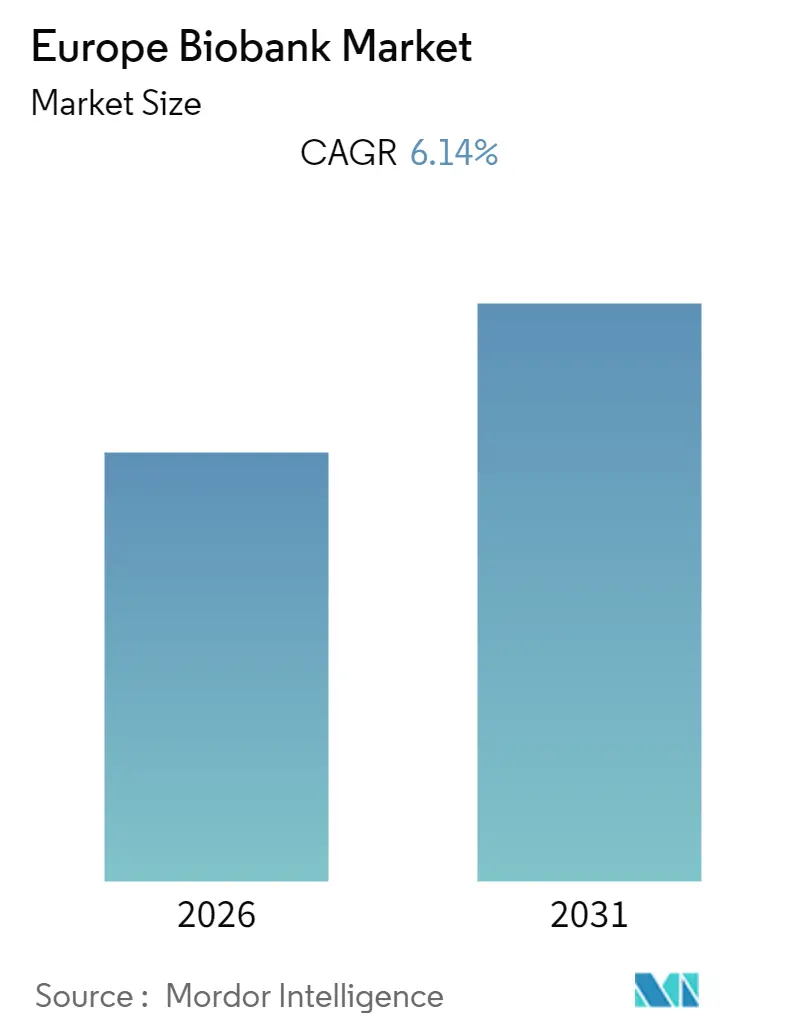
Europe Biobank Market Analysis by Mordor Intelligence
The Europe Biobank Market size is expected to register a CAGR of 6.14% during the forecast period (2026-2031).
Stem cell practices are on the rise in European countries and are contributing to the development of new solutions for various diseases. Several advancements have been made in regenerative medicine through stem cell technology over the past decade. Thus, it has become one of the vital treatment methods for diseases, such as Alzheimer's, diabetes, cancer, and rare genetic diseases. To benefit from the existing therapies, umbilical cord cells and other stem cells are preserved in Biobanks. The European Science Foundation (ESF) has been an active player in advancements made in stem cell and regenerative medicine research and in providing new treatments for incurable diseases and opportunities. With the increase in awareness about stem cell therapies, there have been a larger number of parents, who choose umbilical cord banks for their children. Biobanks not only aid in the therapies for genetic diseases, but also in medical research on rare genetic disorders. However, other factors such as rising burden of chronic diseases and R&D funding and investments by government and non-governmental organizations are also expected to drive the market in the forecast period.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Europe Biobank Market Trends and Insights
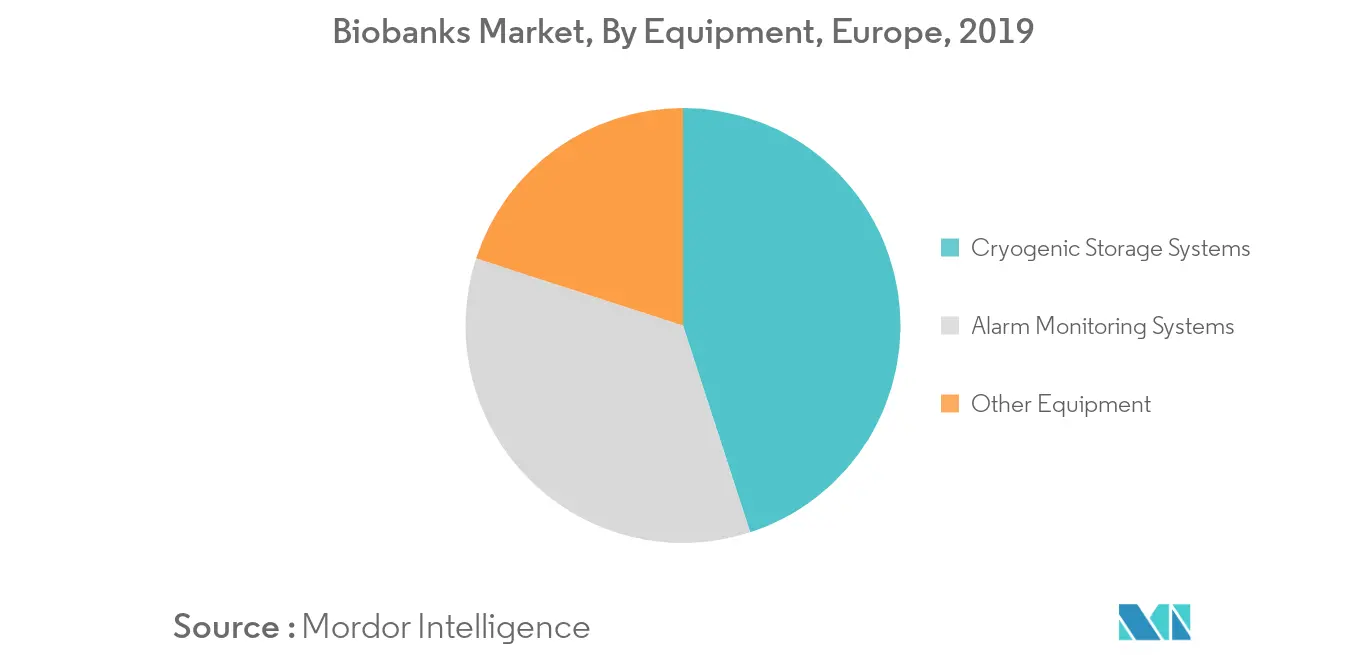
Alarm Monitoring Systems Segment is Expected to Show Better Growth in the Forecast Years
Based on Equipment, it is segmented into cryogenic storage system, alarm monitoring systems and other equipments. Stem cell banking is considered as one of the most promising and emerging fields. The cryogenic storage systems, freezers, refrigerators, and all other storage devices in a biobank require continuous monitoring of various processes. This is effectively done only with an alarm monitoring system. Alarms are used at various stages, from filling to continuous monitoring of temperature. Hence, with advancements in technology and the rising adoption of automated storage systems, there is high demand for automated alarm monitoring devices, which is expected to fuel the market growth.
Competitive Landscape
The market is competitive and with the entrance of key players, the market is expected to grow due to increased competition amongst the existing players. For instance, Atlanta Biologicals Inc. (Bio-Techne Corporation), Becton, Dickinson and Company, BioLifeSolutions Inc., Hamilton Company, Qiagen N.V, , Stemcell Technologies Inc., Thermo Fisher Scientific, etc. are providing these products across the globe.
Europe Biobank Industry Leaders
BioLifeSolutions Inc.
Hamilton Company
Sigma-Aldrich Inc. (Merck KGaA)
Thermo Fisher Scientific Inc.
VWR International LLC
- *Disclaimer: Major Players sorted in no particular order


Europe Biobank Market Report Scope
As per the scope of the report, Biobanks are the bio-repository, wherein various biomaterials (usually human specimens) are stored for research purposes. This study targets gaining a detailed overview of the market dynamics of the contemporary biobank market during the forecast period.
| Cryogenic Storage Systems | Refrigerators |
| Ice Machines | |
| Freezers | |
| Alarm Monitoring Systems | |
| Other Equipment |
| Optimized Media |
| Non-optimized Media |
| Human Tissue Biobanking |
| Stem Cell Biobanking |
| Cord Banking |
| DNA/RNA Biobanking |
| Other Services |
| Regenerative Medicine |
| Drug Discovery |
| Disease Research |
| Equipment | Cryogenic Storage Systems | Refrigerators |
| Ice Machines | ||
| Freezers | ||
| Alarm Monitoring Systems | ||
| Other Equipment | ||
| Media | Optimized Media | |
| Non-optimized Media | ||
| Services | Human Tissue Biobanking | |
| Stem Cell Biobanking | ||
| Cord Banking | ||
| DNA/RNA Biobanking | ||
| Other Services | ||
| By Application | Regenerative Medicine | |
| Drug Discovery | ||
| Disease Research |


Key Questions Answered in the Report
What is the current Europe Biobank Market size?
The Europe Biobank Market is projected to register a CAGR of 6.14% during the forecast period (2026-2031)
Who are the key players in Europe Biobank Market?
BioLifeSolutions Inc., Hamilton Company, Sigma-Aldrich Inc. (Merck KGaA), Thermo Fisher Scientific Inc. and VWR International LLC are the major companies operating in the Europe Biobank Market.
What years does this Europe Biobank Market cover?
The report covers the Europe Biobank Market historical market size for years: 2019, 2020, 2021, 2022, 2023, 2024 and 2025. The report also forecasts the Europe Biobank Market size for years: 2026, 2027, 2028, 2029, 2030 and 2031.
Page last updated on:








