Histology And Cytology Market Size and Share
Histology And Cytology Market Analysis by Mordor Intelligence
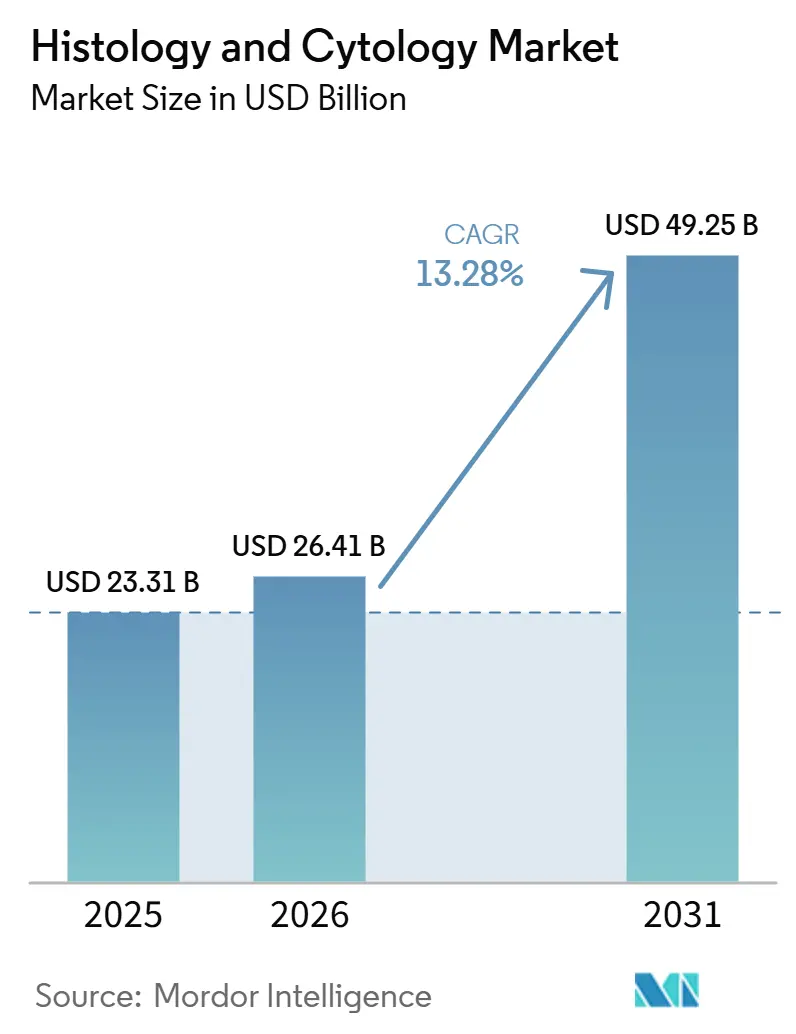
The Histology And Cytology Market size is projected to expand from USD 23.31 billion in 2025 and USD 26.41 billion in 2026 to USD 49.25 billion by 2031, registering a CAGR of 13.28% between 2026 to 2031.
The acceleration arises from AI-enabled whole-slide imaging, liquid biopsy advances, and lab automation that together mitigate global pathologist shortages while meeting higher cancer-screening volumes and precision-medicine demands. Consumables and reagents continue to underpin revenues, yet cloud-hosted diagnostic services post the fastest gains as health systems favor variable-cost outsourcing over capital expenditure. Clinically, histology retains primacy for tissue staging, but cytology benefits from minimally invasive liquid-based methods that enable real-time tumor monitoring. Competitive intensity increases as leading manufacturers close strategic acquisitions that bundle scanners, reagents, and AI software into unified offerings. Regional growth divides along digital-readiness lines: North America leads on share, whereas Asia-Pacific’s leapfrog adoption of automation generates the quickest incremental revenue.
Key Report Takeaways
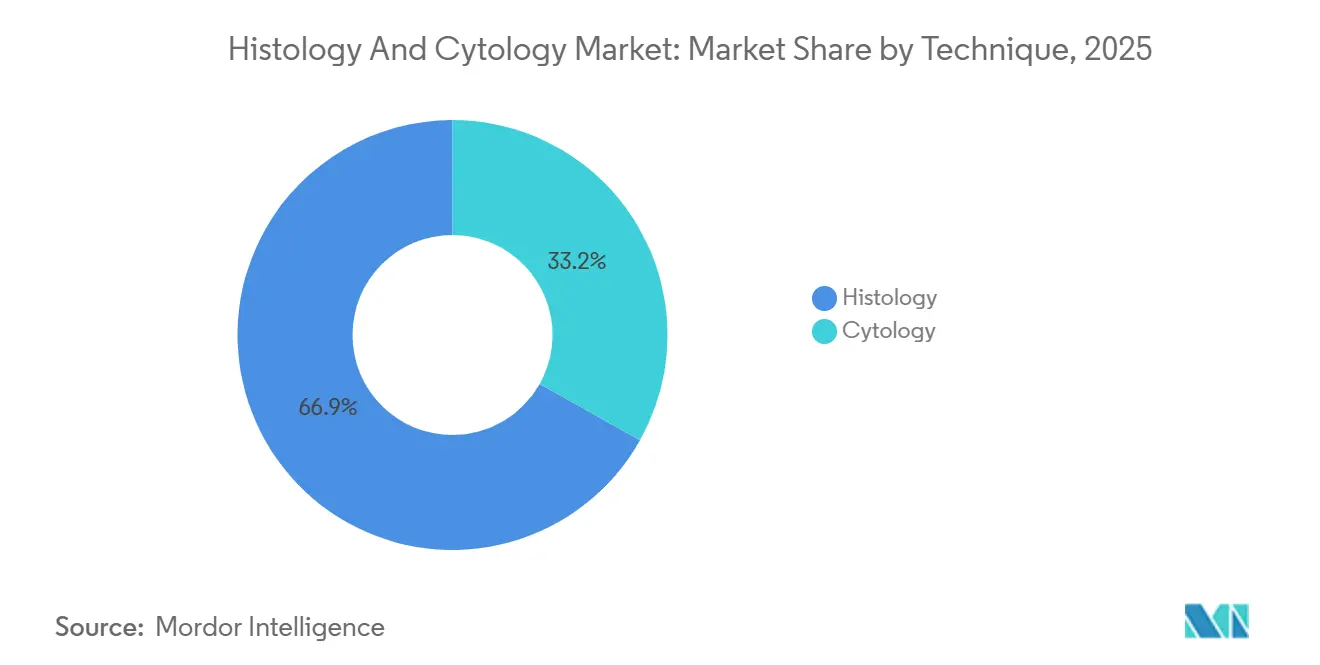
- By technique, histology held 66.85% of the histology and cytology market share in 2025, while cytology is projected to post the fastest 14.01% CAGR through 2031.
- By product, consumables generated 47.58% of 2025 revenue, whereas services are advancing at a 13.62% CAGR over the forecast horizon.
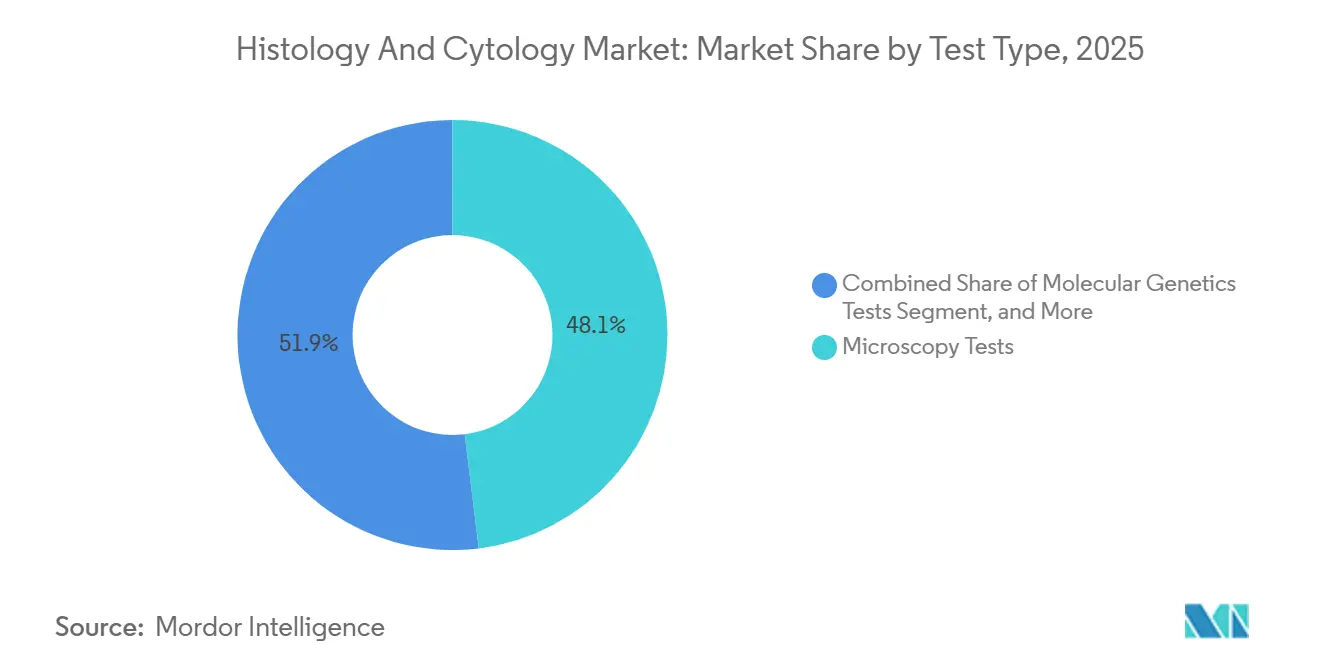
- By test type, microscopy dominated with a 48.06% share in 2025, but molecular genetics tests are on track for a 14.55% CAGR to 2031.
- By application, clinical diagnostics accounted for 53.48% of spending in 2025, while drug discovery and development is expanding at a 13.79% CAGR.
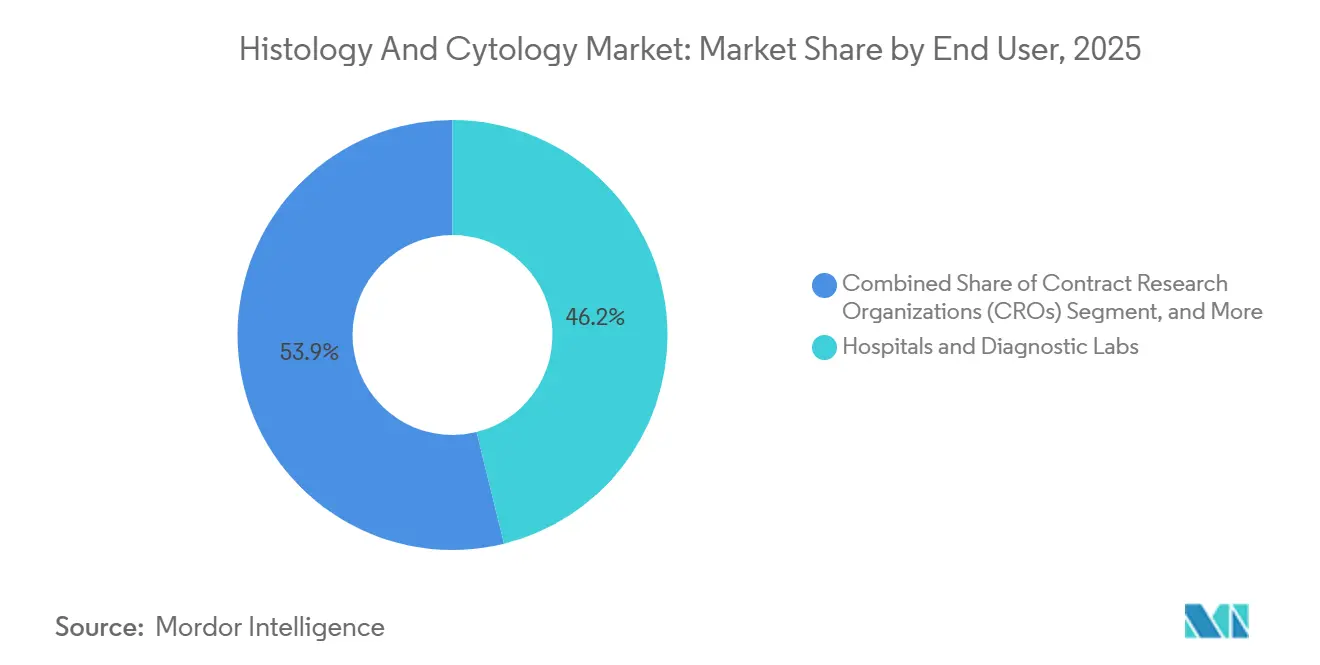
- By end user, hospitals and diagnostic labs captured 46.15% of the outlay in 2025, compared with contract research organizations, which are growing at a 13.95% CAGR.
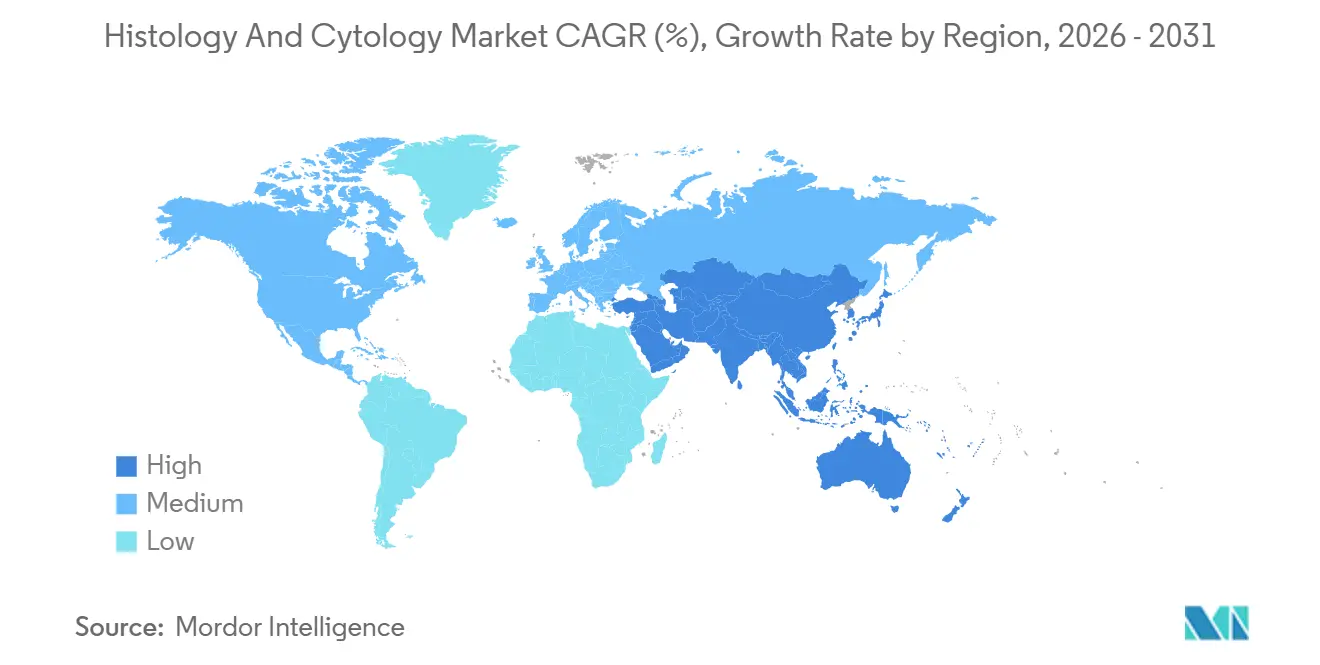
- By geography, North America led with 37.95% of the 2025 base, whereas Asia-Pacific is forecast to grow at a 13.76% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Histology And Cytology Market Trends and Insights
Driver Impact Analysis*
| DRIVER | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| AI-enabled whole-slide imaging adoption | +2.8% | Global, early traction in North America and Western Europe | Medium term (2–4 years) |
| Rise in companion diagnostics for oncology | +2.3% | Global, concentrated in North America, Europe, and Japan | Long term (≥ 4 years) |
| Growing demand for minimally invasive biopsies | +1.9% | Global, rapid uptake in Asia-Pacific urban centers | Short term (≤ 2 years) |
| Rapid lab automation in emerging economies | +1.6% | Core Asia-Pacific, spillover to Latin America | Medium term (2–4 years) |
| Subscription-based scanner-as-a-service models | +1.2% | North America and EU, expanding to Middle East | Short term (≤ 2 years) |
| Spatial-omics integration with digital histology | +1.0% | North America and EU research hubs, select Asia-Pacific sites | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
AI-Enabled Whole-Slide Imaging Adoption
FDA-cleared scanners now digitize entire slides at diagnostic resolution, permitting cloud review that offsets workforce gaps and halves turnaround in high-volume centers. Academic–industry programs, such as Danaher and Stanford’s smart-microscopy venture, generate additional revenue streams by pairing spatial biology with AI scoring algorithms. New CPT codes from the College of American Pathologists allow billing for digital workflow tasks, transforming previous cost centers into reimbursable services. European cost-effectiveness studies show that labs recoup capital outlays within six years by boosting throughput and enabling remote reads. The primary adoption hurdle remains scanner prices of USD 50,000–300,000, so vendors explore subscription models that bundle hardware, software, and maintenance.
Rise in Companion Diagnostics for Oncology
Targeted-therapy pipelines rely on validated biomarker tests; Roche’s PATHWAY HER2 assay for biliary-tract cancer illustrates first-mover advantages in niche indications. Flow-cytometry collaborations such as BD-Labcorp expand conventional cell-analysis platforms into treatment-selection tools. Thermo Fisher’s myeloMATCH trial uses broad NGS panels to match patients to trials, accelerating enrollment and de-risking drug development.[1]Thermo Fisher to Acquire Solventum Unit,” Thermo Fisher Scientific Newsroom, thermofisher.com FDA approvals have quickened as assay–drug pairings demonstrate survival benefits, giving diagnostics firms defensible revenue backed by drug-label inclusion. The trend scales beyond oncology into autoimmune and infectious diseases as therapeutics become biomarker-guided.
Growing Demand for Minimally Invasive Biopsies
Liquid biopsy captures circulating tumor DNA, enabling earlier detection and serial monitoring without surgical sampling. Sensitivity for actionable mutations in lung and breast cancers now rivals tissue biopsy, expanding eligibility for targeted therapy and reducing patient morbidity. Multi-cancer early-detection represents a USD 8 billion U.S. opportunity as payers weigh cost offsets from stage-shift diagnoses. Standardization remains a barrier because platforms vary in capture chemistry and bioinformatics, but AI-aided signal calling is shrinking false-positive rates. As protocols harmonize, cytology specimen volumes rise, reinforcing demand for high-throughput molecular workflows within the histology and cytology market.
Rapid Lab Automation in Emerging Economies
Industry 5.0 concepts deploy collaborative robots that batch-process staining, sectioning, and cover-slipping, allowing technicians to supervise multiple instruments simultaneously. APAC labs bypass legacy infrastructure by integrating IoT-enabled analyzers that auto-route images to centralized pathologists, compressing result times from days to hours. Vendors tailor ruggedized hardware and low-footprint scanners to constrained settings, pairing them with on-site training to sustain uptime. Automation improves consistency in high-volume cervical and breast-screening programs, thereby reinforcing public-health initiatives that depend on reliable diagnostics.
Restraint Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Shortage of pathologists in rural regions | -1.8% | Global, particularly acute in rural areas and developing countries | Long term (≥ 4 years) |
| High capital cost of digital scanners | -1.2% | Global, with greater impact in emerging markets | Medium term (2-4 years) |
| Limited standardization of liquid-based cytology | -0.9% | Global, with stricter regulations in EU and North America | Short term (≤ 2 years) |
| Interoperability gaps between AI tools & VNA archives | -0.7% | Global, with varying impact by region | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Shortage of Pathologists in Rural Regions
Only 3% of UK histopathology departments report adequate staffing, with 78% holding vacancies that delay diagnoses. United States surveys forecast 19.6% cytology retirements by 2027, outpacing the output of training programs.[2]Sharmila Bhatia et al., “The Human Tumor Atlas Network,” Cell, humantumoratlas.org Telepathology can redistribute slides, yet broadband deficits and funding gaps limit reach. AI triage systems lessen manual load but still require human sign-out. Without policy incentives to relocate or train specialists, rural disparities persist, tempering the growth of the histology and cytology market. India trains roughly 1,500 new pathologists per year, against a demand of 4,000, leaving a structural deficit that automation only partially addresses. Chronic workforce gaps restrain tissue testing volumes, especially for complex panels that still need expert interpretation.
High Capital Cost of Digital Scanners
Whole-slide scanners cost USD 50,000–300,000 plus servers and calibration, a hurdle for community labs. Only 33% of U.S. facilities have digitization plans after pandemic budget re-prioritization. ROI calculators predict a 6-year payback period, yet CFOs seek shorter cycles. Subscription and managed-service models lower the entry cost but can exceed the purchase cost over a decade. Grant funding and public-private partnerships are emerging to subsidize first installations in resource-limited regions.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Technique: Histology Dominance Faces Cytology Innovation
Histology accounted for 66.85% of 2025 revenue; however, cytology is surging at a 14.01% CAGR as laboratories embrace non-invasive liquid biopsy protocols that enable serial monitoring without repeated excisions. This demand drives consumption, especially of antigen-specific antibodies required for immunochemistry verification of circulating tumor cells. Cytology expansion also leverages AI-guided image analysis to accelerate Pap slide review and flag atypical cells for human confirmation. Laboratories thereby reallocate skilled staff toward complex differential diagnoses, raising overall productivity.
Cytology’s ascent is encouraging vendors to integrate dual-modality scanners capable of both bright-field tissue and thin-layer cell imaging, thereby maximizing equipment utilization. The histology and cytology market, attributed to cytology, is forecast to expand fastest in outpatient women ’s-health clinics, where cervical self-collection drives higher sample volumes to centralized labs. Nonetheless, histology remains indispensable for surgical-margin assessment and evaluation of in situ architecture, thereby ensuring continued demand for reagents and microtomes.

By Product: Consumables Dominance Meets Services Surge
Consumables and reagents accounted for 47.58% of the 2025 value, as stains, antibodies, and RNA probes remain recurring necessities for every block or slide processed. The histology and cytology market size for this product class scales linearly with biopsy volumes, providing predictable revenue for reagent suppliers. Manufacturers differentiate by offering ready-to-use kits pre-optimized for digital scanners, minimizing manual adjustments.
Services, though smaller today, exhibit a 13.62% CAGR as providers deliver cloud-based slide digitization, AI scoring, and second-opinion consults. Outsourced models appeal to clinics lacking capital or personnel and enable 24-hour turnaround across time zones. Vendors bundle logistics, quality checks, and LIS integration, presenting compelling total-cost-of-ownership savings. Instrument sales plateau because extended hardware lifecycles and shared-use depots satisfy existing capacity, shifting supplier focus toward consumable pull-through and service annuities.
By Test Type: Molecular Genetics Outpaces Microscopy
In 2025, microscopy and cytogenetic assays accounted for 48.06% of the histology and cytology market share, serving as critical tools for leukemia classification, prenatal diagnostics, and evaluation of tissue morphology. While immunohistochemistry panels provide vital insights into protein expression for therapy decision-making, the demand for actionable genomic data is driving a shift. Consequently, molecular genetics tests are experiencing a strong 14.55% CAGR, gradually reallocating budgets previously dedicated to morphology-focused studies.
Indicators such as tumor mutational burden, microsatellite instability, and gene fusion detection are now key in determining eligibility for immunotherapies and targeted small molecules. Advances in automation and sample multiplexing have enabled next-generation sequencing panels to meet five-day turnaround expectations. Although flow cytometry remains essential for lymphoma immunophenotyping, its growth is constrained by the labor-intensive nature of sample preparation. Additionally, quality regulations are becoming stricter, with CAP requiring external proficiency testing for each molecular marker, raising operational standards for smaller facilities.

By Application: Drug Discovery Gains Share from Clinical Diagnostics
Routine biopsies and surgical pathology kept clinical diagnostics at 53.48% of 2025 revenue, but growth decelerated slightly as hospitals optimized utilization rates. Pharmaceutical R&D is driving the fastest growth, with drug discovery and development applications projected to achieve a 13.79% CAGR. Contract research organizations are delivering integrated slide processing, multiplex staining, and AI-powered analysis, enabling sponsors to eliminate weaker candidates earlier and significantly reduce development timelines.
Biomarker discovery acts as a bridge between research and clinical applications, transforming exploratory insights into companion diagnostics. The histology and cytology market for translational applications is expected to grow as each new targeted therapy requires validated tissue-based assays. Although funding variability limits purely academic studies, strategic public-private partnerships are fostering innovative protocols, such as spatial omics, which are anticipated to be commercialized within 5 years.
By End User: Hospitals Lead While CROs Accelerate
Hospitals and diagnostic laboratories commanded 46.15% of 2025 spending due to mandatory on-site diagnostic services. Their adoption of digital workflows remains gradual, constrained by budget cycles and IT integration challenges. Meanwhile, contract research organizations record a 13.95% CAGR as pharma outsourcing intensifies. CROs leverage specialized expertise and scalable infrastructure to provide histopathology, molecular assays, and bioinformatics in unified packages, appealing to biotech clients seeking rapid development timelines.
Academic and research institutes expand moderate volumes through grant-funded studies, whereas pharmaceutical companies internalize select high-value assays for data-security reasons. Over time, hospital consolidation may shift more routine work to centralized reference labs, freeing hospitals to focus on urgent intraoperative consultations.

Geography Analysis
North America maintains a 37.95% share thanks to robust reimbursement, widespread scanner approvals, and early validation of AI algorithms. Leading cancer centers conduct digital-first workflows, and multistate IDNs negotiate value-based contracts that reimburse performance metrics rather than per-slide fees. Nonetheless, rural understaffing persists, prompting telepathology networks that pool subspecialty expertise across state lines. Proposed 2026 Medicare fee reductions inject uncertainty, but private insurers offset the risk by rewarding demonstrable gains in accuracy.
Asia-Pacific posts the highest 13.76% CAGR, driven by China’s nationwide cancer-screening expansion and India’s public-private lab partnerships that deploy automated staining lines. Governments prioritize leapfrog technologies to bridge specialist gaps without replicating legacy infrastructure. Japanese vendors pilot end-to-end digital-pathology suites in local hospitals, combining high-speed scanners with natural-language pathology reporting. ASEAN regulatory harmonization eases device clearance, encouraging multinationals to site regional hubs that cut import duties and service delays.
Europe records steady growth anchored in collaborative research and strong data-privacy safeguards. Germany’s hospital-modernization grants accelerate scanner procurement, and the UK’s NHS Digital Pathology program scales teleconsultation across trusts. France and Italy foster public–academic alliances that commercialize AI de-identification pipelines compliant with GDPR. Brexit prompts UK firms to pursue dual registration in EMA and MHRA pathways, adding regulatory overhead but also opening flexibility for innovative trial designs. Overall adoption aligns with national e-health roadmaps, positioning Europe as a disciplined yet attractive market segment for vendors.
Competitive Landscape
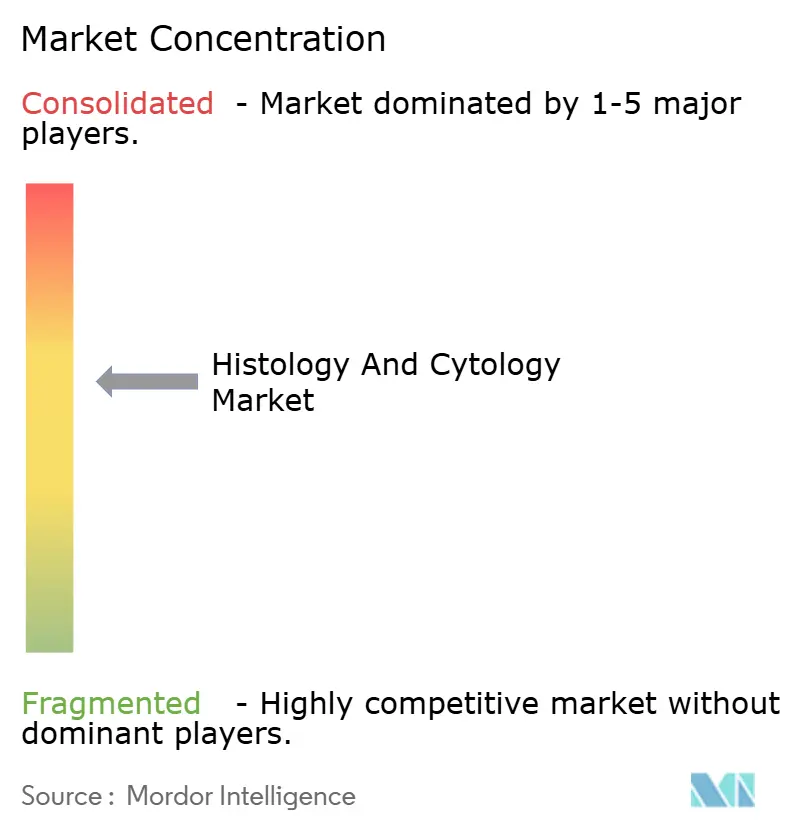
The market displays moderate consolidation: the top five firms collectively hold significant market share, translating to a market-concentration score of 7. Thermo Fisher’s USD 4.1 billion purchase of Solventum’s filtration unit deepens life-science integration and cross-sells consumables to pathology customers. Danaher’s Leica Biosystems partners with Stanford to co-develop smart microscopes that couple spatial biology with AI analytics. BD reorganizes through a USD 4 billion merger of its biosciences assets with Waters, freeing capital to double down on advanced med-tech growth segments.
Cloud-native service providers such as 3DHISTECH and Proscia position themselves as nimble challengers, offering subscription platforms that bypass heavy hardware outlays. Instrument specialists differentiate via open-architecture systems that accept third-party AI plug-ins, courting customers wary of vendor lock-in. Pricing pressure persists in consumables as group-purchasing organizations negotiate bulk discounts, compelling reagent makers to bundle training and QC software for stickier revenue.
Emerging opportunities focus on niche assays, such as dual-stain cytology for HPV triage or rare-disease panels, where intellectual property and clinical evidence create defensible margins. Vendors with regulatory experience gain an advantage as AI algorithms now require clinical-grade validation. Overall, sustained M&A is expected as integrated-solution demand favors companies able to supply scanners, reagents, and analytics in cohesive packages within the histology and cytology market.
Histology And Cytology Industry Leaders
Abbott Laboratories
Becton Dickinson and Company
Merck KGaA
F. Hoffmann-La Roche Ltd
Hologic Inc
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2026: Superb Dx, a CAP-accredited laboratory specializing in advanced diagnostic services, has entered into a strategic collaboration with AIxMed, a leader in AI-driven software for urine cytology. This partnership will integrate AIxMed’s proprietary AI technology into Superb Dx’s digital pathology platform, strengthening its portfolio of advanced diagnostic solutions for the urology market.
- October 2025: AIxMed Inc. and Biopticka Laboratoř signed an MoU to integrate AIxURO, an AI solution for urine cytology that supports The Paris System and enhances bladder cancer detection.
- February 2025: Thermo Fisher Scientific agreed to purchase Solventum’s Purification and Filtration business for USD 4.1 billion, adding roughly USD 1 billion to its Life Sciences Solutions unit.
- January 2025: Roche obtained FDA clearance for the VENTANA Kappa and Lambda Dual ISH mRNA Probe Cocktail covering more than 60 B-cell-lymphoma subtypes on a single slide.
Global Histology And Cytology Market Report Scope
As per the scope of the report, cytology and histology are two different branches of pathology, where cytology mainly involves looking at individual cells or clusters of cells, and histology involves examining an entire section of tissue containing many types of cells.
The histology and cytology market is segmented by type of examination, test type, application, end-user, and geography. By type of examination, the market is segmented into histology and cytology. By test type, the market is segmented into microscopy tests, molecular genetics tests, and flow cytometry. By application, the market is segmented into clinical diagnostics, drug discovery & development, research, and translational & biomarker validation. By end-user, the market is segmented into hospitals and clinics, academic and research institutes, and other end users. By geography, the market is segmented into North America, Europe, Asia-Pacific, Middle East and Africa, and South America. The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers market size and forecasts in value (USD) for the above segments.
| Histology |
| Cytology |
| Instruments |
| Analysis Software Systems |
| Consumables & Reagents |
| Services |
| Microscopy Tests | Cytogenetic Tests | Karyotyping |
| Fluorescent In-situ Hybridization (FISH) | ||
| Polymerase Chain Reaction | ||
| Other Microscopy Tests | ||
| Molecular Genetics Tests | ||
| Flow Cytometry |
| Clinical Diagnostics |
| Drug Discovery & Development |
| Research |
| Translational & Biomarker Validation |
| Hospitals & Diagnostic Labs |
| Academic & Research Institutes |
| Pharmaceutical & Biotechnology Companies |
| Contract Research Organizations (CROs) |
| Reference Laboratories |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Technique | Histology | ||
| Cytology | |||
| By Product | Instruments | ||
| Analysis Software Systems | |||
| Consumables & Reagents | |||
| Services | |||
| By Test Type | Microscopy Tests | Cytogenetic Tests | Karyotyping |
| Fluorescent In-situ Hybridization (FISH) | |||
| Polymerase Chain Reaction | |||
| Other Microscopy Tests | |||
| Molecular Genetics Tests | |||
| Flow Cytometry | |||
| By Application | Clinical Diagnostics | ||
| Drug Discovery & Development | |||
| Research | |||
| Translational & Biomarker Validation | |||
| By End User | Hospitals & Diagnostic Labs | ||
| Academic & Research Institutes | |||
| Pharmaceutical & Biotechnology Companies | |||
| Contract Research Organizations (CROs) | |||
| Reference Laboratories | |||
| By Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| India | |||
| Japan | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||


Key Questions Answered in the Report
What revenue does the histology and cytology market generate in 2026?
The market produces USD 26.41 billion in 2026 and is projected to climb to USD 49.25 billion by 2031.
Which region grows fastest in histology and cytology through 2031?
Asia-Pacific records a 13.76% CAGR as governments fund large-scale screening and automation projects.
Why is cytology gaining ground on histology?
Liquid biopsy and other non-invasive methods create real-time monitoring options that boost cytology volumes at a 14.01% CAGR.
Which product category shows the quickest expansion?
Cloud-delivered diagnostic services rise at a 13.62% CAGR as laboratories outsource slide digitization and AI analysis.
How are AI tools affecting pathology workloads?
Whole-slide imaging with AI triage automates routine screening, letting pathologists focus on complex cases and cutting turnaround times.
What hampers digital-scanner adoption in smaller labs?
Capital costs of USD 50,000300,000 per unit plus IT infrastructure remain the chief barrier despite six-year ROI projections.
Page last updated on:









