Pharmaceutical Contract Development And Manufacturing Organization (CDMO) Market Size and Share
Pharmaceutical Contract Development And Manufacturing Organization (CDMO) Market Analysis by Mordor Intelligence
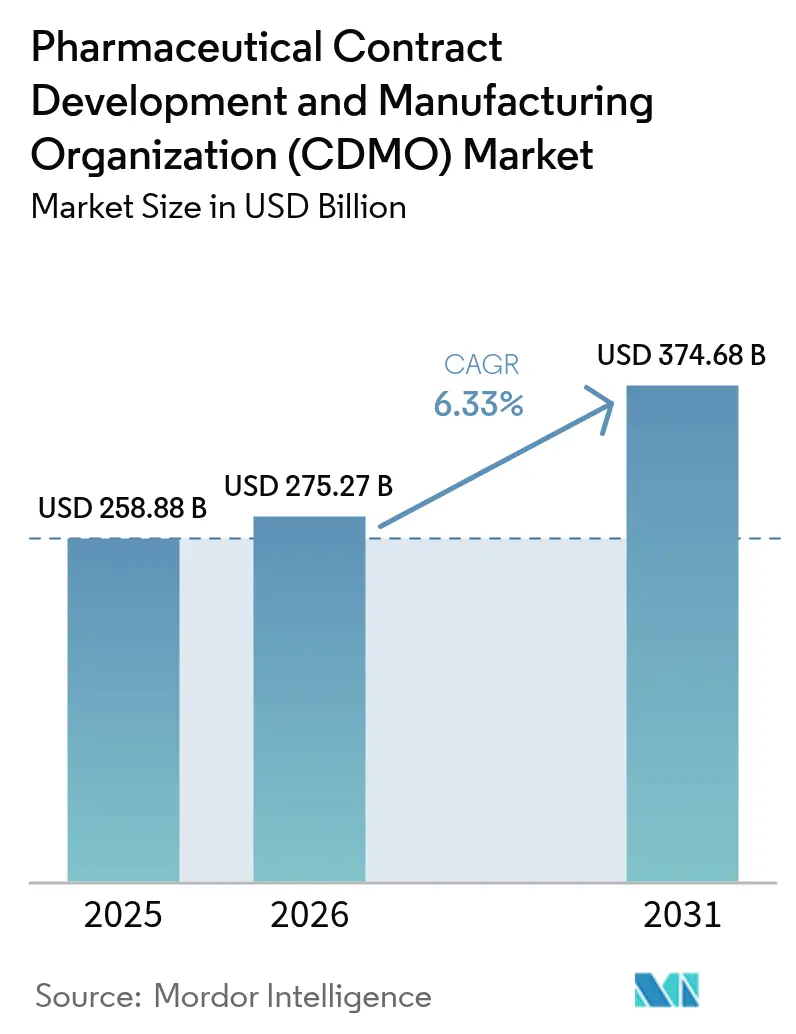
The Pharmaceutical contract development and manufacturing organization (CDMO) market size is expected to grow from USD 258.88 billion in 2025 to USD 275.27 billion in 2026 and is forecast to reach USD 374.68 billion by 2031 at 6.33% CAGR over 2026-2031. Robust outsourcing demand for complex biologics, the rise of high-potency APIs (HPAPIs), and artificial-intelligence-enabled–enabled process-development platforms underpin this trajectory. Peptide-based GLP-1 therapies, expanding vaccine programs, and sustained investment in digitally connected plants further amplify the need for specialist partners able to absorb capital and regulatory risks. North American innovators continue to anchor high-value biologics and gene-therapy work, while Asia-Pacific cost advantages accelerate capacity expansion. Consolidation—typified by Novo Holdings’ USD 16.5 billion purchase of Catalent—signals a decisive shift toward end-to-end providers that combine development, scale-up, and commercial production.
Key Report Takeaways
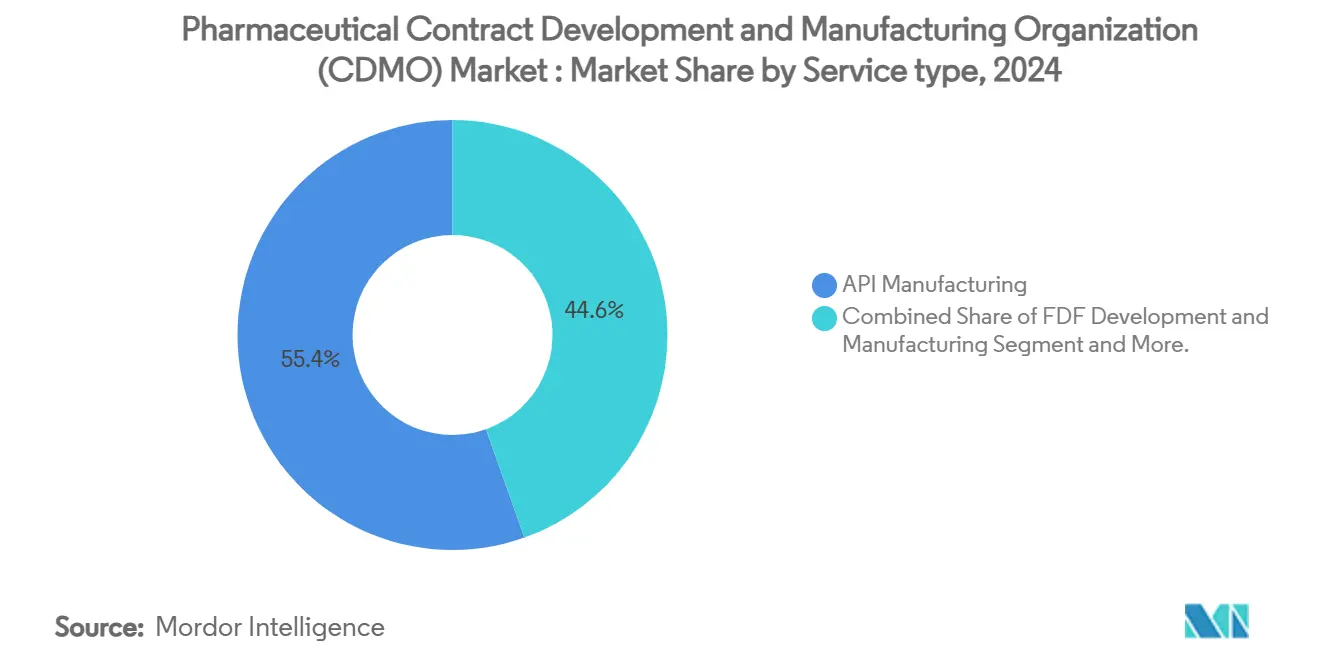
- By service type, API manufacturing held 54.92% of the Pharmaceutical contract development and manufacturing organization (CDMO) market share in 2025, while finished-dosage-form (FDF) development and manufacturing is forecast to expand at 7.18% CAGR through 2031.
- By molecule type, small-molecule APIs captured 61.70% of the Pharmaceutical contract development and manufacturing organization (CDMO) market size in 2025; HPAPIs are projected to grow at an 8.05% CAGR to 2031.
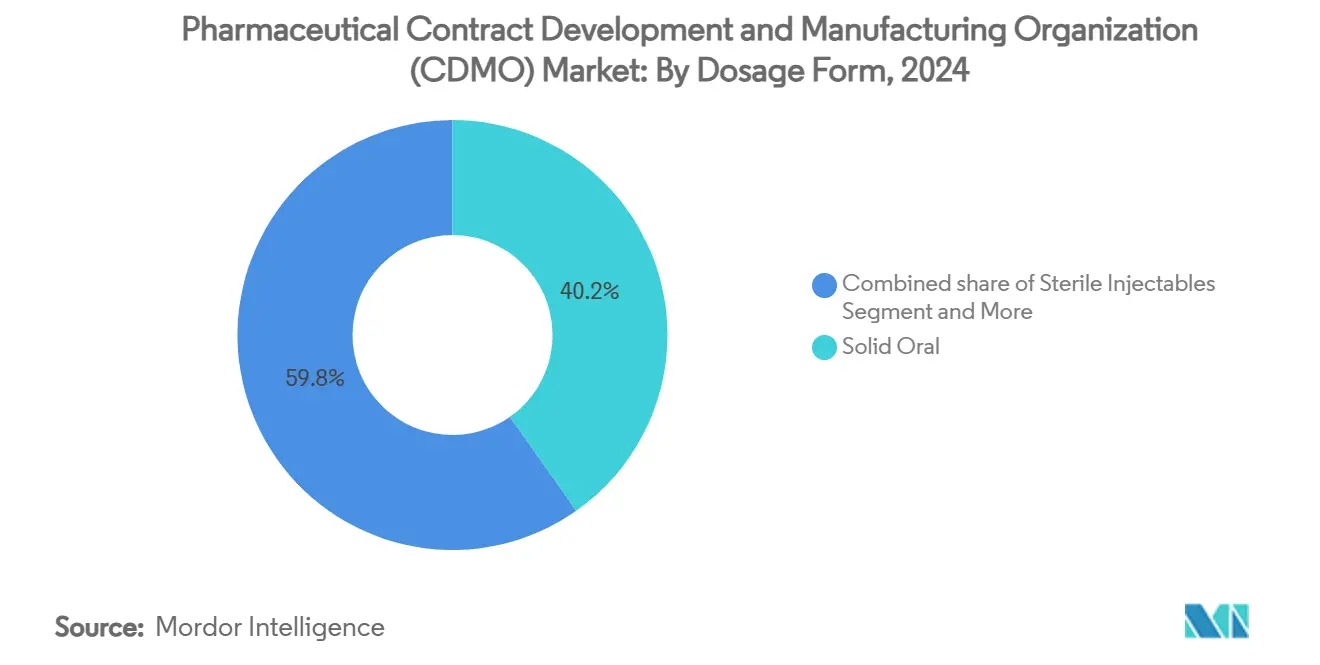
- By dosage form, solid orals accounted for 39.88% share of the Pharmaceutical contract development and manufacturing organization (CDMO) market size in 2025, whereas sterile injectables are advancing at 9.05% CAGR.
- By therapeutic area, oncology commanded 32.15% revenue share in 2025, while infectious-disease and vaccine projects post the fastest 8.16% CAGR outlook.
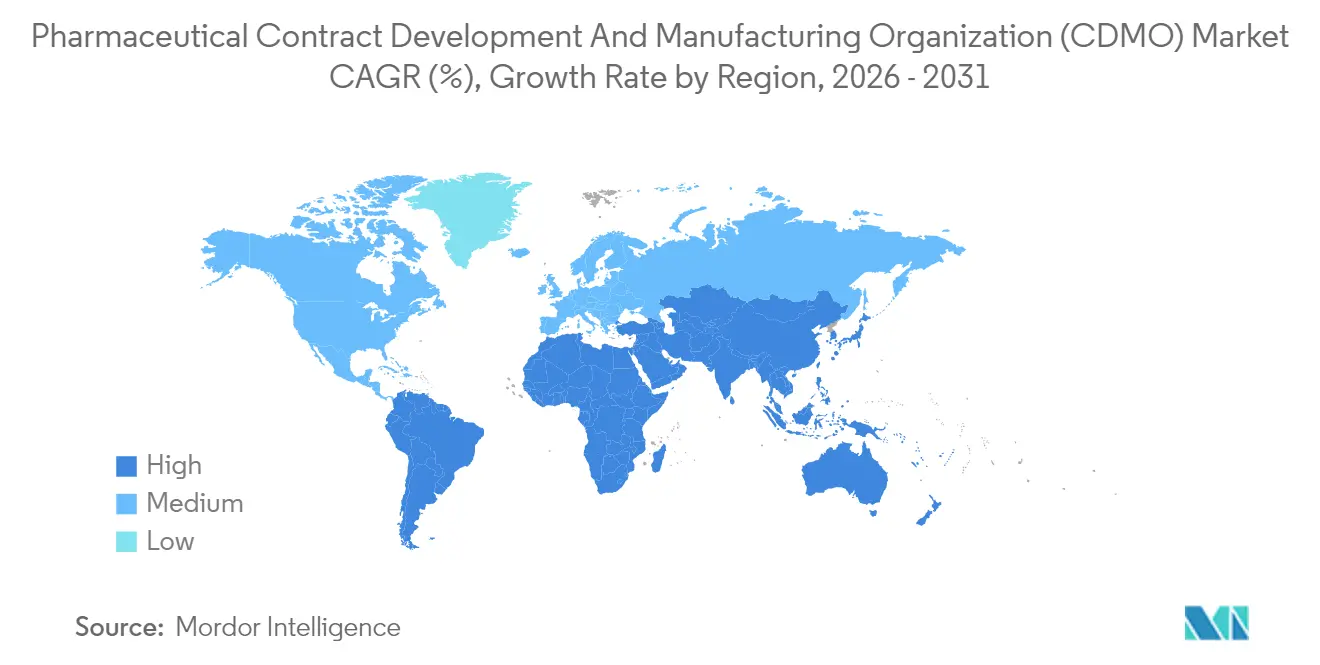
- By geography, North America led with 37.95% of the Pharmaceutical contract development and manufacturing organization (CDMO) market share in 2025; Asia-Pacific records the highest 7.18% CAGR projection.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Pharmaceutical Contract Development And Manufacturing Organization (CDMO) Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) %Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Increasing outsourcing volume by large pharmaceutical companies | +1.8% | North America, Europe | Medium term (2-4 years) |
| Surge in biologics and complex-molecule pipelines | +1.5% | North America, EU; APAC emerging | Long term (≥ 4 years) |
| Cost and speed advantage of manufacturing in emerging markets | +1.2% | Core APAC; MEA & South America spill-over | Short term (≤ 2 years) |
| Consolidation toward one-stop CDMOs | +0.9% | Global | Medium term (2-4 years) |
| AI-enabled rapid process-development platforms | +0.7% | North America, Europe; APAC expanding | Medium term (2-4 years) |
| GLP-1 and peptide HPAPI capacity build-outs | +0.6% | North America, Europe | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Increasing Outsourcing Volume by Large Pharmaceutical Companies
Escalating R&D costs and pipeline complexity drive pharmaceutical majors to off-load non-core manufacturing. Asset-light models free capital for discovery while leveraging CDMO expertise to maintain global supply continuity. Lonza’s USD 1.2 billion Vacaville site purchase from Roche underpins this transition, adding 330,000 L of biologics capacity to support blockbuster antibody demand. Outsourcing is most intense for sterile biologics and gene-editing therapies, where regulatory rigor and technical barriers heighten the value of specialist partners.
Surge in Biologics and Complex-Molecule Pipelines
Biological entities now dominate new-drug filings, propelled by antibody-drug conjugates, mRNA vaccines, and cell-based therapeutics. Samsung Biologics secured USD 1.4 billion in new 2024 contracts and is expanding antibody-drug-conjugate suites, illustrating spiraling demand for cGMP biologics supply [1]Samsung Biologics, “Samsung Biologics Financial Results 2024,” samsung.com Source: Economic Times, “India CDMO market to reach $22-25 billion by 2035: Report,” economictimes.indiatimes.com . Biologics’ stringent cold-chain, contamination-control, and analytics requirements solidify a preference for full-scope CDMOs with proven regulatory track records.
Cost and Speed Advantage of Manufacturing in Emerging Markets
Regional incentives, labor arbitrage, and faster approvals underpin APAC’s ascent. India targets a USD 22–25 billion Pharmaceutical contract development and manufacturing organization (CDMO) market size by 2035, backed by streamlined environmental clearances and tax holidays. Brazil and Saudi Arabia likewise channel public funds into diabetes-drug and vaccine plants to foster local resilience.
Consolidation Toward One-Stop CDMOs
Drug sponsors shave months off timelines by handing a molecule to a single partner from pre-clinical tox through global launch. Novo Holdings’ 2024 Catalent takeover fused development, viral-vector, and fill-finish expertise under one roof. Larger platforms exploit shared quality systems and digital twins to de-risk tech transfer.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Stringent multi-region regulatory requirements | -0.8% | North America, EU | Long term (≥ 4 years) |
| Capacity-utilization and lead-time risk | -0.6% | Global, biologics acute | Medium term (2-4 years) |
| High capex for sterile biologics suites | -0.5% | Developed markets | Medium term (2-4 years) |
| Scarcity of skilled aseptic-manufacturing talent | -0.4% | North America, Europe | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Stringent Multi-Region Regulatory Requirements
Divergent dossiers and rolling updates such as the European Medicines Agency’s new fee rules raise compliance budgets and prolong variation lead times EMA. CDMOs must operate duplicate quality-management systems and align data-integrity protocols across FDA, EMA, and PMDA audits, challenging smaller entrants.
High Capex for Sterile Biologics Suites
Each Class A/B cell-culture wing commands USD 100 million plus validation, locking outsized capital for up to five years. Lonza earmarked CHF 500 million to refurbish Vacaville’s fermentation halls into next-generation antibody production, underscoring significant fixed-cost hurdles. Limited suppliers of single-use bioreactors and isolators compound procurement delays.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Service Type: API Manufacturing Remains the Anchor
Active Pharmaceutical Ingredient (API) Manufacturing is the largest segment in the Pharmaceutical Contract Development and Manufacturing Organization (CDMO) market, holding a significant market share of approximately 68% in 2023. This segment is pivotal due to its role in the production of essential components for pharmaceutical drugs, which are critical for the healthcare industry. The dominance of this segment is driven by the increasing demand for APIs, particularly for small molecule drugs, which constitute a major portion of the pharmaceutical market. Technological advancements in API synthesis and the growing trend of outsourcing API production to specialized CDMOs have further bolstered this segment's growth. Additionally, the rising prevalence of chronic diseases and the need for innovative therapies have amplified the demand for high-quality APIs. The segment also benefits from stringent regulatory requirements, which encourage pharmaceutical companies to partner with experienced CDMOs for API manufacturing. Overall, the API manufacturing segment remains a cornerstone of the CDMO market, driving innovation and ensuring the availability of critical pharmaceutical ingredients.

By Molecule Type: Small Molecules Hold Scale; HPAPIs Accelerate
Small molecules captured 62.34% of 2024 value, aided by mature regulatory pathways and broad therapeutic applicability. Reaction-efficiency gains and green-chemistry mandates encourage hybrid batch-continuous plants that drive cost competitiveness. However, oncology’s migration toward antibody-drug conjugates, selective degrader molecules, and micro-dosed cytotoxics pushes HPAPIs to an 8.32% CAGR. CDMOs retrofit suites with negative-pressure isolators and closed-handling skids to comply with <1 μg/m³ OEL thresholds.
The Pharmaceutical contract development and manufacturing organization (CDMO) market size devoted to HPAPIs is forecast to double over the next five years as peptide-based GLP-1s and next-gen chemotherapeutics move through Phase III. HPAPI projects typically command 25-30% pricing premiums due to containment and analytical complexity, supporting higher margins for specialized providers.
By Dosage Form: Solid Orals Dominate; Sterile Injectables Surge
Solid oral products retained 40.23% share in 2024. Robust tableting lines, wet-granulation flexibility, and unrivaled patient acceptance anchor this format. Efforts to extend franchise exclusivity via abuse-deterrent coatings and multiparticulate capsules sustain demand in mature markets. Further, fixed-dose combinations for metabolic disease streamline adherence.
Sterile injectables register the fastest 9.32% CAGR on the back of biologics, long-acting antipsychotics, and biosimilar launches. The Pharmaceutical contract development and manufacturing organization (CDMO) market size for sterile fill-finish is projected to eclipse USD 70 billion by 2030 as dual-chamber syringes, auto-injectors, and lyophilized vials gain regulatory approvals. Simtra BioPharma’s USD 250 million expansion highlights capital intensity and the allure of premium parenteral margins[2]Simtra BioPharma, “Sterile Fill-Finish Expansion,” simtra.com.

By Therapeutic Area: Oncology Retains the Lead; Vaccines Rise
Oncology accounted for 32.43% of 2024 CDMO revenue, reflecting high value per gram and constant pipeline replenishment. HPAPI suites, single-use perfusion bioreactors, and conjugation lines support small-batch precision medicines. Demand spikes for checkpoint inhibitors and radioligand therapies keep capacity tight and prices firm.
Infectious-disease and vaccine programs grow at 8.42% CAGR. Government pandemic-preparedness funding exemplified by Moderna’s USD 590 million H5N1 mRNA contract—sustains lipid-nanoparticle-formulation and aseptic-fill investments [3]Moderna, “Moderna Receives $590 Million US Government Contract,” modernatx.com. Expanded adult immunization schedules and antimicrobial-resistance initiatives diversify volumes beyond COVID-19 boosters. Meanwhile, GLP-1–driven endocrine products accelerate metabolic-disorder output, further crowding HPAPI slots.
Geography Analysis
North America retained 37.95% revenue share in 2025, buoyed by premier biologics programs, FDA Orphan-Drug incentives, and a deep venture-capital pool. The United States sustains premium pricing as cGMP compliance costs and stringent data-integrity audits elevate barriers to entry. Canada benefits from free-trade access and skilled resources, whereas Mexico lures secondary packaging and regional solid-oral projects. Thermo Fisher’s USD 4.1 billion filtration-business acquisition reinforces North American vertical-integration strategies.Asia-Pacific logs the fastest 7.18% CAGR through 2031. China and South Korea bankroll mega-plants for mAbs and oligonucleotides, though geopolitical risk nudges US sponsors toward India and Southeast Asia. The Pharmaceutical contract development and manufacturing organization (CDMO) market size in India alone could surpass USD 22 billion by 2035, aided by PLI incentives and harmonized quality standards.Samsung Biologics’ fourth plant, topping 600,000 L, cements Incheon as the world’s largest single-site biologics hub. Australia leverages expedited regulatory pathways for early-phase oncology and cell-therapy trials.
Europe presents steady expansion anchored in quality leadership. Germany’s continuous-manufacturing clusters and the United Kingdom’s advanced-therapy corridor offset Brexit friction through mutual-recognition waivers. The EMA’s updated variation fees raise short-term compliance costs but assure global sponsors of consistent review rigor. Eastern Europe gains traction as an overflow destination for solid-oral generics and secondary packaging. Sustainability regulations incentivize solvent-recovery units and low-energy lyophilization, driving process innovation.
Competitive Landscape
The CDMO market is characterized by a high degree of competition and ongoing consolidation, reflecting current CDMO market trends. Global players are expanding their operations in key regions, while local firms are enhancing their capabilities to compete internationally. Mergers and acquisitions are a common strategy, enabling companies to broaden their service offerings and geographical reach. Investment in advanced manufacturing technologies, such as continuous manufacturing and digital integration, is also prevalent, driving CDMO industry growth.
Niche disruptors leverage continuous flow, micro-reactor scale-out, and on-demand formulation to serve precision-medicine pipelines with batch sizes in the tens of grams. Capacity shortages in HPAPI and viral-vector fill-finish create price elasticity that rewards early movers. Competitive intensity will heighten as digital-native entrants compress tech-transfer cycles and as large pharma refines multi-sourcing frameworks to mitigate geopolitical risk.
Pharmaceutical Contract Development And Manufacturing Organization (CDMO) Industry Leaders
Catalent Inc.
Recipharm AB
Jubilant Pharmova Ltd
Patheon Inc. (Thermo Fisher Scientific Inc.)
Boehringer Ingelheim Group
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2025: Boehringer Ingelheim and DotBio form a tri-specific-antibody alliance, with Bora Biologics providing cGMP supply.
- January 2025: Boehringer Ingelheim and DotBio form a tri-specific-antibody alliance, with Bora Biologics providing cGMP supply.
- December 2024: Novo Holdings closes USD 16.5 billion Catalent deal, creating the world’s largest CDMO platform.
- November 2024: Avid Bioservices agrees to USD 1.1 billion buyout by GHO Capital and Ampersand, illustrating sustained private-equity appetite
Global Pharmaceutical Contract Development And Manufacturing Organization (CDMO) Market Report Scope
The study tracks and analyzes the demand for outsourcing CMO and CRO activities within the pharmaceutical industry based on the current trends and market dynamics. The market numbers are derived by tracking the revenue generated by the players operating in the market who are providing CRO & CMO services. The study provides a detailed breakdown of various research phases and service types. This report analyzes the factors based on the prevalent base scenarios, key themes, and end-user vertical-related demand cycles.
The pharmaceutical contract development and manufacturing organization market is segmented by service type CMO segment (active pharmaceutical ingredient (API) manufacturing (small molecule, large molecule, and high potency (HPAPI)), finished dosage formulation (FDF) development and manufacturing (solid dose formulation (tablets), liquid dose formulation, injectable dose formulation), secondary packaging), research phase CRO segment (pre-clinical, phase I, phase II, phase III, phase IV), pharmaceutical CMO geography (North America [United States, Canada], Europe [United Kingdom, Germany, France, Italy, and Rest of Europe], Asia-Pacific [China, India, Japan, Australia, and the Rest of Asia-Pacific], Latin America [Brazil, Mexico, Argentina, and the Rest of Latin America], and the Middle East and Africa [United Arab Emirates, Saudi Arabia, South Africa, and the Rest of Middle-East and Africa]), and pharmaceutical CRO geography (North America, Europe, Asia-Pacific, Latin America, and the Middle East and Africa). The market sizes and forecasts are provided in terms of value USD for all the above segments.
| API Manufacturing | Small Molecule |
| Large Molecule | |
| High-Potency (HPAPI) | |
| FDF Development and Manufacturing | Solid Dose |
| Liquid Dose | |
| Injectable Dose | |
| Secondary Packaging |
| Small Molecule |
| Large Molecule (Biologics and Biosimilars) |
| High-Potency APIs |
| Solid Oral |
| Sterile Injectables |
| Topicals and Transdermals |
| Specialty/Novel (e.g., ODT, Long-acting) |
| Oncology |
| Metabolic and Endocrine |
| Cardiovascular |
| CNS and Psychiatry |
| Infectious Diseases and Vaccines |
| North America | United States | |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Russia | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia and New Zealand | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | Middle East | United Arab Emirates |
| Saudi Arabia | ||
| Turkey | ||
| Rest of Middle East | ||
| Africa | South Africa | |
| Nigeria | ||
| Egypt | ||
| Rest of Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
| By Service Type | API Manufacturing | Small Molecule | |
| Large Molecule | |||
| High-Potency (HPAPI) | |||
| FDF Development and Manufacturing | Solid Dose | ||
| Liquid Dose | |||
| Injectable Dose | |||
| Secondary Packaging | |||
| By Molecule Type | Small Molecule | ||
| Large Molecule (Biologics and Biosimilars) | |||
| High-Potency APIs | |||
| By Dosage Form | Solid Oral | ||
| Sterile Injectables | |||
| Topicals and Transdermals | |||
| Specialty/Novel (e.g., ODT, Long-acting) | |||
| By Therapeutic Area | Oncology | ||
| Metabolic and Endocrine | |||
| Cardiovascular | |||
| CNS and Psychiatry | |||
| Infectious Diseases and Vaccines | |||
| By Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Russia | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| India | |||
| Japan | |||
| South Korea | |||
| Australia and New Zealand | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | Middle East | United Arab Emirates | |
| Saudi Arabia | |||
| Turkey | |||
| Rest of Middle East | |||
| Africa | South Africa | ||
| Nigeria | |||
| Egypt | |||
| Rest of Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||


Key Questions Answered in the Report
What is the current size of the pharmaceutical contract development and manufacturing organization (CDMO) market?
The Pharmaceutical CDMO market size stands at USD 275.27 billion in 2026 and is projected to reach USD 374.68 billion by 2031, reflecting a 6.33% CAGR.
Which CDMO service segment generates the greatest revenue today?
API manufacturing leads with 54.92% of 2025 revenue, supported by entrenched small-molecule infrastructure and rising demand for high-potency APIs.
Which region holds the largest market share and which is expanding the quickest?
North America captured 37.95% of 2025 global revenue, while Asia-Pacific records the fastest 7.18% CAGR thanks to cost advantages and large-scale capacity additions.
How are high-potency APIs (HPAPIs) and sterile injectables performing?
HPAPIs expand at an 8.05% CAGR, propelled by oncology and targeted-therapy pipelines, and sterile injectables advance at a 9.05% CAGR on the back of biologics and vaccine demand.
What is the fastest-growing service category?
Finished-dosage-form (FDF) development and manufacturing posts the highest 7.18% CAGR through 2031 as drug sponsors seek patient-centric formats and accelerated life-cycle management.
Page last updated on:






