Acute Ischemic Stroke Diagnosis Market Size and Share
Acute Ischemic Stroke Diagnosis Market Analysis by Mordor Intelligence
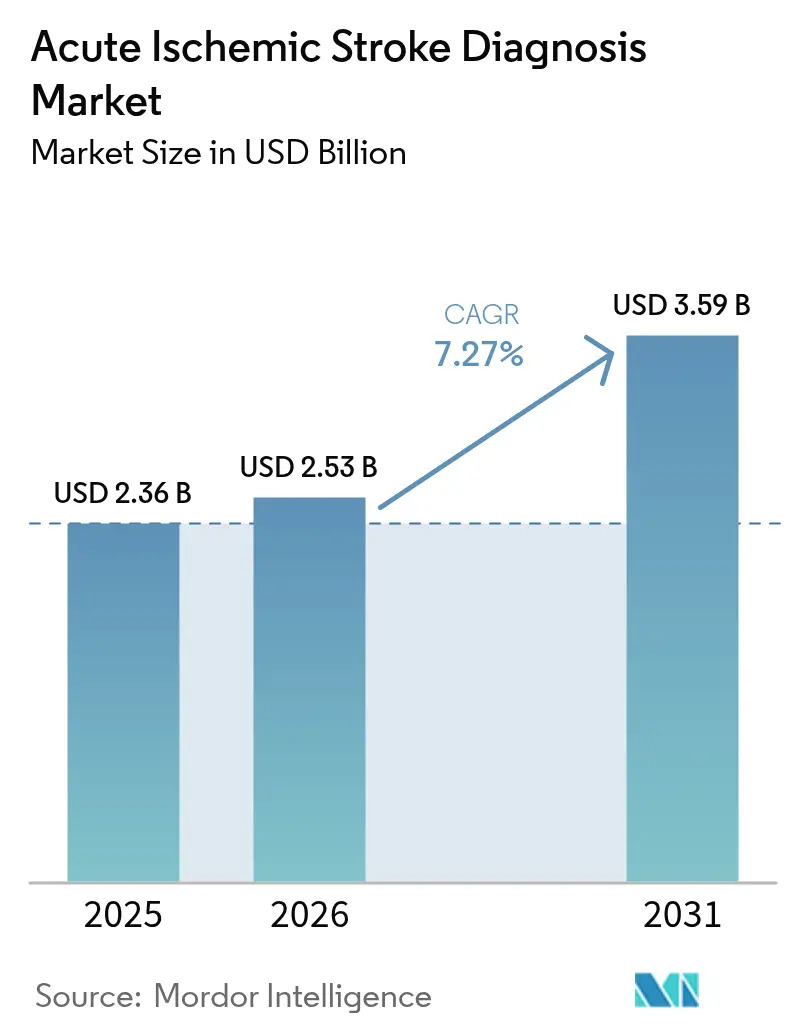
The acute ischemic stroke diagnosis market size is expected to grow from USD 2.36 billion in 2025 to USD 2.53 billion in 2026 and is forecast to reach USD 3.59 billion by 2031 at 7.27% CAGR over 2026-2031. Aging populations, guideline-driven expansion of mechanical thrombectomy time windows, and the steady infusion of artificial intelligence into emergency radiology are lifting procedure volumes and driving software adoption. Computed tomography (CT) remains the dominant front-line modality, yet AI-enabled stroke triage platforms—now cleared by the U.S. Food and Drug Administration—are shifting capital budgets toward subscription software that accelerates large-vessel occlusion routing. Hospitals are prioritizing door-to-needle metrics because Medicare, European payors, and Japanese insurers attach reimbursement bonuses to imaging-guided workflow performance. North America leads revenue because of its broad network of comprehensive stroke centers, while Asia-Pacific shows the fastest growth as national stroke registries in China and India mandate multimodal imaging for suspected large-vessel occlusion.
Key Report Takeaways
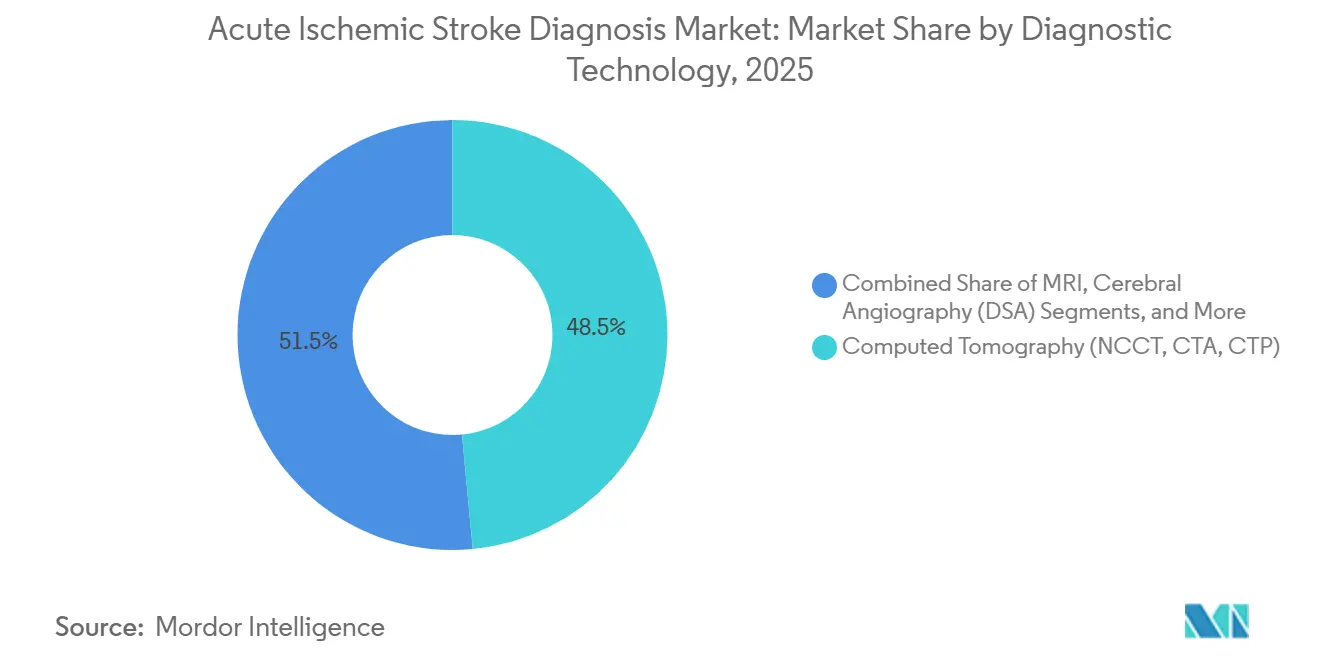
- By diagnostic technology, computed tomography captured 48.55% of 2025 revenue, while AI-enabled stroke decision-support software is projected to expand at 14.25% CAGR through 2031.
- By AI software function, large-vessel occlusion detection algorithms held 40.53% of AI software revenue in 2025; perfusion mismatch quantification tools are advancing at 16.75% CAGR through 2031.
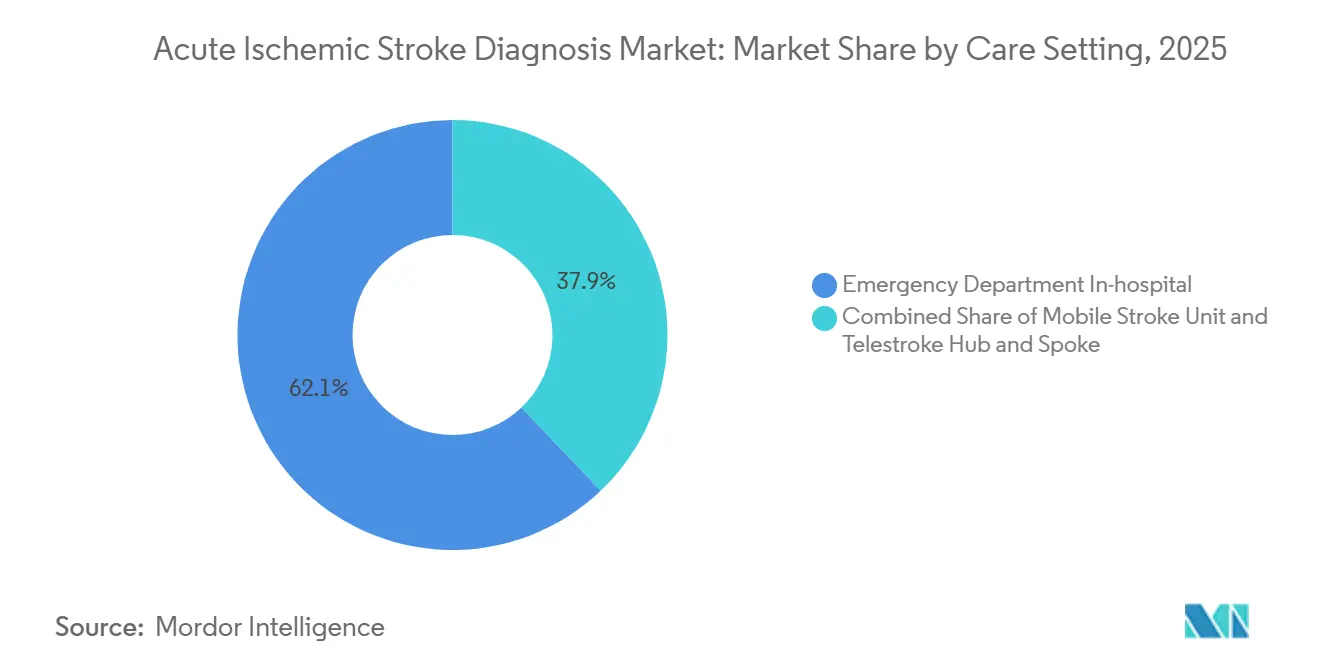
- By care setting, emergency departments generated 62.15% of 2025 care-setting revenue, yet mobile stroke units are forecast to climb at 15.82% CAGR as municipalities deploy CT-equipped ambulances.
- By end user, hospitals accounted for 74.65% of end-user revenue in 2025; ambulatory surgery and diagnostic imaging centers are growing at 12.32% as outpatient facilities add non-contrast CT for rapid hemorrhage exclusion.
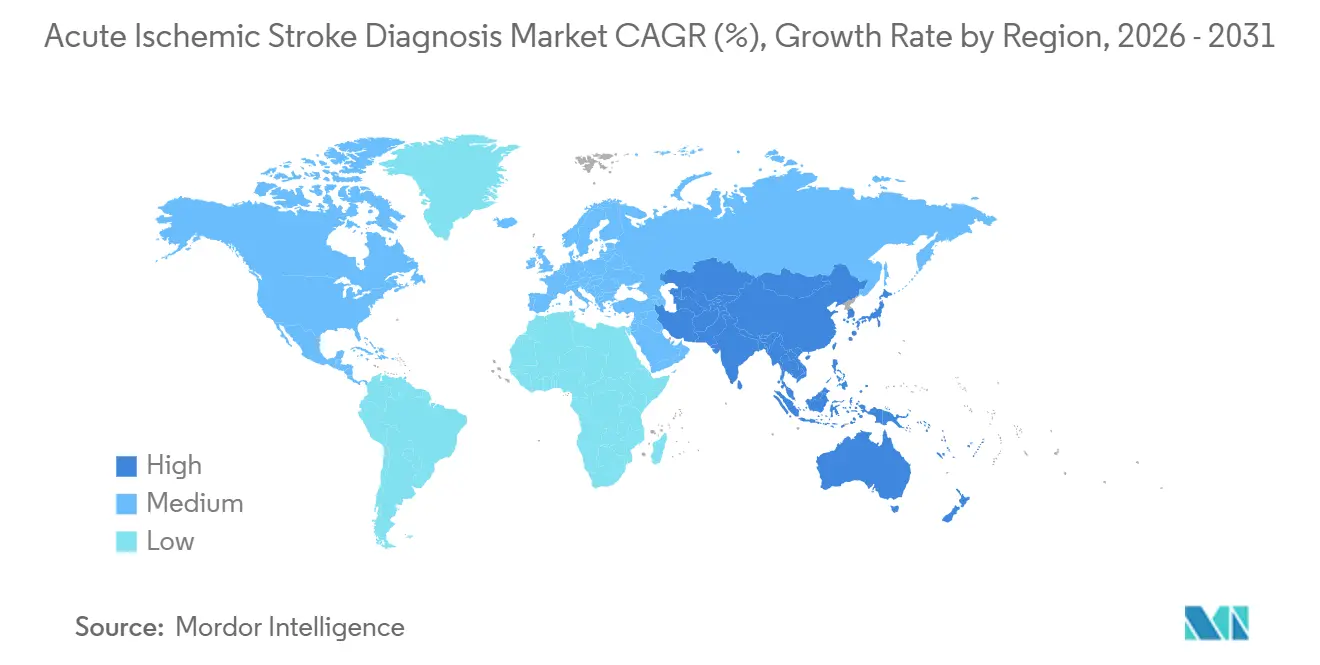
- By geography, North America contributed 36.23% of global revenue in 2025, whereas Asia-Pacific is projected to expand at 11.42% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Acute Ischemic Stroke Diagnosis Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Growing AIS incidence in 65-plus population | +1.2% | North America, Europe, East Asia | Long term (≥ 4 years) |
| Wider adoption of multimodal CT & MR imaging | +1.5% | North America, Western Europe, urban APAC | Medium term (2-4 years) |
| Expanding endovascular-thrombectomy window | +1.8% | Global OECD stroke centers | Medium term (2-4 years) |
| Quality initiatives & reimbursement metrics | +1.3% | United States, Germany, United Kingdom, Australia, Japan | Short term (≤ 2 years) |
| Photon-counting CT & 7 T MRI adoption | +0.9% | Academic centers in North America and Europe | Long term (≥ 4 years) |
| Subscription AI bundles with teleradiology | +1.4% | North America, Western Europe, India, Brazil | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Growing AIS Incidence in 65-Plus Population
The share of the global population aged 65 and older will reach 16% by 2030, and stroke incidence doubles with each successive decade after 65, sustaining diagnostic demand even in regions with plateauing age-adjusted rates. Japan already mandates CT or MRI within 60 minutes of presentation for suspected stroke, a policy response to the fact that 75% of domestic cases occur in seniors. U.S. Medicare data show rising hospitalizations for mild strokes thanks to faster pre-hospital recognition and imaging, further enlarging the pool of patients routed into acute workflows. These demographic realities create a durable tailwind for every segment of the acute ischemic stroke diagnosis market.
Wider Adoption of Multimodal CT & MR Imaging Protocols
Guidelines now recommend CT angiography and CT perfusion for all patients within 24 hours of last-known-well, normalizing tissue-based triage beyond the first 6 hours[1]American Heart Association, “2024 AHA/ASA Stroke Imaging Guidelines,” ahajournals.org. U.S. comprehensive stroke centers increased multimodal CT protocol adoption by 18% in 2025, driven by evidence that perfusion mismatch predicts favorable thrombectomy outcomes. Dual-energy CT platforms compress scan time below 3 minutes and help overcrowded emergency departments meet door-to-imaging targets. Although MRI retains superior sensitivity for posterior circulation infarcts, its longer acquisition time keeps CT in the front-line role.
Expanding Endovascular-Thrombectomy 24-Hour Time-Window
Real-world registries confirm that patients with favorable perfusion profiles benefit from mechanical thrombectomy up to 24 hours after symptom onset, doubling the treatable population[2]New England Journal of Medicine, “DAWN Trial,” nejm.org. U.S. thrombectomy volumes climbed 22% per year between 2020 and 2024, with late-window cases now one-third of all procedures. AI large-vessel occlusion detection shortens door-to-groin puncture by more than 20 minutes, strengthening the ROI case for algorithm subscriptions.
National Stroke-Care Quality Initiatives & Reimbursement Incentives
Medicare bundles, German and Japanese pay-for-performance schemes, and Australia’s federally funded telestroke network all tie payment bonuses to imaging performance metrics, turning AI adoption into a revenue-protection strategy. Hospitals unable to demonstrate CT perfusion use or quick thrombolysis face reimbursement penalties, accelerating technology diffusion across mid-tier facilities.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High capital & maintenance cost of scanners | -0.8% | Emerging APAC, MEA, Latin America, small U.S. hospitals | Medium term (2-4 years) |
| Neuroradiologist shortage in emerging areas | -0.6% | Sub-Saharan Africa, Southeast Asia, rural Latin America | Long term (≥ 4 years) |
| Algorithmic-bias delaying AI clearance | -0.4% | United States and European Union | Short term (≤ 2 years) |
| Xenon-CT consumable supply disruptions | -0.2% | North America and Europe | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
High Capital & Maintenance Cost of Advanced Scanners
Photon-counting CT lists at USD 3.2 million versus USD 1.8 million for a 128-slice CT, and annual service contracts add USD 250,000, numbers that exceed the entire equipment budget of many 200-bed hospitals. District-level hospitals in India illustrate the gap: only 14% possess CT, and fewer than 2% run MRI, constraining adoption despite government subsidies.
Shortage of Neuroradiologists in Emerging Markets
Sub-Saharan Africa averages 0.03 neuroradiologists per 100,000 residents compared with 1.2 in North America, forcing general radiologists to interpret complex perfusion scans and inducing variability that dampens thrombectomy referrals. AI can triage, yet regulations still require physician over-reads, so the bottleneck persists.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Diagnostic Technology: CT Dominance Meets AI Disruption
Multimodal CT imaging generated 48.55% of 2025 technology revenue, underscoring its status as the workhorse of the acute ischemic stroke diagnosis market share during hyperacute triage. The acute ischemic stroke diagnosis market size attributed to AI software, though smaller today, is projected to rise fastest because subscription pricing aligns with cash-flow-constrained hospitals. MRI maintains a foothold for posterior fossa strokes and microbleed detection, but its share will erode marginally as photon-counting CT converges on MRI-like resolution with shorter scan times.
AI decision-support software is set to scale at 14.25% CAGR through 2031 because payors reward time-to-treatment performance and radiologist shortages create demand for automated pre-reads. Early adopters report sensitivity exceeding 94% for large-vessel occlusion, while false positives remain under 8%, metrics that justify per-use fees of USD 50-80. The convergence of CT hardware upgrades with cloud-deployed AI suites positions imaging OEMs and pure-play vendors to compete on workflow speed rather than pixel quality alone.

By AI Software Function: Perfusion Quantification Gains Ground
Large-vessel occlusion detection held 40.53% of 2025 AI revenue as every stroke center now prioritizes rapid occlusion identification for thrombectomy routing. Perfusion mismatch quantification tools will capture incremental share because late-window thrombectomy protocols depend on tissue viability rather than elapsed time alone. Vendors packaging occlusion detection, perfusion maps, and hemorrhage exclusion into a single dashboard win contracts by simplifying IT integration. Mobile push notifications reduce team activation times; early data show a 23-minute median reduction in door-to-groin puncture when algorithms auto-page neurointerventional teams.
By Care Setting: Mobile Units Emerge as High-Growth Niche
Hospital emergency departments retained 62.15% share in 2025 because they remain the primary entry point for stroke patients. The acute ischemic stroke diagnosis market will still find its largest installed base inside brick-and-mortar hospitals, yet mobile stroke units are scaling fastest at 15.82% CAGR. Municipalities justify capital outlays of USD 1.2-1.5 million per ambulance by demonstrating 30-50 minute time savings that translate into better functional outcomes and lower long-term disability costs.

By End User: Hospitals Retain Dominance, Outpatient Sites Expand
Hospitals controlled 74.65% of end-user revenue in 2025, reflecting their monopoly on emergency imaging and thrombectomy infrastructure. Outpatient imaging centers and ambulatory surgery centers, however, are growing at 12.32% as value-based care models push volume toward lower-cost settings. The acute ischemic stroke diagnosis industry expects outpatient sites to install non-contrast CT primarily for hemorrhage exclusion before transferring eligible patients, capturing professional fees while avoiding the complexity of full stroke care.
Geography Analysis
North America retained 36.23% global share in 2025 because comprehensive stroke centers exceed 200 and reimbursement is tightly linked to imaging-guided quality metrics. The United States Food and Drug Administration has cleared 12 AI stroke algorithms since 2024, supplying hospitals with multiple vendor options that compete on speed, cloud integration, and subscription price. Canada’s provincial telestroke networks have shortened remote transfer times, while Mexico added 18 CT scanners to regional hospitals but still struggles with a neuroradiologist deficit that slows interpretation turnaround.
The acute ischemic stroke diagnosis market in Asia-Pacific will expand fastest at 11.42% CAGR because China and India are building stroke registries that compel imaging compliance, and private hospital chains are investing heavily in advanced scanners to appeal to medical tourists. China now operates 1,200 stroke centers, a 35% jump since 2022, and ties reimbursement to imaging protocol adherence. India’s 2024-launched cardiovascular and stroke program earmarked USD 145 million for CT procurement and physician training. Japan, already imaging 92% of stroke suspects within 60 minutes, focuses on ultra-high-field MRI to improve microvascular detection in its rapidly aging society.
Europe generated a significant revenue in 2025 as Germany’s 340 certified stroke units standardized multimodal CT perfusion, and the United Kingdom added GBP 45 million worth of scanners and AI licenses to expand thrombectomy access. France issued CE-Mark approval to eight AI stroke detection platforms in 2024, triggering rapid procurement by public and private hospitals. Middle East and Africa remain nascent but attract targeted investment; the United Arab Emirates installed 14 new CT scanners in 2024, and South Africa’s handful of comprehensive stroke centers are exploring AI to amplify limited neuroradiology capacity. South America is led by Brazil’s private chains, yet public hospitals lag, with median door-to-CT times still above 90 minutes.
Competitive Landscape
The acute ischemic stroke diagnosis market is moderately concentrated. Imaging OEMs—Siemens Healthineers, GE HealthCare, Philips, and Canon Medical Systems—leverage their installed hardware bases to bundle AI modules, while pure-play vendors Viz.ai, RapidAI, and Aidoc pursue direct contracts and teleradiology partnerships. Siemens’ Syngo.via stroke module is live at more than 800 hospitals, and 62% of those customers purchase the AI add-on, paying USD 35,000-50,000 annually. GE HealthCare integrated Viz LVO into its Revolution CT, documenting door-to-groin puncture reductions of 21 minutes across 14 validation sites.
White-space innovators focus on portability: NovaSignal’s Lucid robotic ultrasound automates transcranial Doppler in ambulances, and Hyperfine’s USD 50,000 Swoop portable MRI delivers bedside diffusion-weighted images for unstable ICU patients. Patent filings highlight next-generation photon-counting calibration techniques and federated learning architectures that train AI models without centralizing patient data, a privacy-centric design favored under Europe’s AI Act.
Regulatory clearance speed is a key differentiator; vendors with both FDA 510(k) and CE Mark secure premium pricing and faster hospital uptake, while investigational tools face longer sales cycles and require institutional review board oversight.
Acute Ischemic Stroke Diagnosis Industry Leaders
Fujifilm Holdings Corporation
Koninklijke Philips NV
Siemens Healthcare
GE HealthCare
Canon Medical Systems
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: The Indian Council of Medical Research delivered two mobile stroke units to Assam, equipping them with CT scanners, teleconsultation links, point-of-care labs, and thrombolytic drugs to bring pre-hospital imaging to remote districts.
- January 2026: LMU University Hospital researchers reported that circulating brain-derived tau (BD-tau) tracks ischemic injury progression, opening avenues for blood-based monitoring after reperfusion therapies.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the acute ischemic stroke diagnosis market as the annual revenue generated worldwide from imaging systems, ultrasound devices, cerebral angiography suites, and validated blood-based biomarker tests used for first-line or confirmatory detection of an ischemic event within 24 hours of symptom onset. Value is captured at the equipment manufacturer or assay provider list price and excludes therapeutic products, implantable monitoring devices, and post-stroke rehabilitation services.
Scope exclusions, screening tools for hemorrhagic or transient ischemic attack without an ischemic module, are left outside the frame.
Segmentation Overview
- By Diagnostic Technology
- Computed Tomography (NCCT, CTA, CTP)
- Magnetic Resonance Imaging (DWI, SWI, ASL)
- Carotid & Trans-cranial Ultrasound
- Cerebral Angiography (DSA)
- Blood-based Biomarker Tests
- AI-enabled Stroke Decision-Support Software
- By AI Software Function
- Early-Event Triage & Alerting
- Large-Vessel-Occlusion Detection
- Perfusion Mismatch Quantification
- Hemorrhage Exclusion
- By Care Setting
- Emergency Department In-hospital
- Mobile Stroke Unit
- Telestroke Hub & Spoke
- By End User
- Hospitals
- Diagnostic Imaging Centers
- Ambulatory Surgery Centers / Others
- By Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- India
- Japan
- Australia
- South Korea
- Rest of Asia-Pacific
- Middle East and Africa
- GCC
- South Africa
- Rest of Middle East and Africa
- South America
- Brazil
- Argentina
- Rest of South America
- North America
Detailed Research Methodology and Data Validation
Primary Research
Several conversations with neurologists, interventional radiologists, stroke center administrators, and procurement managers across North America, Europe, China, India, and Brazil helped validate imaging mix shifts, average scan pricing, and practical adoption rates of AI decision-support modules. Structured surveys with modality vendors and regional distributors filled data gaps around average selling prices and replacement cycles, guiding assumption fine-tuning.
Desk Research
Mordor analysts first mapped the diagnostic pathway through publicly available guidelines from bodies such as the American Heart Association, European Stroke Organization, and Japan Stroke Society. They then paired that clinical flow with equipment shipment and install data released by regulators (US FDA 510(k) summaries, European CE mark databases) and customs codes aggregated by UN Comtrade, OECD health statistics, and national ministries of health. Company 10-Ks, investor decks, and Factiva-archived earnings calls supplied benchmark selling prices and utilization commentary, while D&B Hoovers aided in cross-checking vendor revenue splits. Peer-reviewed articles in journals like Stroke and the Journal of Neuroimaging were referenced for prevalence-to-diagnosis conversion factors. This list is illustrative; many other sources informed data collection and verification.
Market-Sizing & Forecasting
A single-year top-down build starts with country-level ischemic stroke incidence, adjusts for emergency presentation rates, and multiplies by diagnostic pathway penetration to derive procedure volumes, which are then converted to value using weighted average selling prices. Sampled supplier roll-ups and channel checks offer bottom-up reasonableness. Key variables feeding the model include stroke incidence per 100,000 population, CT and MRI installed base, emergency department turnaround targets, payer reimbursement ceilings, and adoption curves for rapid blood biomarker panels. Forecasts through 2030 rely on multivariate regression that links procedure growth to aging population trends, CT/MRI replacement waves, and health expenditure elasticity under three scenario bands reviewed with primary experts. Gaps in granular shipment data are bridged with regional proxy ratios anchored to historical install-base audits.
Data Validation & Update Cycle
Outputs undergo variance checks against third-party hospital utilization datasets and time-series consistency reviews by a senior analyst. Anomalies trigger re-contacts with sources before sign-off. Reports refresh yearly, with interim reruns when regulatory or reimbursement events materially move the baseline.
Why Mordor's Acute Ischemic Stroke Diagnosis Baseline Stands Firm
Published estimates often diverge because firms differ in stroke type coverage, bundle laboratory panels with imaging in varying ways, or apply distinct currency conversion cadences.
Key gap drivers in other publications revolve around broader stroke categories, inclusion of monitoring equipment, or unvetted average price progression, whereas Mordor restricts scope strictly to acute ischemic diagnostic modalities, applies incidence-driven demand pools, and refreshes base data every twelve months.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 2.36 B (2025) | Mordor Intelligence | - |
| USD 3.99 B (2025) | Global Consultancy A | Bundles lab assays and metabolic panels beyond acute imaging |
| USD 4.57 B (2024) | Trade Journal B | Adds monitoring devices and all stroke categories, not only acute ischemic |
| USD 3.34 B (2025) | Industry Association C | Combines ischemic and hemorrhagic diagnostics, uses broad device ASP escalator |
The comparison shows that figures swell when wider disease categories or device groups are folded in. By selecting a clinically precise scope, grounding volumes in incidence data, and reconciling price points with field interviews, Mordor Intelligence delivers a balanced, transparent baseline that decision-makers can trace back to clear variables and repeatable steps.


Key Questions Answered in the Report
What CAGR is projected for the acute ischemic stroke diagnosis market during 2026-2031?
The market is forecast to expand at 7.27% CAGR over 2026-2031.
Which diagnostic technology currently holds the highest revenue share?
Multimodal CT imaging commanded 48.55% of 2025 revenue.
Which AI software function is growing fastest?
Perfusion mismatch quantification tools are advancing at 16.75% CAGR through 2031.
What geography is expected to grow most rapidly?
Asia-Pacific is projected to increase at 11.42% CAGR through 2031.
How fast are mobile stroke units expanding?
Mobile units are forecast to rise at 15.82% CAGR by 2031 as municipalities invest in pre-hospital imaging.
What is the key restraint hampering scanner adoption in emerging markets?
The high capital and maintenance cost of photon-counting CT and 7 T MRI systems limits uptake in resource-constrained hospitals.
Page last updated on:










