Canada In-vitro Diagnostics Market Analysis by Mordor Intelligence
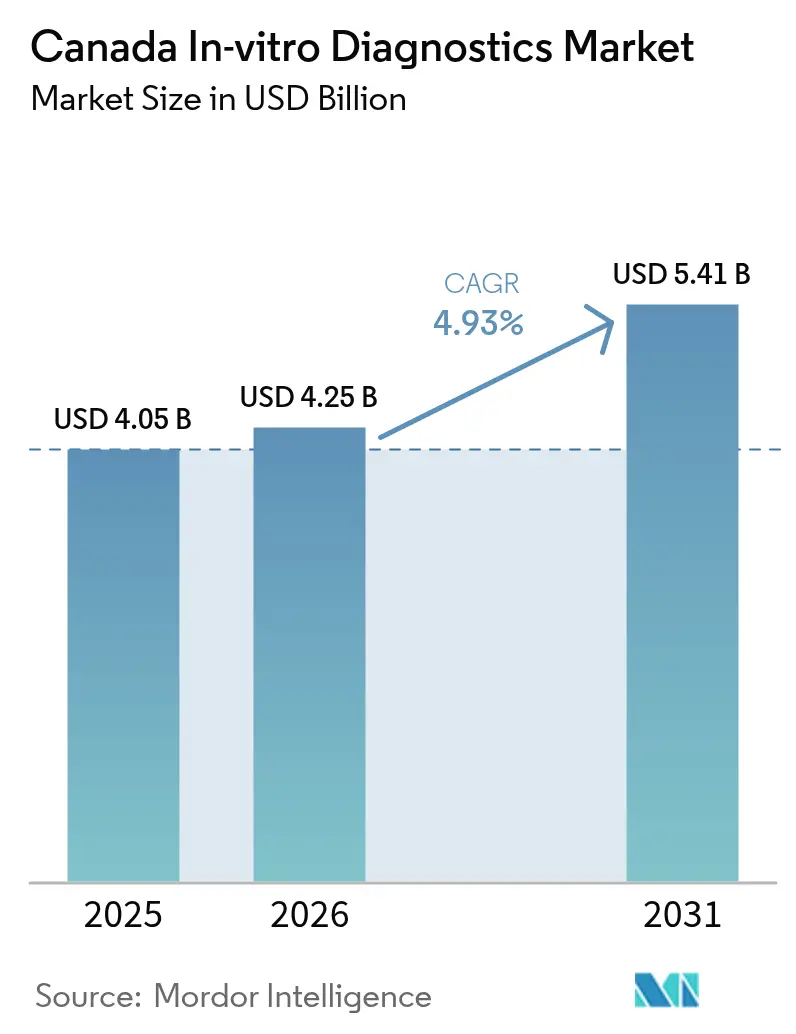
The Canada in-vitro diagnostics market size in 2026 is estimated at USD 4.25 billion, growing from 2025 value of USD 4.05 billion with 2031 projections showing USD 5.41 billion, growing at 4.93% CAGR over 2026-2031. Rising healthcare spending that hit USD 372 billion in 2024, equivalent to 12.4% of national GDP, is creating headroom for test adoption. Demand is further amplified by a growing chronic-disease burden, government commitments exceeding USD 200 billion to modernize the system, and rapid uptake of molecular and digital platforms. Consolidation among large laboratories, the pivot toward decentralized testing, and provincial push for preventive screening programs are reinforcing steady volume growth. Competitive intensity is increasing as global majors introduce precision-oriented solutions and capitalize on Health Canada’s comparatively flexible stance toward laboratory-developed tests.
Key Report Takeaways
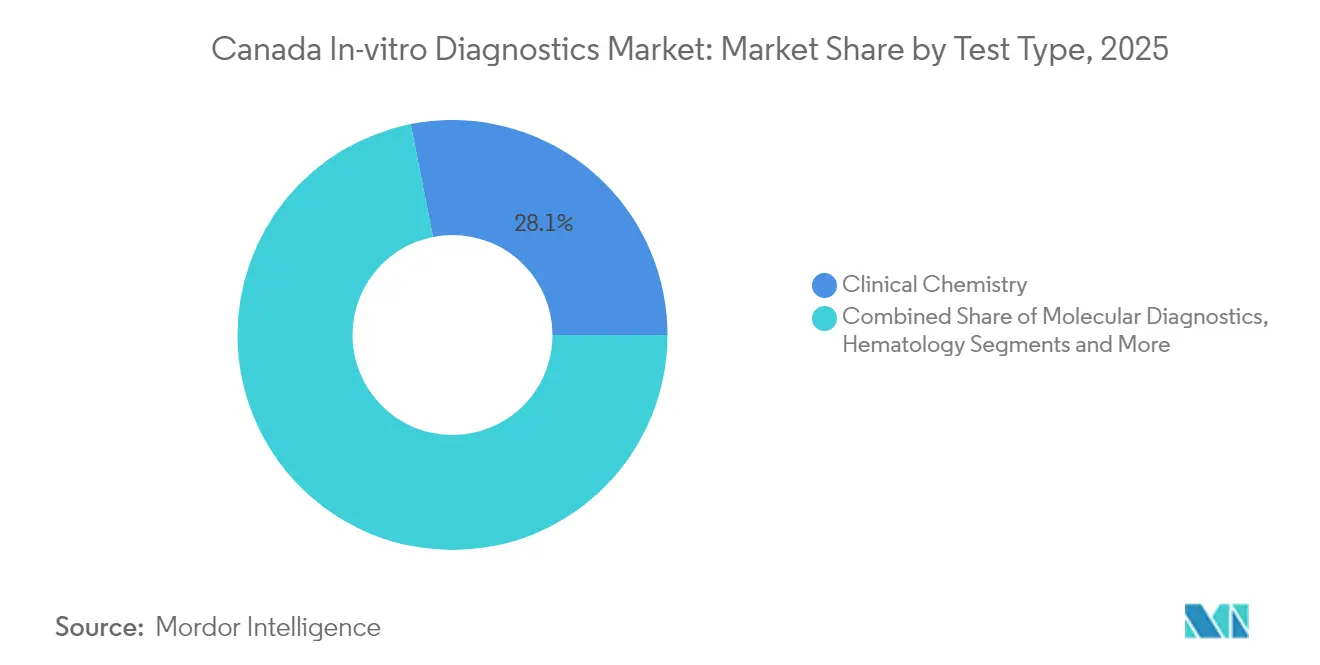
- By test type, Clinical Chemistry led with 28.12% share in 2025, whereas Molecular Diagnostics is projected to advance at a 10.42% CAGR from 2026 to 2031.
- By product, Reagents & Kits captured 62.14% of the Canada in-vitro diagnostics market share in 2025; Software & Services are forecast to grow at a 9.55% CAGR to 2031.
- By usability, Disposable IVD Devices accounted for 70.63% of the Canada in-vitro diagnostics market size in 2025, while Reusable IVD Devices are set to expand at a 9.05% CAGR through 2031.
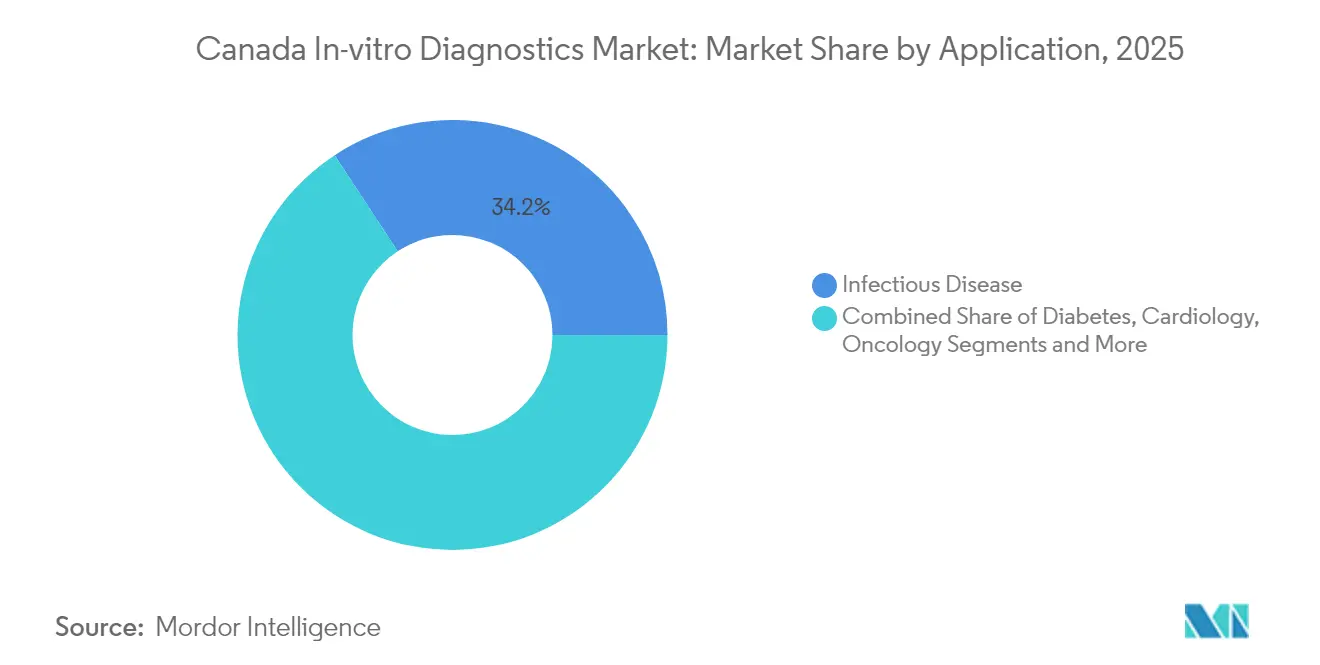
- By application, Infectious Disease testing held 34.23% revenue share in 2025, but Cancer/Oncology diagnostics will rise fastest at a 10.55% CAGR to 2031.
- By end user, Diagnostic Laboratories secured 45.74% share in 2025, whereas Home Care Settings are projected to grow quickest at an 8.25% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Canada In-vitro Diagnostics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Growing Burden of Chronic & Infectious Diseases | +1.2% | National, with higher impact in provinces with aging populations (Ontario, Quebec, British Columbia) | Long term (≥ 4 years) |
| Government-Led Healthcare Capacity Expansion & Modernization | +0.9% | National, with emphasis on underserved provinces and territories | Medium term (2-4 years) |
| Rising Adoption of Advanced Diagnostic Technologies | +0.7% | Urban centers initially, with gradual expansion to rural areas | Medium term (2-4 years) |
| Favorable National Screening & Preventive Health Programs | +0.5% | National, with provincial variations in implementation | Medium term (2-4 years) |
| Expanding Health-Insurance Coverage Including Mandatory Expat Benefits | +0.4% | National, with emphasis on provinces with higher immigrant populations | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Growing Burden of Chronic & Infectious Diseases
Nineteen percent of Canadians were aged 65 or older in 2024, a ratio expected to climb to 22.5% in coming years, intensifying demand for early diagnostic services. Long-COVID adds complexity, with 1 in 5 adults reporting persistent symptoms and 100,000 unable to work as of December 2023[1]Office of the Chief Science Advisor, “Post-COVID Condition and its Continued Impact,” Science.gc.ca. These overlapping pressures are funneling public and private investment toward molecular and point-of-care assays capable of detecting chronic and infectious conditions earlier. As molecular diagnostics penetrate routine workflows, demand is shifting from static chemistry panels to high-value genomic and proteomic tests. This structural change underpins long-run volume and value growth for the Canada in-vitro diagnostics market.
Government-Led Healthcare Capacity Expansion & Modernization
Federal and provincial authorities have pledged more than USD 200 billion over ten years to widen family health services, clear backlogs, fortify mental-health supports, and digitize systems. Laboratory upgrades and cloud-enabled data platforms form a core element of this strategy, opening opportunities for integrated testing ecosystems. New policy guidance for machine-learning medical devices signals Health Canada’s intent to nurture innovation while maintaining safety standards. Collectively, these initiatives expand addressable budgets and accelerate procurement cycles for advanced testing instruments and software across the Canada in-vitro diagnostics market.
Rising Adoption of Advanced Diagnostic Technologies
Molecular assays are growing at twice the overall market pace, reflecting their central role in precision-therapy decisions. A 2024 survey of oncologists highlighted gaps in biomarker testing access linked to limited standardization and financial hurdles. Manufacturers are responding with platforms such as Roche’s Digital LightCycler and self-collection HPV assays that combine automation, high sensitivity, and seamless data flow. Broader availability of next-generation sequencing is expected to embed genomic profiling into routine cancer care, supporting sustained expansion of the Canada in-vitro diagnostics market.
Favorable National Screening & Preventive Health Programs
Pan-Canadian newborn-screening recommendations released in March 2025 standardized testing for 25 conditions, with scope to add 29 more[2]Canada’s Drug Agency, “Newborn Screening Recommendations,” cda-amc.ca. Digital pathology pilots in British Columbia, funded at USD 2.65 million, illustrate the push to leverage AI for earlier detection, especially in rural communities. Preventive initiatives create predictable demand for assay kits, reagents, and analytics platforms, bolstering the medium-term outlook for the Canada in-vitro diagnostics market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Technical Complexity and Need for Skilled Personnel | -0.7% | National, with greater impact in rural and remote areas | Medium term (2-4 years) |
| High Cost of Diagnostic Equipment and Tests | -0.4% | National, with greater impact in smaller provinces with limited healthcare budgets | Short term (≤ 2 years) |
| Stringent Regulatory and Approval Processes | -0.3% | National, with particular impact on innovative diagnostic technologies and smaller companies | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Technical Complexity and Need for Skilled Personnel
Advanced molecular and digital platforms require specialized operators, yet expertise is unevenly distributed. A 2025 systematic review flagged infrastructure gaps and staffing shortages as key barriers to community-based molecular testing. Certification frameworks advocated by the Canadian Society of Clinical Chemists underscore the training burden for widespread point-of-care deployment. These manpower constraints slow test adoption in underserved areas, tempering growth potential for the Canada in-vitro diagnostics industry.
High Cost of Diagnostic Equipment and Tests
New MRI and CT scanners cost roughly USD 2 million, surpassing the USD 1.5 million tax-depreciation cap under the Immediate Expensing Incentive program. Two-thirds of imaging equipment is already over five years old, driving wait times and prompting calls for USD 1 billion in capital upgrades. Termination of federal funding for HIV self-testing kits after spending USD 17 million in two years highlights vulnerability of access programs to fiscal pressures. These cost headwinds could dampen near-term expansion of the Canada in-vitro diagnostics market.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Test Type: Molecular Diagnostics Redefining Clinical Practice
Clinical Chemistry retained 28.12% of Canada in-vitro diagnostics market share in 2025, reflecting its entrenched role in chronic-disease management. Molecular Diagnostics, projected to grow at a 10.42% CAGR, is gradually reshaping care pathways by enabling rapid pathogen identification and comprehensive genomic profiling. Investments such as the USD 18.9 million PREPARED initiative will deploy molecular testing across 50 sites, embedding PCR and sequencing workflows into frontline practice. Hematology, Microbiology, Immuno-Diagnostics, Coagulation, and Urinalysis remain critical for specific clinical decisions, yet none match the velocity of molecular uptake. As assay menus widen and reimbursement stabilizes, Molecular platforms are expected to claim a larger slice of the Canada in-vitro diagnostics market size by 2030.
Hematology analyzers underpin efficient blood-disorder work-ups, while Microbiology laboratories diversify into multiplex PCR to tackle antimicrobial resistance. Immuno-Diagnostics supports autoimmune monitoring and therapeutic-drug management, whereas Coagulation testing gains relevance in an aging population with cardiovascular risk. Affordable dipstick Urinalysis persists in primary care due to simplicity. However, the precision and speed of sequencing and digital PCR offer clear clinical advantages, positioning Molecular Diagnostics as the signature growth engine of the Canada in-vitro diagnostics market.

By Product: Digital Integration Transforming Test Ecosystems
Reagents & Kits generated 62.14% of revenue in 2025, underlining their consumable nature and resilient demand curve. Instruments face slower turnover because capital budgets remain constrained; many facilities operate legacy analyzers older than five years. Software & Services, though smaller, will register the fastest 9.55% CAGR as laboratories integrate cloud analytics, artificial intelligence, and workflow automation. Roche’s navify suite exemplifies the shift toward data-driven diagnostics that connect disparate sites and improve clinical decisions.
Ongoing supply-chain reinforcement since the pandemic has prompted multi-vendor sourcing strategies for reagents, while proposed tax changes to raise the expensing limit to USD 5 million could accelerate instrument upgrades. Altogether, digitization is expanding the value pool beyond wet chemistry, enlarging long-term potential for the Canada in-vitro diagnostics market.
By Usability: Disposable Devices Dominate Despite Sustainability Concerns
Disposable IVD Devices captured 70.63% of the segment in 2025 thanks to infection-control benefits and minimal maintenance requirements. Large-scale distribution of rapid antigen tests during COVID-19 embedded disposables into public health routines. Yet Reusable Devices are forecast to climb at a 9.05% CAGR as hospitals weigh environmental and cost implications. Advances in material science facilitate rigorous decontamination, reducing contamination risk and extending device lifespans.
Health Canada regulates both categories under the Medical Devices Regulations (SOR/98-282), with risk-based classification dictating evidence thresholds. Sustainability-oriented procurement guidelines may nudge laboratories toward reusable analyzers where feasible, gradually balancing the usability mix within the Canada in-vitro diagnostics market.
By Application: Precision Oncology Driving Diagnostic Innovation
Infectious Disease assays led with 34.23% revenue share in 2025, anchored by expanded respiratory-virus testing capacity. Cancer/Oncology testing is set to outpace all other applications at a 10.55% CAGR, propelled by biomarker-guided therapy protocols and funding such as Pfizer Canada’s USD 1.1 million grant program. Diabetes, Cardiology, Autoimmune, Nephrology, Blood Screening, and Prenatal segments continue to address population-specific needs, yet oncology’s escalation in test volume and complexity will increasingly influence revenue mix.
The proposed USD 25 million national molecular profiling network aims to standardize oncology diagnostics, potentially broadening patient access and reducing geographic disparities. These initiatives solidify oncology’s role as a primary catalyst for the Canada in-vitro diagnostics market size expansion.

By End User: Home Care Settings Disrupting Traditional Models
Diagnostic Laboratories accounted for 45.74% of test volume in 2025, fortified by Quest Diagnostics’ planned CAD 1.35 billion acquisition of LifeLabs. Hospitals and Clinics maintain substantial demand for acute-care assays, while Point-of-Care Centers bridge access gaps with rapid turnaround. Home Care Settings, projected to grow at an 8.25% CAGR, represent the vanguard of decentralized healthcare. Wearable sensors, mail-in genetic panels, and digitally guided rapid tests foster patient autonomy and reduce system load.
Quality-assurance frameworks such as MDSAP audit requirements are being adapted to cover decentralized platforms. As reimbursement catches up, home-based testing is expected to command a larger proportion of the Canada in-vitro diagnostics market.
Geography Analysis
Ontario, Quebec, and British Columbia collectively account for more than half of Canada in-vitro diagnostics market revenue, reflecting dense populations, teaching hospitals, and research centers. Alberta and Saskatchewan contribute meaningful volume via centralized laboratory networks that serve widely dispersed communities. Atlantic provinces and the territories trail in absolute terms but are experiencing above-average uptake of point-of-care devices due to infrastructure constraints. Government programs targeting underserved regions are funneling funds toward mobile clinics and cloud-linked laboratories, gradually narrowing service gaps. Urban centers lead early adoption of molecular assays, whereas rural areas initially rely on traditional chemistry panels due to staffing limitations. Investments in broadband and provincial cloud services are expected to redistribute digital testing capacity over the forecast horizon. The PREPARED pathogen-surveillance network, operating at 50 sites across the country, exemplifies nationwide initiatives designed to harmonize diagnostic standards and real-time data sharing. Altogether, geographical expansion of advanced testing will underpin steady growth in Canada in-vitro diagnostics market penetration across every province.
Competitive Landscape
The top manufacturers— Becton, Dickinson and Company, Abbott, Siemens Healthineers, and Danaher —hold a sizeable slice of the Canada in-vitro diagnostics market, enabling scale advantages in R&D, distribution, and regulatory affairs. Competitive pressures escalated after Quest Diagnostics moved to acquire LifeLabs, bringing 382 collection centers and 16 laboratories under its umbrella and signaling aggressive expansion by U.S. majors into Canadian territory. Emerging players are expected to pursue niche segments such as liquid biopsy or AI-enabled image analysis to differentiate.
Innovation remains the primary weapon: Roche’s cobas HPV self-collection and Digital LightCycler platforms target high-growth niches in women’s health and rare-mutation detection. Siemens is integrating Atellica CI analyzers for mid-volume labs, while Abbott expands point-of-care portfolios to home settings. Health Canada’s lighter oversight of laboratory-developed tests, relative to imminent FDA regulations, offers domestic labs flexibility to partner with diagnostic firms on bespoke assays, potentially lowering entry barriers for innovative SMEs.
Pricing competition remains moderate due to the importance of bundled reagent-service contracts and the technical switching costs associated with analyzer platforms. However, purchasing coalitions among provincial health authorities are negotiating volume discounts, nudging vendors toward value-based pricing models and integrated service agreements. Over the forecast period, digital connectivity, automation, and sample-to-answer molecular platforms will define competitive advantage within the Canada in-vitro diagnostics market.
Canada In-vitro Diagnostics Industry Leaders
Bio-Rad Laboratories Inc.
Danaher Corporation
Siemens Healthineers
Abbott Laboratories
Becton, Dickinson and Company
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2025: Canada’s Drug Agency issued pan-Canadian newborn-screening recommendations covering 25 conditions and signaling future expansion to 54 conditions, creating fresh demand for rare-disease testing platforms.
- February 2025: Becton Dickinson announced intent to divest its diagnostics and life-sciences divisions, a move that could spawn a new global competitor by 2026 with ripple effects on Canadian laboratory procurement.
Canada In-vitro Diagnostics Market Report Scope
As per the scope of the report, in-vitro diagnostics involves medical devices and consumables that are utilized to perform in vitro tests on various biological samples. They are used for the diagnosis of various medical conditions, such as chronic diseases. The Canada In-vitro market is segmented by Test Type (Clinical Chemistry, Molecular Diagnostics, Hematology, Immuno Diagnostics, and Other Tests), Product (Instrument, Reagent, and Other Products), Usability (Disposable IVD Devices and Reusable IVD Devices), Application, (Infectious Diseases, Diabetes, Cancer/Oncology, Cardiology, Autoimmune Disease, Nephrology, and Other Applications), and End Users (Diagnostic Laboratories, Hospitals and Clinics, and Other End Users). The report offers the value (in USD) for the above segments.
| Clinical Chemistry |
| Immuno-Diagnostics |
| Molecular Diagnostics |
| Hematology |
| Microbiology |
| Coagulation |
| Urinalysis |
| Other Tests |
| Instruments |
| Reagents & Kits |
| Software & Services |
| Disposable IVD Devices |
| Reusable IVD Devices |
| Infectious Disease |
| Diabetes |
| Cancer / Oncology |
| Cardiology |
| Autoimmune Disorders |
| Nephrology |
| Blood Screening |
| Prenatal / NIPT |
| Other Applications |
| Diagnostic Laboratories |
| Hospitals & Clinics |
| Point-of-Care Centers |
| Home Care Settings |
| Other End Users |
| By Test Type | Clinical Chemistry |
| Immuno-Diagnostics | |
| Molecular Diagnostics | |
| Hematology | |
| Microbiology | |
| Coagulation | |
| Urinalysis | |
| Other Tests | |
| By Product | Instruments |
| Reagents & Kits | |
| Software & Services | |
| By Usability | Disposable IVD Devices |
| Reusable IVD Devices | |
| By Application | Infectious Disease |
| Diabetes | |
| Cancer / Oncology | |
| Cardiology | |
| Autoimmune Disorders | |
| Nephrology | |
| Blood Screening | |
| Prenatal / NIPT | |
| Other Applications | |
| By End User | Diagnostic Laboratories |
| Hospitals & Clinics | |
| Point-of-Care Centers | |
| Home Care Settings | |
| Other End Users |


Key Questions Answered in the Report
What is the value of Canada's in-vitro diagnostics sector in 2026?
It is worth USD 4.25 billion, reflecting sustained investment in clinical testing infrastructure.
What compound annual growth rate is projected for Canada's in-vitro diagnostics through 2031?
The sector is forecast to grow at a 4.93% CAGR, reaching USD 5.41 billion by the end of the period.
Which test type is expanding quickest within Canada's IVD landscape?
Molecular diagnostics leads, advancing at a 10.42% CAGR on the back of precision-medicine demand.
How dominant are disposable IVD devices compared with reusable alternatives across Canada?
Disposable formats hold 70.63% of current use, although reusable devices are rising at a 9.05% CAGR.
What factors are driving the rapid uptake of diagnostic services in home care settings across Canada?
Patient preference for convenience, telehealth expansion, and reliable point-of-care technologies are propelling an 8.25% CAGR in home testing.
How is federal funding influencing adoption of advanced diagnostics across Canadian provinces?
More than USD 200 billion committed to healthcare modernization is accelerating procurement of molecular platforms, digital tools, and cloud-linked laboratory systems.
Page last updated on:








