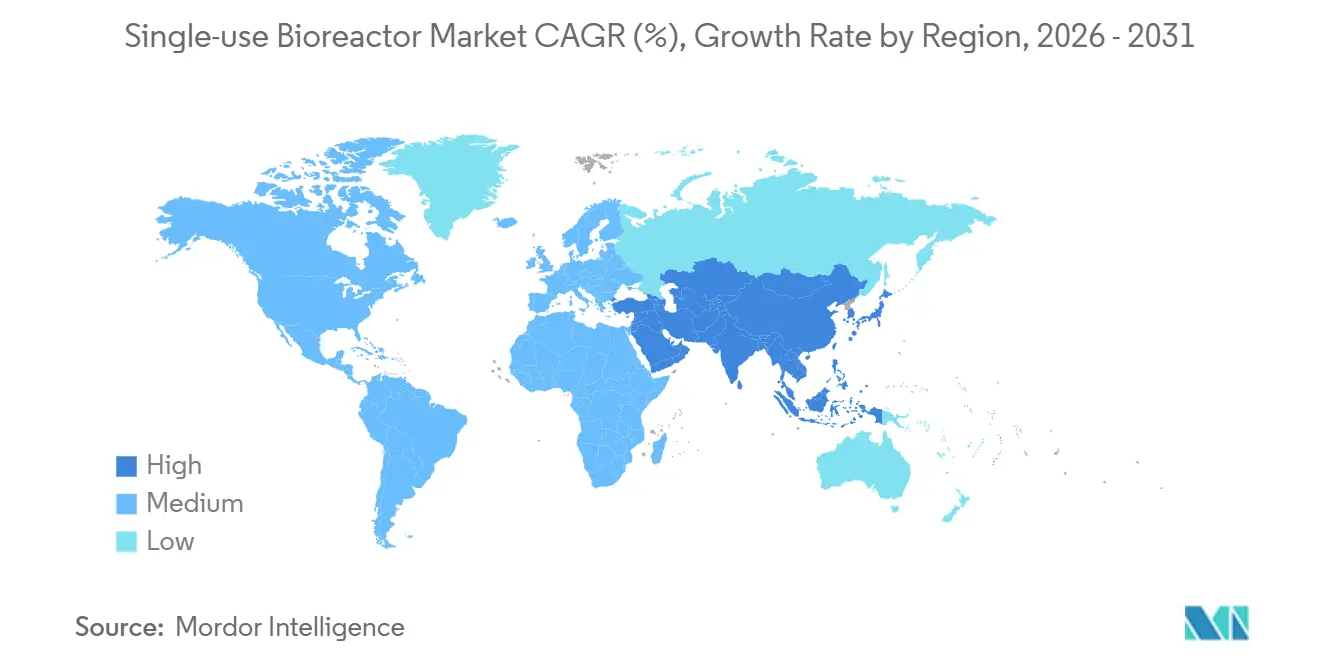
- By geography, North America led with 36.11% revenue share in 2025, and Asia-Pacific is projected to register the fastest growth at a 17.02% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Single-use Bioreactor Market Trends and Insights
Driver Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Shift from stainless-steel to disposable bioprocessing | +2.5% | North America, Europe, Global | Long term (≥ 4 years) |
| Rapid growth of cell & gene therapies | +3.2% | North America, Europe, Asia-Pacific | Medium term (2–4 years) |
| Cost-saving potential in early-stage clinical production | +1.8% | Global | Short term (≤ 2 years) |
| Accelerated vaccine programs post-COVID-19 | +2.1% | Global | Medium term (2–4 years) |
| Decentralized micro-facility models in emerging markets | +1.5% | Asia-Pacific, Middle East & Africa, South America | Long term (≥ 4 years) |
| Expanded γ-irradiation capacity | +1.2% | North America, Europe | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Intensifying Shift From Stainless-Steel to Disposable Bioprocessing Systems
Pharmaceutical manufacturers are retiring legacy stainless trains in favor of single-use platforms that remove cleaning validation, lower water use by up to 80%, and cut turnaround cycles from weeks to days. Danaher’s Cytiva division reported resurgent disposable-consumable sales in late 2024, underscoring dependence on bag assemblies and sensors. Sartorius indicated 75% of Bioprocess Solutions revenue stems from consumables, highlighting customer lock-in once validation is complete. Labor costs for steaming and documentation also tilt economics toward pre-sterilized bags in Western markets, although Indian and Chinese biosimilar firms still favor hybrid plants because import tariffs inflate consumable costs.
Rapid Growth of Cell & Gene Therapies Elevating Demand for Flexible Small-Batch Manufacturing
Autologous CAR-T treatments require 50 mL-10 L runs, volumes incompatible with 500 L stainless reactors. Bristol Myers Squibb reserved USD 380 million in capacity with Cellares in 2024 for its automated Cell Shuttle platform that pairs single-use bioreactors with closed cell-processing hardware.[3] Bristol Myers Squibb, “Capacity Reservation Agreement With Cellares,” Bristol Myers Squibb, bms.com FDA approvals for multiple CAR-T constructs in 2024-2025 and the agency’s Advanced Manufacturing Technologies pathway spur decentralized manufacturing near oncology centers. Univercells’ 50 L NevoLine system can be shipped in a container to remote clinics, reinforcing modular deployments.
Cost-Saving Potential in Early-Stage Clinical Production
Phase I/II campaigns often need 10-50 kg of drug substance. A single-use line eliminates the USD 5-15 million capex for stainless equipment and shortens build times by up to 60%. Thermo Fisher’s 5 L DynaDrive SUB, launched in 2025, lets universities deploy GMP-ready reactors without hiring cleaning-validation staff. Utility savings compound in Europe where wastewater rules add treatment costs, yet at ≥200 annual batches stainless steel regains a cost edge, explaining ongoing hybrid adoption.
Accelerated Vaccine Programs Post-COVID-19
Governments now require domestic capacity able to deliver 100 million doses within 100 days of pathogen sequencing. WHO technology-transfer hubs in South Africa, Argentina, and Bangladesh use modular single-use trains that pivot between strains within weeks. Merck KGaA’s 50-200 L Mobius ADC reactor supports potent payloads while addressing vaccine and oncology overlap.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Leachables & extractables compliance uncertainty | -1.5% | Global | Medium term (2–4 years) |
| High recurring consumable costs | -2.0% | Global | Long term (≥ 4 years) |
| PFAS regulatory scrutiny on single-use plastics | -0.8% | North America, Europe | Medium term (2–4 years) |
| Limited scalability beyond 6,000 L | -1.3% | Global | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Leachables & Extractables Compliance Uncertainty
Multilayer polyethylene films may leach plasticizers that delay approvals. FDA and EMA require up to 18 months of studies costing USD 0.5-1 million per assembly. High-potency products heighten risk, prompting some Japanese sponsors to retain stainless tanks for commercial lots.
High Recurring Consumable Costs
A 2,000 L bag costs USD 3,000-8,000. Facilities running 50 batches spend USD 150,000-400,000 yearly before downstream consumables. Biosimilar makers in India, China, and Brazil struggle to protect margins when ex-factory prices undercut USD 100/g.
Segment Analysis
By Product Type: Stirred-Tank Dominance Meets Niche Innovation
Stirred-tank systems commanded 57.26% of 2025 revenue, reinforcing their role as the workhorse for mammalian cell culture. The single-use bioreactor market size for stirred-tank units is sustained by antibody titers exceeding 5 g/L. Orbitally-shaken vessels are expanding at 18.68% annually on adoption by academic labs, while wave-induced designs serve seed-train and viral-vector work. PBS Biotech’s vertical-wheel approach improves oxygen transfer with lower shear. Thermo Fisher’s sensor-embedded DynaDrive extends digital monitoring into benchtop volumes.
Adoption patterns reflect validation stringency. Commercial antibody makers value historical data from stirred tanks, but early-stage gene therapy teams favor orbitals that fit inside biosafety cabinets. Fixed-bed reactors fill perfusion niches, while the single-use bioreactor industry continues to test hybrid perfusion/fed-batch models that reduce media use.

By Volume Range: Mid-Scale Anchors, Micro-Scale Surges
The 500-2,000 L band represented 43.72% of 2025 revenue and remains the core of CDMO offerings. The single-use bioreactor market share for this range aligns with Phase III batch sizes that yield 5-10 kg per run. Danaher’s 500 L and 2,000 L X-platform models shorten installation to eight weeks, accelerating client onboarding.
Sub-100 L systems are forecast to grow at 19.42% as CAR-T makers and universities deploy benchtop tools. Conversely, designs above 2,000 L face engineering limits; experimental 4,000 L bags remain scarce and expensive, capping growth despite demand for large-scale biosimilars.
By Application: Commercial Manufacturing Anchors, Clinical Scale Accelerates
Commercial-scale manufacturing captured 63.33% of 2025 usage, fueled by marketed monoclonal antibodies. The single-use bioreactor market size for clinical production, however, is rising quickly as sponsors race through Phase I/II studies using disposable lines that eliminate cleaning downtime. Research and process-development labs now migrate from shake flasks to 5-50 L reactors with cloud-linked analytics, compressing tech-transfer timelines.
Clinical programs in cell and gene therapy drive flexible footprints. Univercells’ containerized 50 L units deliver GMP capacity to remote sites, echoing a broader pivot toward decentralized trials. Commercial antibody makers still lean on hybrid fleets because consumable costs climb when annual production rises above 300 kg.

By End-User: Pharma Giants Lead, CDMOs Surge
Pharma and biopharma firms held 69.21% of demand in 2025, leveraging global networks exceeding 100,000 L of single-use capacity. CDMOs, expanding at 17.53%, win contracts as sponsors outsource to conserve cash. Bristol Myers Squibb’s USD 380 million pact with Cellares typifies long-term capacity reservations that anchor the single-use bioreactor market. Academic institutes remain small yet influential early adopters of 5-10 L systems that support translational research.
Large pharma negotiates volume discounts and co-develops sensors, deepening supplier lock-in. CDMOs face heavier cross-contamination oversight because they run multiproduct facilities, but ISO 13408 and FDA guidelines provide a maturing framework.
Geography Analysis
North America accounted for 36.11% of 2025 revenue, buoyed by U.S. headquarters and an FDA program that expedites advanced manufacturing reviews. Growth is plateauing as capacity saturates, prompting suppliers to pivot to Asia-Pacific, projected to grow at 17.02% through 2031. Thermo Fisher’s 2025 design centers in Singapore and Shanghai reflect this tilt.
Europe ranks second, led by Germany, Switzerland, and the UK, where water-usage rules encourage disposable systems. Merck KGaA’s EUR 300 million Darmstadt hub and Sartorius’ consumable-centric model illustrate the focus on high-margin supplies. Post-Brexit dual approvals add modest complexity, yet mutual recognition eases trade.
Asia-Pacific governments pursue sovereign production. South Korea’s Samsung Biologics signed USD 3.3 billion in contracts in 2024, and China’s Suzhou cluster scales single-use suites for domestic biosimilars. India’s Serum Institute uses disposable lines to meet WHO prequalification for African supply.
Middle East & Africa and South America nurture containerized facilities. WHO hubs in South Africa and Argentina rely on modular reactors, and Brazil’s Farmanguinhos uses single-use equipment to localize vaccine output. The United Arab Emirates’ biotech free-zones mirror these moves, signaling continued diffusion.
Competitive Landscape
The single-use bioreactor market is moderately to highly concentrated. Danaher’s Xcellerex expansion in 2025 delivers pre-validated 500 L and 2,000 L units that cut qualification from a year to two months. Thermo Fisher’s acquisition of Solventum filtration and the 5 L DynaDrive launch combine geographic and product diversification. Sartorius’ 75% consumables revenue underscores sticky customer relationships.
Niche innovators attack white-space. PBS Biotech’s vertical-wheel design reduces shear and energy use, useful in perfusion research. Univercells’ NevoLine enables shipping-container factories adopted in WHO hubs, meeting emerging-market self-sufficiency goals. Cellares weaves robotics and AI into closed CAR-T workflows, securing major pharma contracts.
Regulatory incentives matter: FDA’s Advanced Manufacturing Technologies pathway offers speed for perfusion and continuous models, potentially accelerating uptake of disruptive configurations.
Single-use Bioreactor Industry Leaders
-
Sartorius AG
-
Thermo Fisher Scientific, Inc.
-
Danaher (Cytiva & Pall)
-
Merck KGaA
-
Eppendorf SE
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: Fujifilm Biotechnologies opened a GBP 400 million single-use facility in Teesside, the largest of its kind in the UK.
- April 2025: Thermo Fisher launched the 5 L DynaDrive SUB, providing seamless scalability from 1 L to 5,000 L for rapid bench-to-commercial transitions.
- March 2025: Danaher broadened its Xcellerex X-platform to 500 L and 2,000 L, halving installation times for CDMOs.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the global single-use bioreactor (SUB) market as the annual revenue generated from disposable, pre-sterilized bioreactor vessels (<=6,000 L) and their dedicated control hardware supplied for cell-culture or fermentation campaigns in biopharmaceutical R&D, clinical, and commercial production environments.
Scope Exclusion: Traditional glass or stainless-steel multi-use reactors and generic single-use fluid-handling bags are not counted.
Segmentation Overview
-
By Product Type
- Stirred-Tank SUBs
- Wave-Induced SUBs
- Fixed-Bed SUBs
- Orbitally-Shaken & Other SUBs
-
By Volume Range
- Less than 100 L
- 100 – 500 L
- 500 – 2 000 L
- More than 2,000 L
-
By Application
- Research & Process Development
- Clinical-Scale Production
- Commercial-Scale Biopharmaceutical Manufacturing
-
By End-User
- Pharmaceutical & Biopharmaceutical Companies
- Contract Development & Manufacturing Organisations (CDMOs/CMOs)
- Academic & Research Institutes
-
By Geography
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Rest of Europe
-
Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia-Pacific
-
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
-
South America
- Brazil
- Argentina
- Rest of South America
-
North America
Detailed Research Methodology and Data Validation
Primary Research
Mordor analysts next held structured calls with bioprocess engineers at vaccine makers, global CDMOs, filter film suppliers, and regional regulators across North America, Europe, and Asia. These interviews clarified average selling prices, usable lifespan assumptions, and the realistic ceiling for single-use working volumes. This then guided variable selection and sanity checks.
Desk Research
We began by mapping the installed base and shipment flows using open datasets such as FDA Biologic License Application files, European Medicines Agency product lists, UN Comtrade HS-9021 export codes for bioprocess equipment, and trade association briefs from BioPlan Associates and BPOG. Market ratios were enriched with company 10-Ks, investor decks, and peer-reviewed papers on SUB adoption economics. Where public detail thinned, analysts accessed D&B Hoovers for private-firm revenue breakouts and Dow Jones Factiva for deal pipelines. This combination gave our team a grounded starting lattice; many additional sources were tapped as cross-checks during model refinement.
Market-Sizing & Forecasting
A top-down capacity-utilization model converts biologics production volumes (liters of cell-culture capacity by scale) into SUB demand. Then, results are corroborated through bottom-up samples of vendor shipments and typical ASP × volume by scale band. Key inputs include: 1) commercial-scale biologics pipeline expansions, 2) penetration rates of SUBs versus steel reactors, 3) average batch sizes in mAbs and vaccines, 4) regional cap-ex incentives for fast-track plants, and 5) unit ASP erosion as volumes climb. Multivariate regression with scenario analysis is applied to forecast 2025-2030 trajectories; gaps in supplier roll-ups are bridged with imputed volumes from import data and primary-source price bands.
Data Validation & Update Cycle
Outputs run through variance screens against historical adoption curves; anomalies trigger analyst review and, if required, re-contact of earlier respondents. Reports refresh yearly, with mid-cycle updates when material events (large capacity additions, regulatory shifts) occur.
Why Mordor's Single-use Bioreactor Baseline Commands Reliability
Published numbers often diverge because firms bundle upstream disposables, apply different ASP ladders, or extrapolate short-run survey data.
Key Gap Drivers: 1) Some studies merge SUBs with single-use bags and mixers, inflating totals. 2) Others annualize aggressive >18% growth seen in venture-backed CDMOs without tempering for regulatory lag. 3) Refresh cycles vary; Mordor's annual rewrites capture new 2,000 L+ launches that older datasets miss.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 4.74 B (2025) | Mordor Intelligence | - |
| USD 5.27 B (2025) | Global Consultancy A | Includes media bags & filtration assemblies inside reactor total |
| USD 3.90 B (2024) | Trade Journal B | Uses conservative pipeline count and excludes SUBs above 2,000 L |
Taken together, these contrasts show that Mordor's disciplined scope and multi-step validation deliver a balanced, transparent baseline that decision-makers can trace back to clear variables and repeatable steps.


Key Questions Answered in the Report
What is the projected value of the c in 2031?
It is forecast to reach USD 13.07 billion by 2031.
Which volume range currently leads sales?
Vessels sized 500-2,000 L captured 43.72% of 2025 revenue.
Why are CDMOs investing heavily in disposable equipment?
Multi-year capacity-reservation deals lock in recurring consumable revenue while sparing sponsors from large capex.
What regulatory program supports advanced manufacturing adoption in the United States?
FDA’s Advanced Manufacturing Technologies Designation Program expedites reviews for qualified innovations.
Which region will grow fastest through 2031?
Asia-Pacific is projected to expand at a 17.02% CAGR on the back of national biomanufacturing mandates.
What main factor restrains commercial-scale uptake?
High recurring consumable costs push sponsors toward stainless-steel once annual batches exceed 200-300.
Page last updated on:









