France Diabetes Devices Market Analysis by Mordor Intelligence
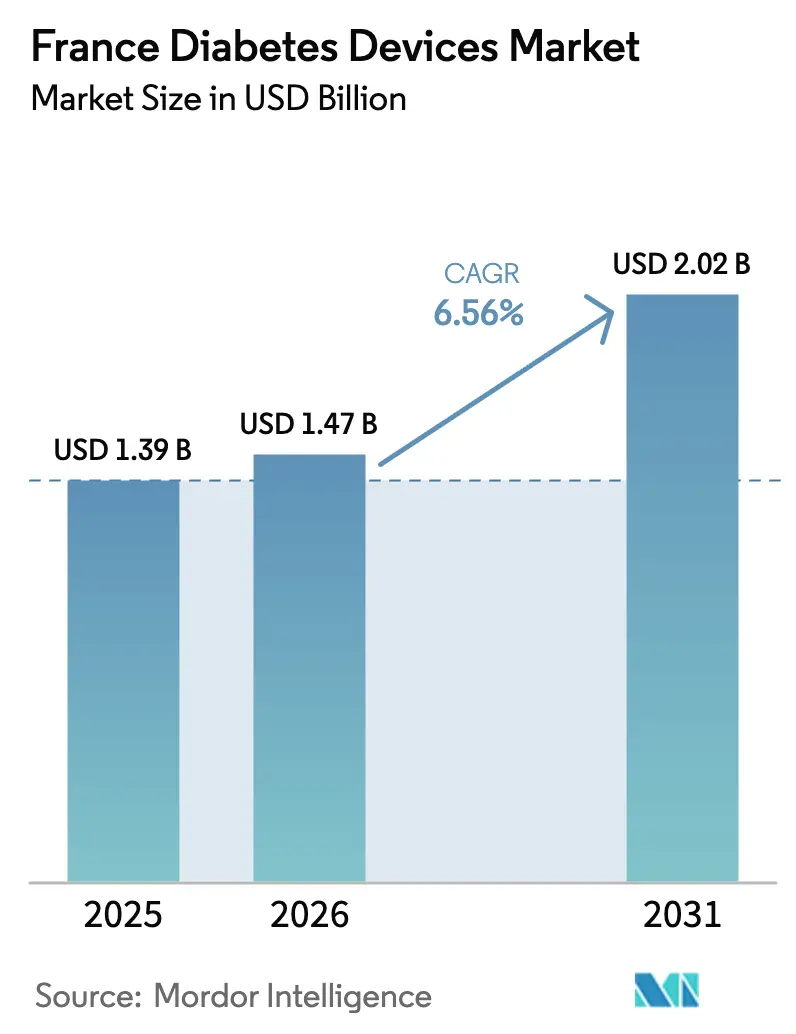
The France Diabetes Devices Market size is projected to expand from USD 1.39 billion in 2025 and USD 1.47 billion in 2026 to USD 2.02 billion by 2031, registering a CAGR of 6.56% between 2026 to 2031.
Reimbursement for continuous glucose monitoring (CGM) in basal-insulin Type 2 patients, strong uptake of tubeless patch pumps, and mandatory telemonitoring quotas are shaping near-term demand. CGM commanded 57.23% revenue in 2025, yet insulin delivery devices show faster 7.87% annual growth as automated insulin delivery (AID) systems penetrate pediatric and adult Type 1 segments. Price-volume clauses negotiated by the Comité Économique des Produits de Santé compress sensor margins, while the December 2025 global FreeStyle Libre 3 recall exposes supply-chain fragility. Online channels, expanding at a 12.57% CAGR, are redrawing distribution as patients embrace subscription fulfillment and direct-to-consumer support. The France diabetes devices market remains moderately concentrated, yet domestic innovators such as Diabeloop and Biocorp inject competitive tension through algorithm and connectivity advances.
Key Report Takeaways
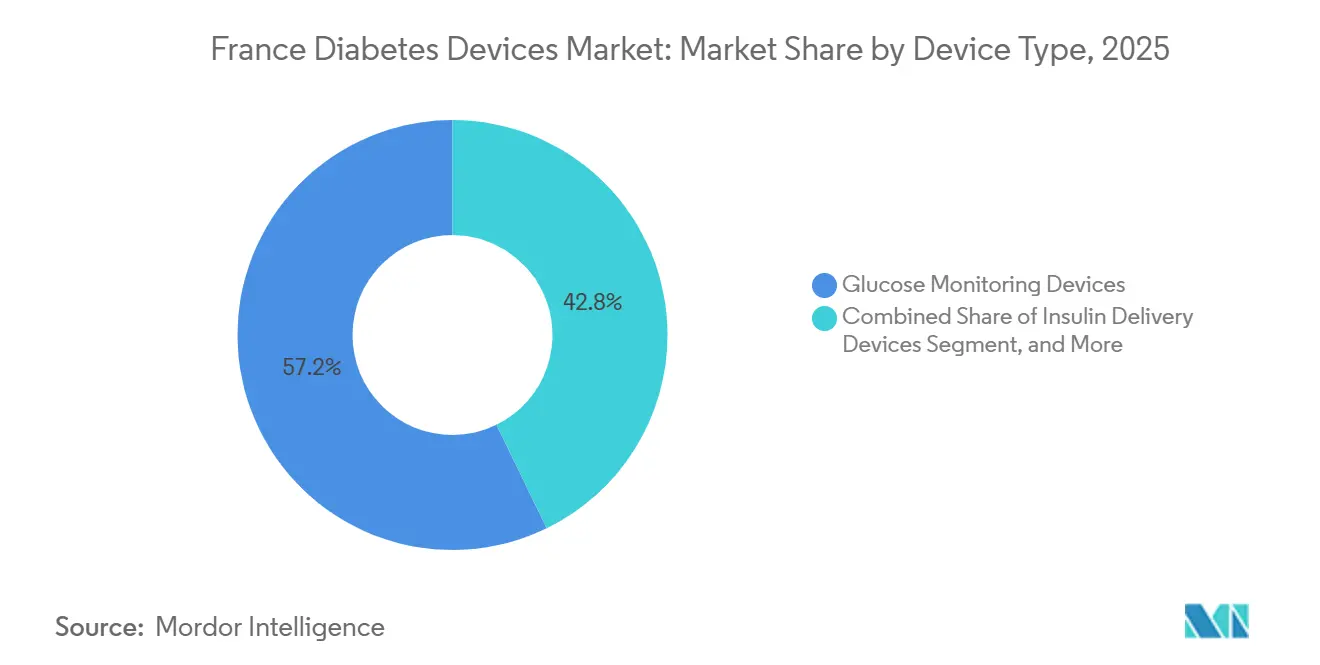
- By device category, CGM held 57.23% of France diabetes devices market share in 2025, while insulin delivery devices are forecast to expand at a 7.87% CAGR through 2031.
- By diabetes type, Type 2 products accounted for 83.03% of 2025 revenue, whereas Type 1 devices are projected to register a 10.73% CAGR through 2031.
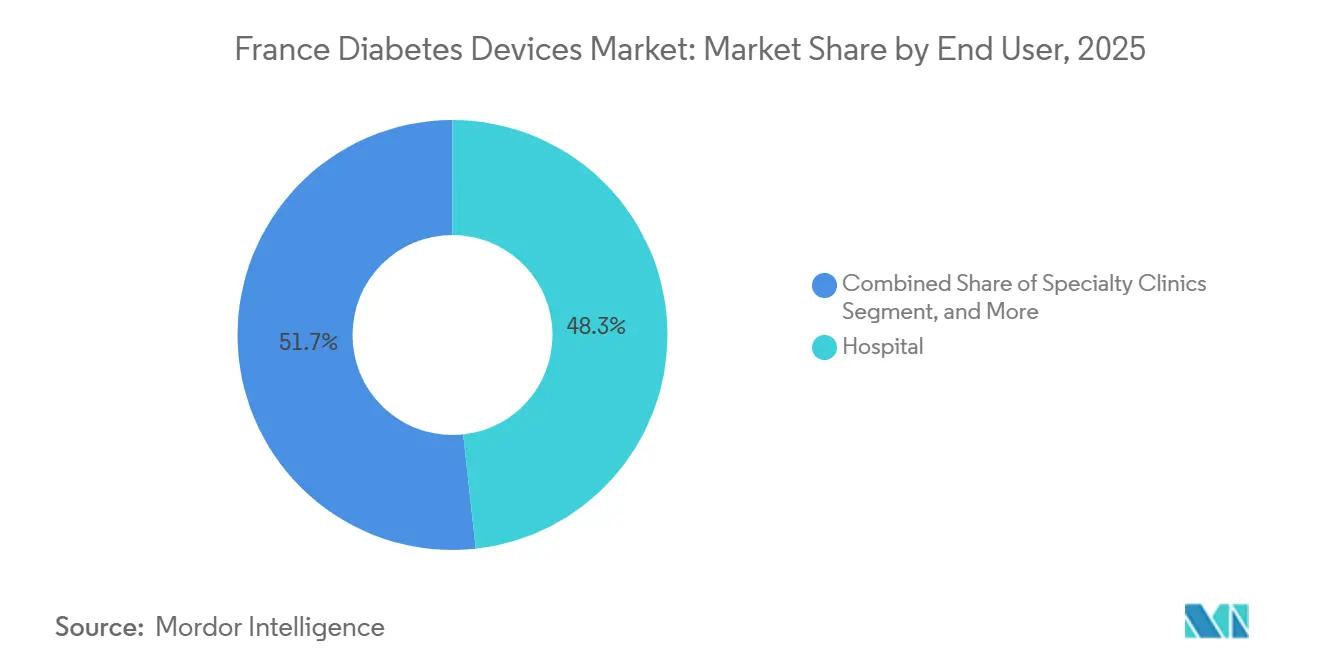
- By end user, hospital led with 48.27% revenue share in 2025, yet home care settings are advancing at a 12.57% CAGR during 2026-2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
France Diabetes Devices Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) %Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Mainstream Reimbursement Expansion for CGM Sensors | +1.8% | National metropolitan areas | Medium term (2-4 years) |
| Rapid Uptake of Tubeless and Patch Insulin Pumps | +1.2% | Type 1 cohorts nationwide | Medium term (2-4 years) |
| Smartphone-Integrated SMBG Ecosystems | +0.6% | Urban France | Short term (≤ 2 years) |
| Rapid GLP-1 Adoption Shifting Device Mix | +1.4% | National | Medium term (2-4 years) |
| Primary-Care Telemonitoring Quotas (LATM) | +0.9% | Rural regions | Short term (≤ 2 years) |
| Diabetic-Elderly Fall-Prevention Mandates | +0.5% | Geriatric facilities | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Mainstream Reimbursement Expansion for CGM Sensors
France approved full reimbursement of Dexcom ONE for 100,000 Type 2 patients on basal insulin in June 2024, opening the first European CGM pathway for non-intensive users. Health-economic studies place the incremental cost-effectiveness ratio within national thresholds, reinforcing payer support.[1]Francisco Gomez-Peralta et al., “Cost-utility of real-time continuous glucose monitoring versus self-monitoring of blood glucose in people with insulin-treated Type II diabetes in France,” PMC, PMC.NCBI.NLM.NIH.GOV As volumes rise, price-volume clauses lower per-sensor reimbursement, limiting manufacturer margins. Integrated sensor-transmitter ecosystems help offset this pressure by locking users into subscription refill models. The 2025 revision of the EU Medical Device Regulation (MDR) embeds post-market surveillance requirements, so real-world CGM outcomes feed directly into reimbursement renewal.
Rapid Uptake of Tubeless and Patch Insulin Pumps
Insulet introduced Omnipod 5 in June 2024, meeting the demand for a discreet, adhesive-based therapy among lifestyle-conscious Type 1 patients. Real-world surveys of 14,765 users show satisfaction driven by convenience rather than superior glycemic metrics. Grenoble-based Diabeloop secured EUR 15 million in 2024 to speed DBLG1 adoption and prepare the smartphone-native DBLG2, which gained FDA clearance by January 2026. The MiniMed 780G spin-off announced by Medtronic in June 2025 underscores escalating competition as agile software innovators shorten upgrade cycles. Future pump growth depends on algorithmic personalization, patch-format miniaturization, and bundle offers that pair hardware, sensor, and support.
Smartphone-Integrated SMBG Ecosystems
Pen-cap sensors such as Biocorp’s Mallya, acquired by Novo Nordisk for EUR 154 million in 2023, automatically log insulin doses and transmit data to mobile dashboards. Abbott and Biocorp are aligning FreeStyle Libre data with connected pens, reinforcing platform stickiness. Smartphone-enabled self-monitoring blood glucose (SMBG) meters still serve gestational and early Type 2 cases but face declining volumes as CGM reimbursement widens. Device makers counter commoditization by embedding coaching nudges and predictive analytics into companion apps. National Health Data System analysis links CGM adoption with switches to longer-acting insulins, illustrating how digital data reshapes prescribing behaviors.
Rapid GLP-1 Adoption Shifting Device Mix
Glucagon-like peptide-1 receptor agonists enable down-titration or cessation of basal insulin, shrinking pen and strip demand. Yet CGM placement is still rising because clinicians exploit sensor trends to optimize GLP-1 titration and detect nocturnal lows. Once-weekly basal insulins further trim injection frequency, prompting pen makers to focus on reusable hardware and connectivity. The July 2025 carbon-footprint methodology rates an insulin pen at 0.7 kg CO₂eq, adding environmental scoring to procurement and favoring low-material options.[2]Direction Générale des Entreprises, “Carbon Footprint of Medicines: Assessment Methodology,” ENTREPRISES.GOUV.FR Manufacturers must therefore balance environmental impact with feature differentiation.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Price-Volume Clauses Squeezing Margins | -0.9% | National | Medium term (2-4 years) |
| Sensor Cybersecurity and GDPR Compliance Burden | -0.4% | EU aligned | Short term (≤ 2 years) |
| GLP-1-Driven Reduction in Basal-Insulin Users | -0.7% | National | Medium term (2-4 years) |
| CEPS Claw-Backs Linked to Carbon Footprint KPIs | -0.3% | Pilot procurement | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Price-Volume Clauses Squeezing Margins
French price tariffs fall once sales thresholds are crossed, curbing profit despite rising CGM penetration. Dexcom ONE reimbursement illustrates this pinch: higher volumes trigger steeper rebates. Manufacturers introduce premium variants with longer wear or predictive alerts to escape existing caps. The 2025 FreeStyle Libre 3 recall shows how speed-to-market gambits carry quality risk that can magnify financial strain.[3]European Commission, “Guidance on post-market surveillance of medical devices and in vitro diagnostic medical devices: MDCG 2025-10,” HEALTH.EC.EUROPA.EU
Sensor Cybersecurity and GDPR Compliance Burden
Connected pumps and sensors must notify CSIRTs and ENISA of exploited vulnerabilities within 30 days of December 2025, adding audit and patching costs. Data hosting must meet GDPR and ISO 13485 requirements, increasing fixed overhead that smaller firms struggle to cover.
Segment Analysis
By Device Category: CGM Dominance and Pump Upswing
The French diabetes devices market size remained at 57.23% of the French diabetes devices market share that year. Sensors remain volume leaders, yet their growth trails pumps, as margin pressure from price-volume clauses tempers revenue gains. AID-capable patch pumps underpin the 7.87% CAGR in insulin delivery, helped by Omnipod 5 and DBLG1 rollouts. Self-monitoring glucometers continue to lose ground as CGM reimbursement widens and fingerstick avoidance intensifies.
Second-generation pumps are split between tethered (MiniMed 780G, Tandem Control-IQ) and tubeless (Omnipod 5, Kaleido) designs. Recall-driven sensor shortages can temporarily re-inflate SMBG demand, but a structural shift favors sensors and smart pumps.

By Diabetes Type: Expanding Type 1 AID and Broadening Type 2 CGM
Type 2 products accounted for 83.03% of revenue in 2025, and type 1 diabetes is set to grow faster, with a 10.73% CAGR, as AID becomes standard of care. OB2F registry data indicated MiniMed 780G and Control-IQ each held close to half of installed AID bases, with only 2.8% discontinuation, confirming high persistence. Type 2 devices, despite owning the majority France diabetes devices market share, grow more slowly because GLP-1 usage reduces injection frequency. Pandemic-accelerated telemonitoring, however, lifts sensor adoption among basal-only cohorts, balancing the insulin volume drop.
Gestational and other forms of diabetes remain a niche. Once national CGM thresholds are widened to cover oral-therapy populations, Type 2 sensor revenue should accelerate, though pen volumes may not recover. Carbon-footprint scoring may further tilt purchasing toward reusable pens within the France diabetes devices industry.
By End User: Home-Care Settings Gain Momentum
Hospitals and clinics accounted for 48.27% of revenue in 2025, reflecting the concentration of specialist expertise and peri-operative glucose monitoring. Closed-loop initiation during inpatient stays sustains pump attachment rates, while CEPS procurement channels favor bulk purchases. Yet staff shortages, especially outside metro hubs, press facilities to offload stable patients sooner, indirectly bolstering remote monitoring adoption across the French diabetes devices market.
Home-care settings are recording the fastest growth at a 12.57% CAGR through 2030. Reimbursement introduced in July 2023 allows physicians to bill remote glucose reviews via the PPDP program. Seniors equipped with tablets receive coaching from regional diabetes-nurse networks, reducing travel costs. The France diabetes devices market share for home settings is poised to surpass 30% by 2029 as broadband penetration widens. Pharmacies and ambulatory surgical centers add secondary volumes: pharmacies handle lancets and low-cost glucometers, while day-surgery centers offer sensor insertions and pump starts, creating cross-selling avenues for multinational suppliers focused on the France diabetes devices market.

Geography Analysis
Abbott, Dexcom, Medtronic, Insulet, and Novo Nordisk dominate most revenue streams, but the December 2025 Libre 3 recall allowed Dexcom to capture share amid supply gaps. Novo Nordisk bolstered its connected-device portfolio via the Biocorp purchase, integrating Mallya pen caps into its insulin ecosystem.
Insulet leverages tubeless convenience to woo Type 1 converts, while Diabeloop’s algorithmic expertise targets both hardware-paired (DBLG1) and phone-only (DBLG2) AID niches, reporting 3,500 European users by 2026. Medtronic’s MiniMed spin-off seeks agility to counter rapid software iterations from digital natives.
Sensor and pump makers race to harden cybersecurity, cut carbon footprints, and secure ISO 13485 re-certification ahead of 2028 MDR roll-over deadlines. White-space opportunities include implantable CGM, where Senseonics filed for CE mark of Eversense 365 in February 2026, promising 12-month wear and fewer insertions.
Competitive Landscape
The France diabetes devices market blends global titans and agile local entrants. Abbott, Dexcom, and Medtronic lead unit sales, jointly accounting for a significant share of 2025 revenue. Medtronic’s August 2024 data-sharing pact with Abbott joins Libre sensors to MiniMed pumps, unifying patient dashboards and clinician portals. Dexcom differentiates with 10-day G7 sensors that warm up in 30 minutes and auto-pair to smartphones, while Abbott counters with 1-hour warm-up Libre 3 at a lower list price, sustaining competitive tension that benefits payers.
Domestic innovators capitalize on France 2030 grants. Grenoble-based Sensorion develops silicon-nitride membranes extending sensor life to 21 days, targeting CE mark by 2026. Paris start-up GlucoSurf integrates haptic alerts for visually impaired users, piloting devices under PECAN fast-track before nationwide roll-out. These firms often license patents to majors, gaining channel access while adding diversity to the French diabetes devices market.
Strategic manufacturing investments amplify local presence. Novo Nordisk expanded a Penfill cartridge plant in Chartres in 2024 to serve European demand. Embecta outsources cannula molding to a Dijon-based contract manufacturer that recently installed EU-MDR-compliant clean rooms, cutting lead times for European deliveries. Competitive differentiation shifts from hardware alone to service layers: remote coaching, A1c predictive analytics, and seamless “Mon Espace Santé” integration. GLP-1 receptor agonists reshape device usage, but CGM enhances titration efficacy; a 2024 study showed A1c declines of 0.5 percentage points when GLP-1 therapy was paired with CGM. Manufacturers therefore position sensors as medication-agnostic companions, sustaining relevance inside the France diabetes devices market.
France Diabetes Devices Industry Leaders
Dexcom
Novo Nordisk
Medtronic
Roche Diabetes Care
Abbott Diabetes Care
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2025: Glucotrack joined the FORGETDIABETES consortium to co-develop a bionic pancreas.
- January 2025: Glooko received French reimbursement for remote gestational-diabetes monitoring, expanding digital-health coverage.
France Diabetes Devices Market Report Scope
More user-friendly and unobtrusive devices are available, from improved insulin pumps to continuous glucose monitors (CGM) and blood glucose meters. The France diabetes device market comprises component management devices and monitoring devices. The report offers the value (in USD) and volume (in units) for the above segments.
| Glucose Monitoring Devices | Self-Monitoring Blood Glucose (SMBG) Devices | Glucometers |
| Test Strips | ||
| Lancets | ||
| Continuous Glucose Monitoring (CGM) Devices | Sensors | |
| Durables (Receivers & Transmitters) | ||
| Insulin Delivery Devices | Insulin Pens | |
| Insulin Pumps | ||
| Insulin Syringes | ||
| Jet Injectors | ||
| Other Diabetes-Care Devices | ||
| Type 1 Diabetes |
| Type 2 Diabetes |
| Gestational & Others |
| Hospitals |
| Specialty Clinics |
| Home Care Settings |
| Other End Users |
| By Device Category | Glucose Monitoring Devices | Self-Monitoring Blood Glucose (SMBG) Devices | Glucometers |
| Test Strips | |||
| Lancets | |||
| Continuous Glucose Monitoring (CGM) Devices | Sensors | ||
| Durables (Receivers & Transmitters) | |||
| Insulin Delivery Devices | Insulin Pens | ||
| Insulin Pumps | |||
| Insulin Syringes | |||
| Jet Injectors | |||
| Other Diabetes-Care Devices | |||
| By Diabetes Type | Type 1 Diabetes | ||
| Type 2 Diabetes | |||
| Gestational & Others | |||
| By End User | Hospitals | ||
| Specialty Clinics | |||
| Home Care Settings | |||
| Other End Users | |||


Key Questions Answered in the Report
What is the projected value of the France diabetes devices market by 2031?
The market is forecast to reach USD 2.02 billion by 2031 at a 6.56% CAGR.
Which device segment is expanding faster than the overall market?
Insulin delivery devices, propelled by tubeless patch pumps and AID systems, are growing at a 7.87% CAGR.
How did France change CGM reimbursement in 2024?
National coverage was extended to 100,000 basal-insulin Type 2 patients, boosting CGM volumes.
Why are home care settings gaining share by end user?
Home care settings fulfillment drive a 12.57% CAGR during the forecast period.
What regulatory change in 2025 affects connected diabetes devices?
The EU MDR revision introduces mandatory cybersecurity incident reporting within 30 days.
Page last updated on: