PCR Molecular Diagnostics Market Size and Share
PCR Molecular Diagnostics Market Analysis by Mordor Intelligence
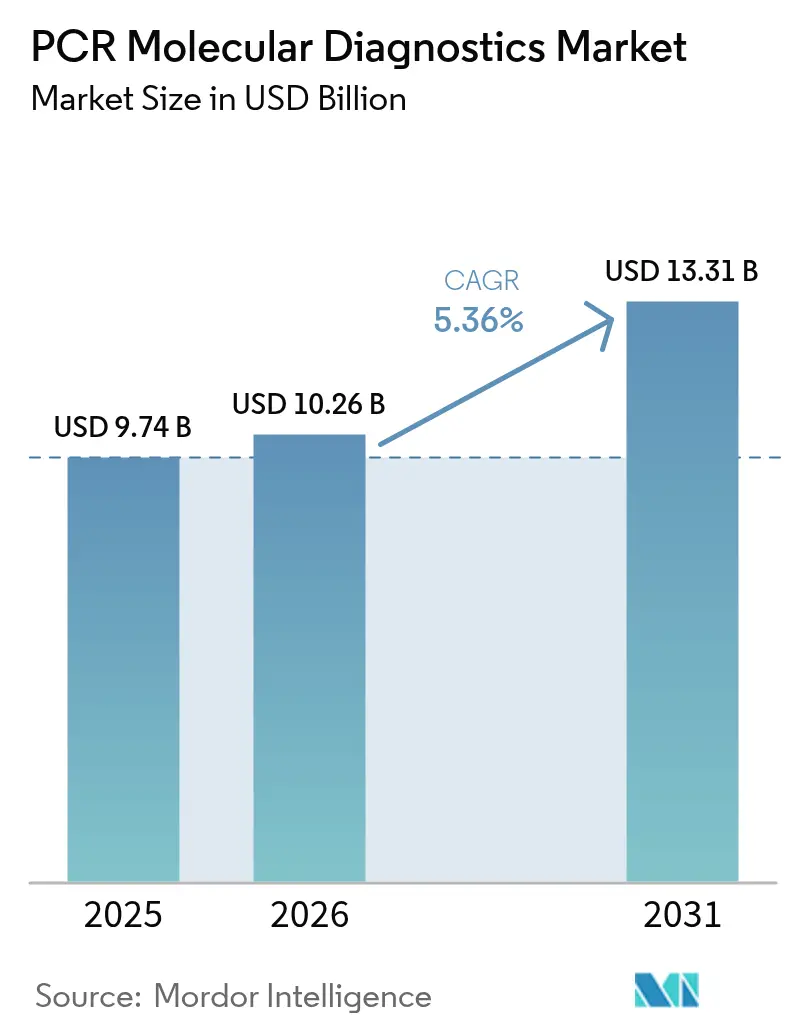
The PCR Molecular Diagnostics Market size was valued at USD 9.74 billion in 2025 and estimated to grow from USD 10.26 billion in 2026 to reach USD 13.31 billion by 2031, at a CAGR of 5.36% during the forecast period (2026-2031).
Reagents and consumables underpin revenue expansion because every amplification run requires fresh chemistry, while demand for digital PCR systems is accelerating thanks to their 0.01% variant-allele detection thresholds that unlock liquid-biopsy and minimal-residual-disease use cases. Hospitals and reference laboratories continue to dominate test volumes, yet miniaturised platforms are pushing sophisticated assays into emergency departments, oncology clinics and community health centres. North America still captures the largest share at 42% on the strength of well-funded laboratories and supportive reimbursement, but Asia-Pacific represents the fastest-growing geography at a 6.54% CAGR as governments scale precision-medicine budgets. Competitive intensity is rising as incumbents such as F. Hoffmann-La Roche and Thermo Fisher Scientific refresh instrument portfolios, while specialist firms turn to AI-guided assay design and cloud-linked workflows to differentiate. Meanwhile, the United States Food and Drug Administration (FDA) is ending decades of enforcement discretion for laboratory-developed tests, adding compliance costs that will catalyse consolidation across the PCR molecular diagnostics market[1]Food and Drug Administration, “Medical Devices; Laboratory Developed Tests – Federal Register,” Federalregister.gov.
Key Report Takeaways
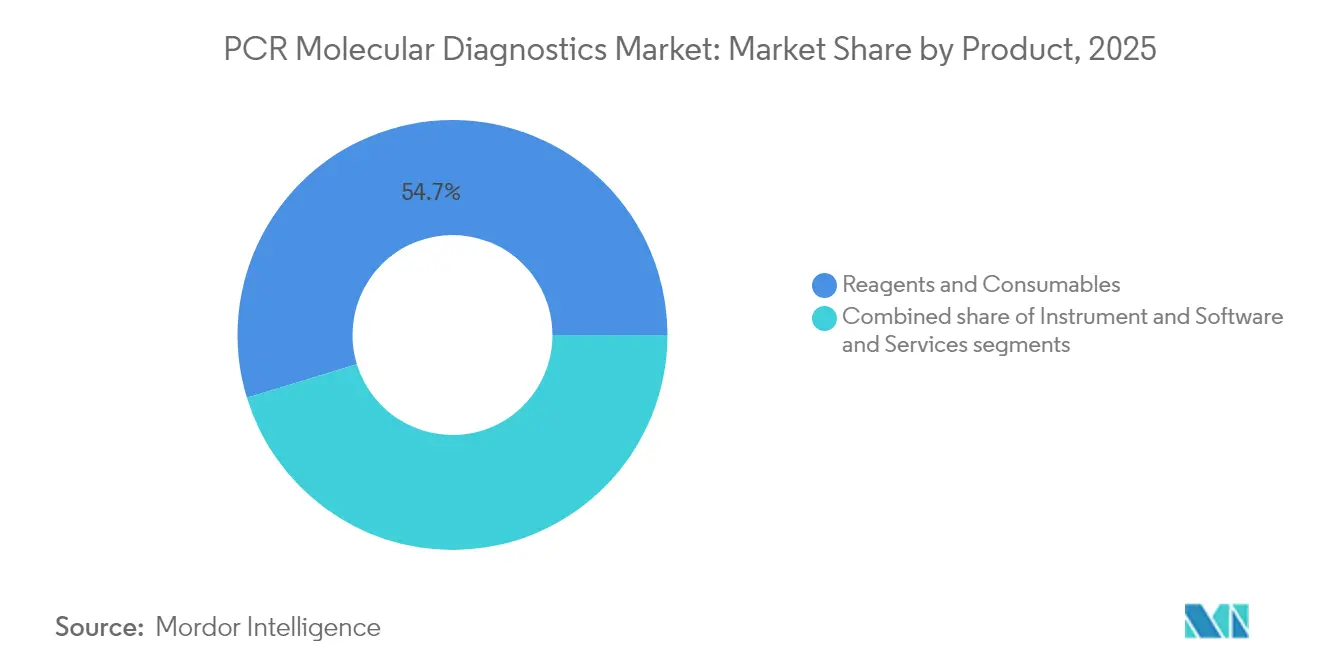
- By product type, reagents and consumables held 54.72% of the PCR molecular diagnostics market share in 2025, whereas digital PCR instruments are on track for the highest 2026-2031 CAGR at 6.86%.
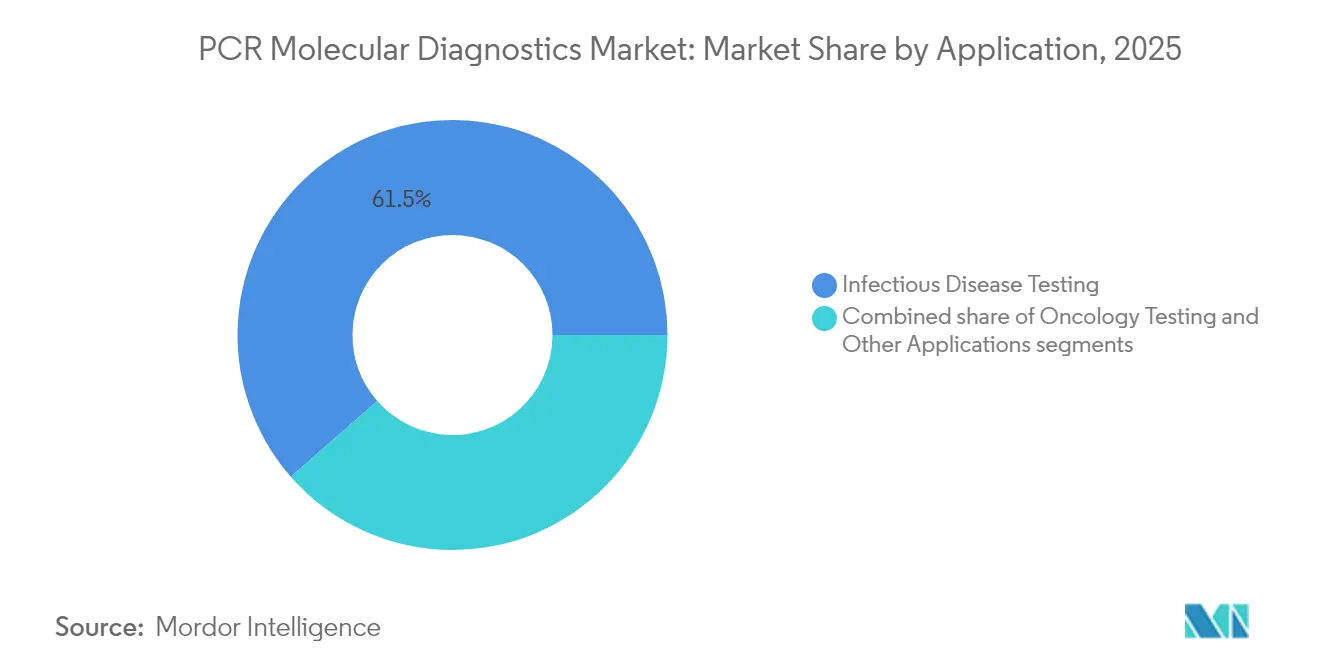
- By application, infectious disease assays led with 61.45% revenue share in 2025; oncology testing is projected to grow at an 7.33% CAGR to 2031.
- By end user, hospitals and reference laboratories contributed 47.35% of the PCR molecular diagnostics market size in 2025, while decentralised point-of-care settings are forecast to post the fastest 7.74% CAGR through 2031.
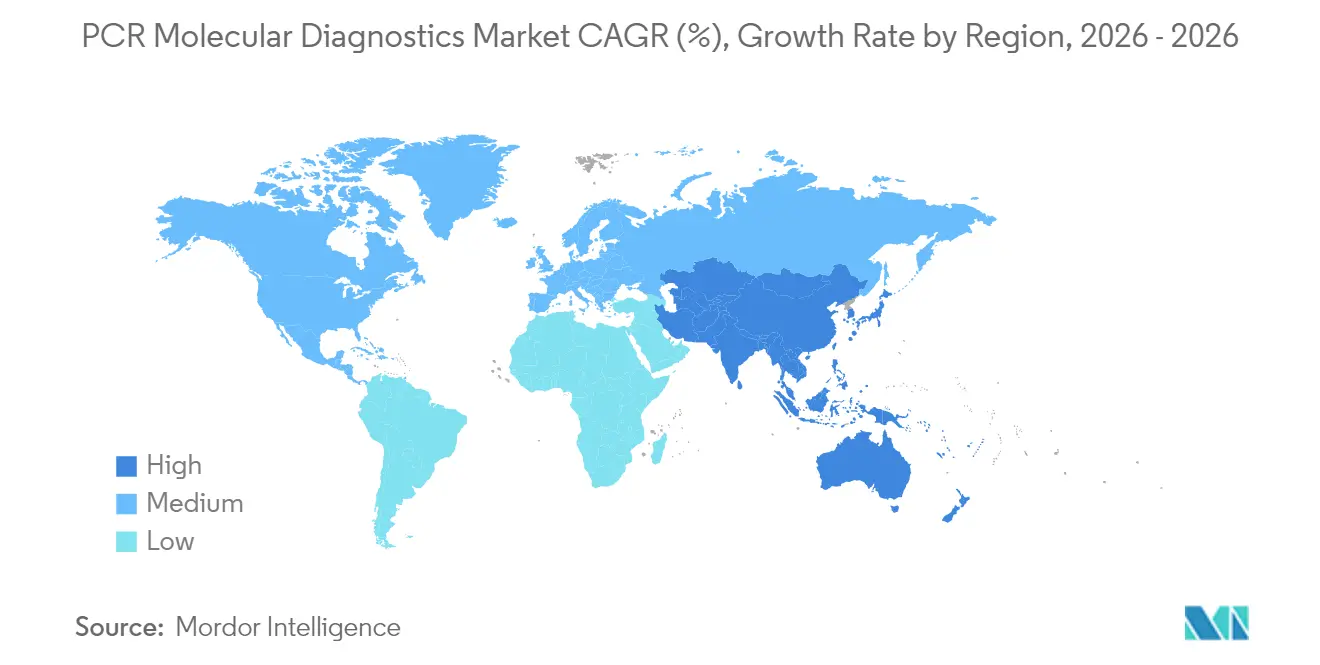
- By geography, North America commanded 41.62% of 2025 revenue; Asia-Pacific is projected to post the quickest 6.27% CAGR between 2026 and 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global PCR Molecular Diagnostics Market Trends and Insights
Driver Impact Analysis*
| Drivers Impact Analysis | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising infectious-disease and cancer incidence | +1.8% | Global, stronger in North America & Asia-Pacific | Medium term (2-4 years) |
| Technological innovation in PCR chemistry & instrumentation | +1.2% | Global, earliest in North America & Europe | Medium term (2-4 years) |
| Adoption of syndromic & multiplex panels | +0.8% | North America & Europe, growing Asia-Pacific uptake | Short term (≤2 years) |
| Decentralised and point-of-care molecular testing | +0.7% | Rural & underserved regions worldwide | Medium term (2-4 years) |
| Government screening programs & public-health funding | +0.6% | North America, Europe, developed Asia-Pacific | Short term (≤2 years) |
| Rising demand for precision medicine & companion diagnostics | +1.1% | North America, Europe, emerging Asia-Pacific | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Infectious-Disease and Cancer Incidence
Escalating global disease burdens anchor demand for molecular assays across the PCR molecular diagnostics market. The American Cancer Society anticipates 2,041,910 new cancer diagnoses and 618,000 deaths in the United States during 2025, heightening the urgency for early detection strategies[2]American Cancer Society, “Cancer Facts & Figures 2025,” Cancer.org. Concurrently, paediatric hospitalisations linked to human metapneumovirus rose 17% versus 2023 across the United States and China, underscoring the need for rapid respiratory-virus differentiation. PCR offers attomolar sensitivity, enabling clinicians to guide antivirals, isolate patients more precisely and monitor minimal residual disease after therapy. Because 40% of new cancer cases are considered preventable through earlier detection, payers and policymakers increasingly treat molecular diagnostics as cost-saving rather than discretionary. The result is sustained throughput growth across centralised and near-patient laboratories within the PCR molecular diagnostics market.
Technological Innovation in PCR Chemistry & Instrumentation
Rapid-cycle enzymes, microfluidics and digitisation are rewriting performance benchmarks within the PCR molecular diagnostics market. Extreme-PCR protocols now compress complete thermocycles into 15 seconds, raising hourly sample capacity without raising instrument footprints. Digital PCR partitions reaction volumes into thousands of nanodroplets, reaching 0.01% variant calls that were once the sole province of next-generation sequencing. AI is becoming integral to quality control; Seegene is co-developing a Digitalized Development System with Microsoft that automates primer design and detects signal anomalies in real time. These advances collectively elevate sensitivity, shorten turn-around-time and lower operator variability, thereby expanding oncology monitoring, transplant surveillance and wastewater-based epidemiology within the PCR molecular diagnostics market.
Adoption of Syndromic and Multiplex Panels
Physicians are shifting from single-pathogen ordering to broad, symptom-centred panels that interrogate dozens of targets simultaneously. The BioFire FilmArray Pneumonia Panel demonstrates 96.3% sensitivity and 97.2% specificity while reducing time-to-result from ≥48 hours for culture down to about one hour. Fast, comprehensive data underpin antibiotic-stewardship programmes because clinicians can de-escalate broad-spectrum therapy once the causative organism is known. Laboratories also trim hands-on-time because multiplex cartridges consolidate extraction, amplification and detection. Point-of-care iterations such as BIOFIRE SPOTFIRE deliver answers in 15 minutes, further integrating syndromic testing into emergency and paediatric settings. As antibiotic resistance intensifies, hospitals use multiplex data to update local antibiograms, strengthening the long-run competitive position of the PCR molecular diagnostics market.
Decentralised and Point-of-Care Molecular Testing
Technological miniaturisation is unbundling diagnostics from core laboratories within the PCR molecular diagnostics market. The First Nations Molecular Point-of-Care Testing Program now reaches 100 remote Australian communities, running SARS-CoV-2, HIV and sexually transmitted infection panels directly in rural clinics. Systems such as GeneXpert and SPOTFIRE integrate sample preparation, thermal cycling and result interpretation inside sealed disposable cartridges, limiting biohazard exposure and training requirements. Policymakers increasingly subsidise such deployments to close healthcare-access gaps and strengthen outbreak surveillance. Cloud connectivity further allows real-time result aggregation for regional situational awareness, an advantage that creates continuous reagent demand across the PCR molecular diagnostics market.
Restraints Impact Analysis*
| Restraints Impact Analysis | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High capital & consumable costs | –1.0% | Global, strongest in emerging economies | Short term (≤2 years) |
| Regulatory complexity & reimbursement uncertainty | –0.9% | Primarily North America; ripple effects worldwide | Short term (≤2 years) |
| Limited availability of skilled workforce | –0.7% | Global, higher impact in rural & underserved regions | Medium term (2-4 years) |
| Competition from isothermal amplification & CRISPR-based diagnostics | –0.6% | Global, higher impact in point-of-care settings | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
High Capital and Consumable Costs
Cutting-edge digital PCR instruments can exceed USD 250,000 per unit, a barrier for district hospitals that operate under tight capital budgets. Ongoing reagent outlays compound the burden because chemistry often represents 55-65% of lifetime testing expenses. Liquid-biopsy panels cost USD 500–3,000 each, limiting routine oncology follow-up outside high-income markets. Although manufacturing scale and open-channel chemistries are nudging prices downward, the near-term reality is a two-tier diagnostic ecosystem where sophisticated assays cluster in major medical centres. Smaller laboratories therefore outsource samples, extending turn-around-times and tempering volume growth in parts of the PCR molecular diagnostics market.
Regulatory Complexity and Reimbursement Uncertainty
The FDA’s July 2024 final rule ends enforcement discretion for laboratory-developed tests, sequencing compliance deadlines over four years. Each assay must now clear quality-system, adverse-event and pre-market review requirements akin to commercial kits, raising time-to-launch for hospital laboratories. Simultaneously, the Protecting Access to Medicare Act continues to ratchet fee-schedule reductions, complicating cost-recovery for innovative assays. Europe is tightening in-vitro diagnostic regulations under IVDR, while divergent policies across Asia create additional harmonisation headaches. The cumulative burden favours well-capitalised firms and accelerates mergers as smaller players seek regulatory expertise, consolidating power within the PCR molecular diagnostics market.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product: Digital PCR Drives Next-Generation Precision
Reagents and consumables produced the largest slice of 2025 revenue at 54.72% because every test run mandates fresh primers, probes and buffers, creating a predictable annuity business model inside the PCR molecular diagnostics market. Digital PCR instruments, though currently a minority in installed base, logged the quickest adoption pace due to absolute quantification and orders-of-magnitude gains in analytical sensitivity. For example, Bio-Rad’s droplet partitioning routinely detects rare alleles down to 0.01%, enabling detection of circulating tumour DNA in a standard blood draw. Standard thermocyclers still populate many regional hospitals thanks to lower acquisition costs, whereas real-time systems occupy a middle tier by offering quantitation without the capital intensity of digital.
Software and analytical services are emerging revenue streams as laboratories wrestle with data lakes generated by high-throughput systems. Roche’s LightCycler PRO integrates automatic calibration and robotics to lift walk-away time, and QIAGEN plans more than 100 new QIAcuity assays by 2028 after obtaining CE mark for its syndromic respiratory menu in late 2024. These developments highlight how engineering and bioinformatics advances continue to extend the value proposition of each instrument generation within the PCR molecular diagnostics market.

By Application: Infectious Disease Testing Maintains Dominance
Infectious-disease assays constituted 61.45% of 2025 turnover because clinicians rely on PCR to differentiate pathogens that share overlapping symptoms yet require distinct therapies. Tuberculosis remains a headline example; rapid molecular detection compresses diagnostic timeframes from weeks on solid culture to hours, informing targeted treatment and limiting transmission. Syndromic respiratory panels have become routine during influenza-like-illness seasons, especially where antimicrobial-resistance surveillance is a public-health priority.
Oncology testing is the fastest-growing application, reflecting expanding use of cell-free DNA assays and minimal-residual-disease monitoring. The Shield liquid-biopsy test, for instance, showed an 83.1% sensitivity for colorectal-cancer detection in peer-reviewed evaluation. As targeted therapies and immuno-oncology regimens proliferate, companion diagnostics based on PCR mutation panels are being co-developed with drugs to identify responders earlier. The move toward tumour-agnostic drug approvals further elevates the role of genomic profiling in directing treatment, sustaining double-digit growth for oncology sub-segments of the PCR molecular diagnostics market size through 2031.

By End User: Diverse Settings Expand Testing Access
Hospitals and reference labs generated 47.35% of 2025 revenue. Their economies of scale allow multishift operation of high-throughput thermocyclers and aligned clinical interpretation teams. Reference lab order volumes climbed alongside outreach testing; NeoGenomics, for example, reported 34% clinical next-generation-sequencing revenue growth in its 2024 filing, signalling appetite for complex molecular work-ups.
Academic and research institutes represent the most rapidly increasing user group because translational projects increasingly spin out as laboratory-developed tests. Meanwhile, decentralised environments—urgent-care centres, oncology infusion suites and mobile clinics—are adopting cartridge-based platforms that preserve analytical rigor yet eliminate complex pipetting. The First Nations program in Australia exemplifies how remote site testing can integrate into electronic health records, shrinking diagnostic deserts and growing sample throughput for all vendors operating within the PCR molecular diagnostics industry.
Geography Analysis
North America generated 41.62% of global revenue in 2025. Federal funding streams reinforce this leadership position; the U.S. Defense Health Program’s FY 2025 budget allocates substantial outlays for precision-medicine and genomic-surveillance projects. Large hospital networks frequently run dual platforms—high-throughput core-lab thermocyclers for routine testing and mobile units for operating rooms—creating layered demand across the PCR molecular diagnostics market size. FDA regulatory tightening is expected to spur lab consolidation but also encourage standardisation, possibly elevating U.S. export competitiveness for cleared kits.
Asia-Pacific is forecast to record a 6.27% CAGR through 2031, the fastest worldwide. Governments in China, India and South Korea earmark expanding precision-medicine budgets, while private laboratories race to meet oncology-panel demand. Japan currently leads regional adoption in oncology-focused testing, holding roughly 41% share of that application niche during 2023. Public-health authorities in Southeast Asia embrace point-of-care PCR for dengue and respiratory outbreaks to compensate for central-lab shortages. This multifaceted growth portfolio positions the region as the most dynamic frontier for the PCR molecular diagnostics market.
Europe maintains robust share on the back of universal-health-coverage frameworks and strong academic-industry R&D consortia. Implementation of the In Vitro Diagnostic Regulation is prompting earlier engagement between manufacturers and notified bodies, lengthening approval timelines yet raising device quality and post-market surveillance. In contrast, Latin America, Middle East and Africa constitute nascent but high-potential territories where decentralised platforms circumvent infrastructural deficits. Pilots deploying GeneXpert units in remote clinics illustrate the impact of real-time tuberculosis confirmation on treatment initiation and antibiotic stewardship, gradually stitching these regions into the global PCR molecular diagnostics market value chain.
Competitive Landscape
Competition blends global scale with niche specialisation. F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific, Inc., and Abbott Laboratories command extensive instrument and reagent portfolios that ensure lock-in across clinical chemistry, immunoassay and molecular disciplines, reinforcing cross-selling synergies. F. Hoffmann-La Roche Ltd. announced a USD 50 billion, five-year U.S. investment programme in April 2025 to enlarge domestic manufacturing, an explicit hedge against supply-chain disruptions.
Mid-sized innovators concentrate on technical differentiation. QIAGEN’s QIAcuity digital PCR platform partitions 96 wells into 26,000 nanoplates, boosting throughput for oncology liquid biopsies, and the firm secured CE mark for its QIAstat-Dx respiratory menu in September 2024. Bio-Rad advances droplet-emulsification IP, while bioMérieux leverages syndromic cartridge know-how to launch SPOTFIRE, a 15-minute respiratory test system targeting urgent-care clinics.
Strategic alliances and acquisitions continue apace. Hologic paid USD 795.0 million for Mobidiag to gain rapid gastrointestinal and sepsis panels, reinforcing its acute-care footprint. Myriad Genetics teamed with PathomIQ in February 2025 to co-develop oncology-focused PCR panels that marry Myriad’s lab network with PathomIQ’s AI analytics. Funding flows into start-ups remain healthy; Deepull Diagnostics raised EUR 50 million in April 2025 to commercialise its UllCore rapid pathogen platform. Those moves illustrate how intellectual-property depth and menu breadth drive bargaining power across the PCR molecular diagnostics market.
PCR Molecular Diagnostics Industry Leaders
Danaher Corp. (Cepheid)
QIAGEN N.V.
Abbott Laboratories
Thermo Fisher Scientific Inc.
F. Hoffmann-La Roche Ltd.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2025: Roche committed to invest USD 50 billion in United States pharmaceutical and diagnostics operations, enlarging domestic manufacturing capacity.
- April 2025: Deepull Diagnostics raised EUR 50 million Series C funding to advance its UllCore rapid pathogen detection system.
- February 2025: Myriad Genetics and PathomIQ entered a collaboration to expand oncology precision diagnostics, pooling bioinformatics expertise for new PCR-based panels.
- February 2025: Sysmex Astrego won a GBP 8 million award to finalise a rapid urinary-tract-infection antibiotic-susceptibility test.
- September 2024: QIAGEN received CE mark for the QIAstat-Dx system, enabling broader European deployment in syndromic testing.
- October 2024: LabGenomics USA acquired Integrated Molecular Diagnostics, strengthening its oncology testing footprint.
- June 2024: QIAGEN outlined a strategic plan targeting 7% compound annual sales growth from 2024 to 2028 with more than 100 future assays for QIAcuity.
- May 2024: George Mason University secured USD 1.2 million from the U.S. Army to develop a urine-based Lyme-disease PCR assay, highlighting military interest in field-deployable tests.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the polymerase chain reaction (PCR) molecular diagnostics market as all revenue generated from instruments, reagents, consumables, and enabling software that amplify and detect human nucleic acid targets for clinical decision-making across hospital, reference, and point-of-care settings. We include standard, real-time, and digital PCR platforms.
We explicitly exclude veterinary, food testing, and research-only PCR workflows from the scope.
Segmentation Overview
- By Product
- Instrument
- Standard PCR Systems
- Real-time PCR Systems
- Digital PCR Systems
- Reagents & Consumables
- Software & Services
- Instrument
- By Application
- Infectious Disease Testing
- Oncology Testing
- Other Applications
- By End User
- Hospitals
- Diagnostic & Reference Laboratories
- Academic & Research Institutes
- Other End Users
- Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia-Pacific
- Middle-East and Africa
- GCC
- South Africa
- Rest of Middle East and Africa
- South America
- Brazil
- Argentina
- Rest of South America
- North America
Detailed Research Methodology and Data Validation
Primary Research
We spoke with lab directors, infectious disease physicians, oncology pathologists, and service engineers across North America, Europe, and Asia. Their insights on run rate volumes, kit reorder cycles, and reimbursement shifts helped us validate secondary estimates and fine-tune cost per test assumptions.
Desk Research
We began by collating open datasets such as United States CMS test utilization files, WHO tuberculosis and HPV surveillance reports, European Centre for Disease Prevention bulletins, and FDA 510(k) IVD clearances, which gave us disease burden, regulatory velocity, and lab adoption signals. Our analysts then reviewed trade body briefs from the Association of Molecular Pathology and the Asian Clinical Microbiology network, plus company 10-Ks and peer-reviewed papers, to triangulate unit shipments and average selling prices. Selected proprietary tools, D&B Hoovers for revenue splits and Questel for PCR patent trends, added further depth. These sources are illustrative; many additional references supported interim checks and clarifications.
Market-Sizing & Forecasting
Our team built a top down model that reconstructs global PCR demand from reportable disease incidence and cancer screening cohorts, multiplied by tests per patient and blended cost per test to reach baseline revenue. Supplier roll ups of instrument installations and reagent pull through served as a bottom up sense check. Key variables like annual TB, HPV, and COVID-19 case counts, installed thermocycler inventory, reagent ASP progression, healthcare spending growth, and new IVD approvals feed a multivariate regression for 2025 to 2030. Regional penetration rates from interviews filled residual data gaps.
Data Validation & Update Cycle
Before sign off, we run variance flags, re contact sources when deviations exceed preset bands, and perform dual analyst reviews. The model refreshes annually, with interim tweaks for material events such as major assay launches or guideline revisions, ensuring clients receive the latest calibrated view.
Why Mordor's PCR Molecular Diagnostics Baseline Is Dependable
Published estimates often differ because firms apply distinct product mixes, base years, and refresh rhythms, creating understandable confusion for planners.
Key gap drivers we observed include whether software and service contracts are counted, how inpatient versus outpatient volumes are treated, and the month chosen for currency conversion.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 9.74 B (2025) | Mordor Intelligence | - |
| USD 11.20 B (2023) | Global Consultancy A | Wider scope covers all molecular technologies, not PCR only |
| USD 8.80 B (2024) | Trade Journal B | Omits software and post warranty service revenue; updates every two years |
The comparison shows that Mordor's clearly defined scope, yearly refresh, and transparent variable selection give decision makers a balanced, traceable baseline they can rely on.


Key Questions Answered in the Report
How large is the PCR molecular diagnostics market today?
The market generated USD 10.26 billion in 2026 and is set to climb to USD 13.31 billion by 2031, reflecting a 5.36% CAGR.
Which product category generates the highest revenue?
Reagents and consumables account for 54.72% of total revenue because every amplification run consumes fresh primers, probes and buffers.
Why are digital PCR systems gaining popularity?
Digital platforms detect variant alleles down to 0.01%, enabling liquid-biopsy and minimal-residual-disease monitoring that traditional real-time PCR cannot support.
Which region is growing the fastest?
Asia-Pacific is projected to expand at a 6.27% CAGR through 2031, driven by healthcare-infrastructure upgrades and rising precision-medicine budgets.
How will new FDA regulations affect the market?
The FDA will phase out enforcement discretion for laboratory-developed tests, increasing compliance costs and prompting consolidation among smaller laboratories.
What is the main barrier to wider adoption in emerging markets?
High capital investment for instruments and ongoing consumable costs remain the primary hurdles for laboratories operating under constrained budgets.
Page last updated on: