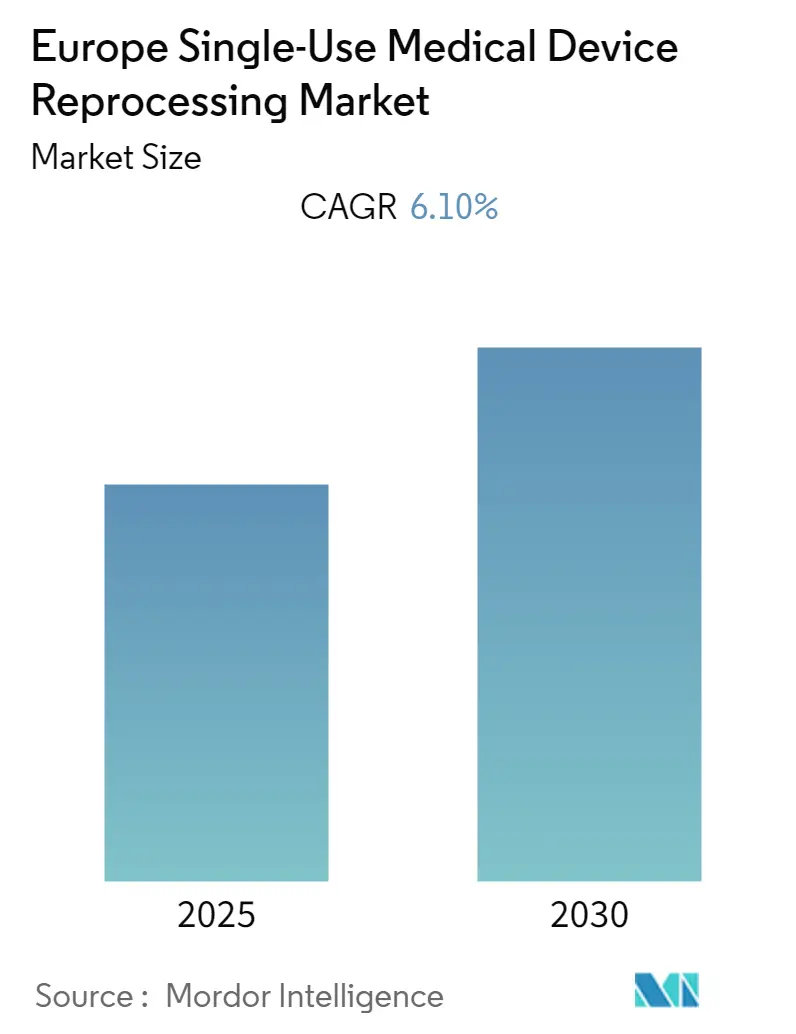
Europe Single-Use Medical Device Reprocessing Market Analysis by Mordor Intelligence
The Europe Single-Use Medical Device Reprocessing Market is expected to register a CAGR of 6.1% during the forecast period.
The COVID-19 pandemic resulted in postponements and cancellations of non-essential and elective procedures, such as dental procedures and orthopedic surgeries, which led to a significant decline in the number of surgeries during the pandemic. This led to a significant decline in the demand for reprocessed medical devices. For instance, according to an article published by PubMed Central in January 2022, during the first COVID-19 pandemic wave, elective procedures were significantly reduced in Europe to increase critical care capacity for patients with COVID-19 and release the surgical team to support other medical areas in need, including intensive care. Hence, the pandemic significantly impacted the demand for surgical medical devices, including reprocessed devices. However, as the pandemic subsided, the market studied is expected to witness stable growth during the forecast period.
Rising clinical urgency to minimize the generation of medical waste in healthcare settings and hospitals, cost savings through reprocessing single-use devices, and regulatory pressure to reduce volume of medical waste are among the key factors driving the market growth. The lowering of the overall cost of healthcare through the reprocessing of single-use devices is also a primary factor driving this market. In Germany, reprocessing has been regulated for over a decade with a large number of hospitals showing significant cost savings. It has been strictly subjected to monitoring and surveillance by public authorities and the European Union (EU)-accredited notified body. Similarly, in the United Kingdom, the Medicines and Healthcare products Regulatory Agency (MHRA) legalized the use and manufacture of reprocessed single-use medical devices for use. In addition, it helps insurers and hospitals for better management and reduced healthcare costs with the improvement of the healthcare system.
Furthermore, the rising number of surgeries taking place is also a major factor driving the growth of the market as it is increasing the demand for reprocessed single-use medical devices. For instance, according to the data published by NHS in 2021, an estimated 64,211 surgical procedures were recorded by the public health of Engalnd. Hence, it is expected to increase the adoption of reprocessed medical devices.
Moreover, along with cost reduction, it also results in environmental sustainability. SUD reprocessing is one of the top healthcare supply chain strategies used to reduce costs and optimize resources. The aforementioned factors such as the rising number of surgeries taking place, rising clinical urgency to minimize the generation of medical waste in healthcare settings and hospitals, and the lowering of the overall cost of healthcare through the reprocessing of single-use devices, are anticipated to contribute towards the development of market in the immediate future.
However, the potential of material alteration and cross infection with reprocessed device and preconceived notions regarding the quality of reprocessed single-use medical devices (SUDs) are the major factors that are expected to impede the growth of this market.
Europe Single-Use Medical Device Reprocessing Market Trends and Insights
Scalpels Segment is Poised to Register Significant Growth During the Forecast Period
A scalpel is a small, sharp-bladed instrument used for surgical purposes in various clinical settings. The major factors driving the growth of the scalpel segment are the increasing prevalence of diseases that requires surgeries and the growing number of surgeries in Europe.
For instance, according to the data by Breast Cancer in 2021, an estimated 55,000 women and 370 men are diagnosed with breast cancer annually in the United Kingdom. Out of which, around 46,000 people in England, 4,700 people in Scotland, 2,800 people in Wales, and 1,500 people in Northern Ireland are diagnosed with breast cancer every year. Hence, as scalpels are majorly used in the surgical process involving breast cancer, the demand for reprocessed scalpels is expected to rise with the rising incidence of breast cancer in the United Kingdom.
Furthermore, according to the data by Penningtons Manches Cooper LLP in February 2022, a cesarean section surgery is done when the baby is delivered through an abdominal surgery rather than pushed through the birth canal, and around 25% of births in the United Kingdom are cesarean sections. As scalpels are often used for skin incisions during cesarean surgeries, the region's high number of cesarean sections is expected to enhance segment growth.
Hence, the aforementioned factors, such as the increasing prevalence of diseases requiring surgeries and the large number of surgeries taking place in Europe, are expected to boost the segment growth.
Germany is Expected to Hold a Significant Share in the Market During the Forecast Period
Factors such as the increasing number of hospitals in the country, the rising prevalence of diseases that require surgery, and the rising number of surgeries taking place in Germany are expected to drive the growth of the market in the country.
For instance, according to an article published by Springer in September 2022, Germany has an estimated incidence of approximately 69,000 breast cancer cases every year. It is also considered the most common cancer type in women in Germany. Moreover, according to an article published by Elsevier in September 2022, colorectal cancer is the third most common cancer in Germany. Hence, due to the increasing prevalence of such diseases, the adoption of reprocessed devices for surgeries in these diseases is expected to increase during the forecast period of the study. Moreover, as old age is associated with the rising prevalence of various diseases, the increasing geriatric population in the country is also a significant factor driving the market growth.
According to the data published by Destatis in March 2021, the rate of cesarean section surgeries in Germany was 29.6% in 2021. The high rate of cesarean sections performed in the country is expected to increase the demand for reprocessed medical devices as they are often required for cesarean surgery.
Hence, the aforementioned factors, such as the rising prevalence of various diseases requiring surgery, the rising number of surgeries, and the increasing geriatric population, are expected to boost the market growth in Germany.
Competitive Landscape
The European single-use medical device reprocessing market is moderately competitive and has several major players. A few of the major players currently dominate the market in terms of market share. The major market players include Stryker Corporation, Medline Industries Inc., Arjo, and Sterilmed Inc. (Johnson & Johnson), among others.
Europe Single-Use Medical Device Reprocessing Industry Leaders
Stryker Corporation
Medline Industries, Inc
Sterilmed Inc. (Johnson & Johnson)
Arjo
NEScientific
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- June 2022: The Medicines and Healthcare products Regulatory Agency of the United Kingdom advised against the reprocessing of single-use medical devices in its guidance.
- May 2022: The President of Poland signed the Act on Medical Devices. Under the new law, reprocessing of single-use medical devices has been allowed in Poland.
Europe Single-Use Medical Device Reprocessing Market Report Scope
As per the scope of the report, single-use medical device (SUD) reprocessing includes cleaning, disinfection, and sterilization, as well as testing and restoration of the technical and functional safety of the used device. Reprocessing of SUDs makes it possible for hospitals to maintain patient care quality while saving substantial amounts of money. The European single-use medical device reprocessing market is segmented by device type (class I devices and class II devices) and geography (Germany, United Kingdom, France, Italy, Spain, and the Rest of Europe). The report offers the value (in USD million) for the above segments.
| Class I Devices | Laparoscopic Graspers |
| Scalpels | |
| Tourniquet Cuffs | |
| Other Class I Devices | |
| Class II Devices | Pulse Oximeter Sensors |
| Sequential Compression Sleeves | |
| Catheters and Guidewires | |
| Other Class II Devices |
| Germany |
| United Kingdom |
| France |
| Italy |
| Spain |
| Rest of Europe |
| By Device Type | Class I Devices | Laparoscopic Graspers |
| Scalpels | ||
| Tourniquet Cuffs | ||
| Other Class I Devices | ||
| Class II Devices | Pulse Oximeter Sensors | |
| Sequential Compression Sleeves | ||
| Catheters and Guidewires | ||
| Other Class II Devices | ||
| By Geography | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||


Key Questions Answered in the Report
What is the current Europe Single-Use Medical Device Reprocessing Market size?
The Europe Single-Use Medical Device Reprocessing Market is projected to register a CAGR of 6.1% during the forecast period (2025-2030)
Who are the key players in Europe Single-Use Medical Device Reprocessing Market?
Stryker Corporation, Medline Industries, Inc, Sterilmed Inc. (Johnson & Johnson), Arjo and NEScientific are the major companies operating in the Europe Single-Use Medical Device Reprocessing Market.
What years does this Europe Single-Use Medical Device Reprocessing Market cover?
The report covers the Europe Single-Use Medical Device Reprocessing Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Europe Single-Use Medical Device Reprocessing Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:








