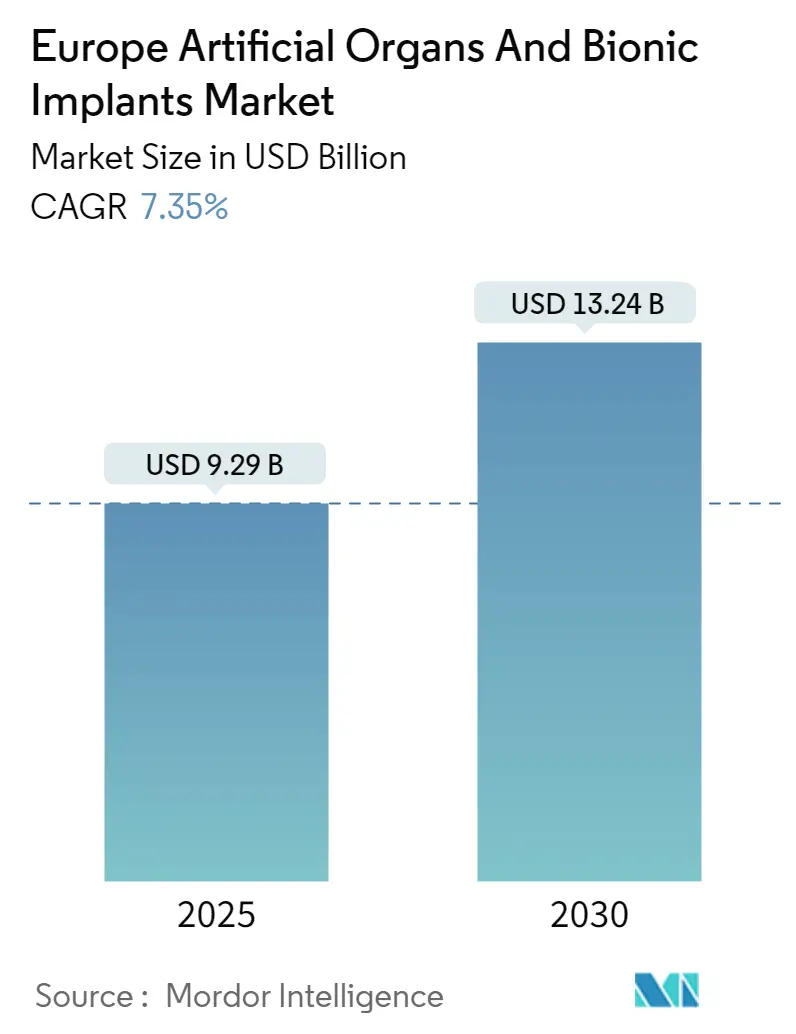
Europe Artificial Organs And Bionic Implants Market Analysis by Mordor Intelligence
The Europe Artificial Organs And Bionic Implants Market size is estimated at USD 9.29 billion in 2025, and is expected to reach USD 13.24 billion by 2030, at a CAGR of 7.35% during the forecast period (2025-2030).
Factors such as the rising incidence of organ failures and disabilities, scarcity of donor organs, and technological advancements leading to enhanced applications are anticipated to drive market growth over the forecast period.
Major organs in a body, such as kidneys, liver, lungs, heart, central nervous system, and hematologic system, tend to fail among the target population. In Europe, the number of organ failures has been increasing consistently over the past few decades, but the number of organ donors remains few. This is creating demand for artificial organs in the region. For instance, a study published in the World Congress of Nephrology in April 2024 predicts a significant rise in advanced-stage chronic kidney disease cases (up to 59.3%) by 2032 across eight major countries, including the United Kingdom, Spain, Germany, and the Netherlands.
Furthermore, according to the same source, dialysis requirements are expected to surge by over 75%, increasing the economic and environmental burden. These factors are expected to create opportunities for adopting artificial organs and bionic implants, specifically artificial kidneys, to address the growing CKD crisis. Hence, the market is expected to grow considerably over the analysis period.
With a strong focus on research and development, coupled with growing demand due to an aging population and rising chronic diseases, this market is expected to experience significant growth in the coming years. For instance, according to the European Union in May 2023, the Breakthrough Technologies for an Implantable Artificial Kidney project funded with EUR 3.38 million (USD 3.63 million) by the Horizon European Innovation Council (EIC) in 2022 exemplified the ongoing research efforts toward a viable alternative. This indicates a strong focus on developing implantable artificial kidneys in Europe. Therefore, the European artificial organs and bionic implants market is poised for significant growth, driven by increasing funding for research.
In addition, the development of innovative dialysis technologies, including portable, wearable, and implantable artificial kidney systems, is set to transform this market, offering more effective and patient-friendly solutions. For instance, a study published in Nature Reviews Nephrology in June 2023 examined the integration of bioartificial kidneys (BAKs) with advanced membranes in the European market. This study emphasized BAKs, which merge artificial membranes with functional kidney cells and aim to restore critical biological functions that are typically managed through conventional dialysis. These functions include erythropoietin production, which is vital for red blood cell production, and vitamin D synthesis, which is essential for calcium metabolism and bone health. The study underscores the potential of these bioartificial systems to enhance patient outcomes by offering a more physiologically relevant alternative to traditional dialysis methods.
Thus, innovations such as portable and wearable artificial kidneys, advanced dialysis membranes, and bioartificial kidneys are poised to meet the growing demand, and they are anticipated to contribute to the growth of the artificial organs and bionic implants market over the forecast period. However, expensive procedures and fear of device malfunction and its consequences are expected to hinder the market's growth.
Europe Artificial Organs And Bionic Implants Market Trends and Insights
The Artificial Kidney Segment is Expected to Register Significant Growth During the Forecast Period
An artificial kidney is a wearable dialysis machine. An individual with end-stage renal disease may use one daily or even continuously. The increasing incidence and prevalence of renal failures in Europe are expected to drive the segment's growth.
Kidney disease has become a burden that is leading to kidney failure and causing a significant healthcare concern in the region in recent years. For instance, the 2023 International Society of Nephrology (ISN) Global Kidney Health Atlas (ISN-GKHA) reported that the prevalence of chronic kidney disease (CKD) in Western Europe reached 10.6% in 2023, slightly exceeding the global median. Thus, the burden of CKD is expected to create demand for the adoption of artificial kidneys for patient survival, thereby fueling the segment's growth during the forecast period.
Furthermore, organ donation and transplantation remain the foremost and cost-effective clinical solutions for end-stage organ failure. For instance, in January 2023, NextKidney secured EUR 4.1 million (USD 4.5 million) through one of the largest crowdfunding efforts in a decade on Oneplanetcrowd. This substantial investment is set to revolutionize the lives of kidney patients by bringing the world’s first portable hemodialysis device, the Neokidney, to market. Hence, the increasing interest of the public and investors in technologies is driving the growth of the artificial kidney segment within the European artificial organs and bionic implants market.
In conclusion, innovations in artificial kidney structures, increasing funds, and the growing burden of chronic kidney diseases (CKDs) are expected to boost the growth of the market during the forecast period.


The United Kingdom is Anticipated to Contribute Significantly to the Market Growth
Among the other countries, the United Kingdom is anticipated to witness growth during the analysis period. Factors such as increasing disability and accidental injuries requiring amputations, such as limbs, fingers, and hands, are major contributors to the growth of the artificial organs and bionic implants market in the United Kingdom.
Aging, diabetes, and vascular diseases are all predicted to cause an increase in the number of amputations. Amputations are among the most destructive complications associated with diabetes, and the number of major lower limb amputations in diabetes is increasing in the country. As per the datasheet published in June 2022, a total of 7,957 major diabetic lower limb amputations were reported in a period of four years, and the cost of healthcare for ulceration and amputation in diabetes is approximately USD 1,230 million (GBP 1 billion) per year in the United Kingdom. Thus, the burden of lower limb amputations is expected to accelerate the growth of the artificial organs and bionic implants market in the country.
The UK healthcare industry is witnessing significant advancements in the market studied, driven by the integration of cutting-edge technologies to improve patient outcomes. For instance, in November 2023, the National Health Service announced that over 150,000 adults and children with type 1 diabetes in England and Wales will be offered an artificial pancreas, a groundbreaking device that uses a hybrid closed-loop system to automate insulin delivery based on real-time blood glucose readings. The device uses advanced algorithms to continuously monitor blood glucose levels and automatically administer the precise amount of insulin needed to maintain optimal levels.
Therefore, increasing adoption of such advanced technology in the United Kingdom is expected to drive the European artificial organs and bionic implants market.
In addition, according to the UK Parliament, in December 2023, organ donation activity fell severely in the region. The overall consent/authorization rate for organ donation dropped from 66% to 62% during 2022/23. A significant disparity was observed in consent rates between white patients (65%) and ethnic minority groups (35%). This increase in transplant waitlists signifies a substantial population of patients for whom traditional transplants are not readily available. This creates a market for artificial organs and bionic implants as patients seek alternative treatments, thereby driving the demand in the country.
Thus, due to the shortage of organs for transplantation, the demand for advanced artificial organs is increasing in the country, further fueling the overall market.


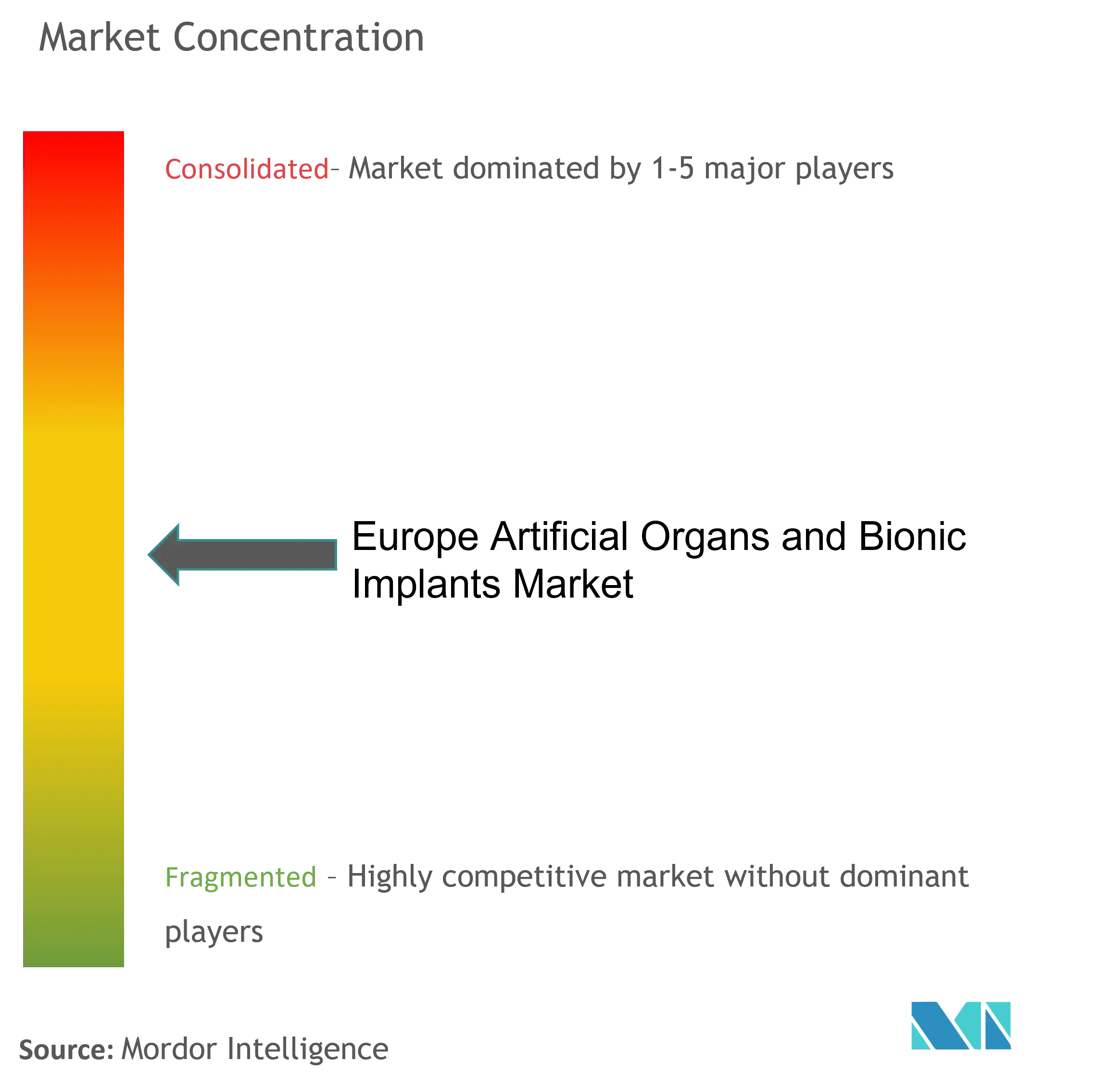
Competitive Landscape
The European artificial organs and bionic implants market is semi-consolidated with several major players. Some strategies implemented by market players include agreements, collaborative models, business expansion, and product development. Most market players focus on bringing technologically advanced products for maximum market share. The key market players include Sonova, Medtronic, Ekso Bionics, Ossur, and Ottobock.
Europe Artificial Organs And Bionic Implants Industry Leaders
Medtronic plc
Zimmer Biomet
Ottobock
Sonova
Ossur
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- June 2024: PrintyMed, a Latvian biomedical startup, made a significant breakthrough by developing biocompatible artificial spider silk. This innovative biomaterial has the potential to revolutionize various medical applications, particularly in the field of artificial organs.
- January 2024: Carmat, a French medical technology company, announced a key development in its artificial heart technology. The company launched a software enhancement for its Aeson heart, designed to significantly improve its safety profile. The new software allows the Aeson heart to detect malfunctions in real time. This is a critical feature as it enables prompt intervention and avoids serious complications.


Europe Artificial Organs And Bionic Implants Market Report Scope
As per the scope of the report, artificial organs are mechanical devices that can temporarily or permanently substitute for a body organ. These devices can replicate or augment the organ's natural function. Bionics refers to the replacement or improvement of body parts by mechanical means.
The European artificial organs and bionic implants market is segmented by type (artificial organs and bionics) and geography (Germany, the United Kingdom, France, Italy, Spain, and Rest of Europe). The report offers market sizes and forecasts in terms of value (USD) for all the above segments.
| Artificial Organs | Artificial Heart |
| Artificial Kidney | |
| Cochlear Implants | |
| Other Artificial Organs | |
| Bionics | Vision Bionics |
| Ear Bionics | |
| Orthopedic Bionic | |
| Cardiac Bionics | |
| Other Bionic Types |
| Germany |
| United Kingdom |
| France |
| Italy |
| Spain |
| Rest of Europe |
| By Product | Artificial Organs | Artificial Heart |
| Artificial Kidney | ||
| Cochlear Implants | ||
| Other Artificial Organs | ||
| Bionics | Vision Bionics | |
| Ear Bionics | ||
| Orthopedic Bionic | ||
| Cardiac Bionics | ||
| Other Bionic Types | ||
| Geography | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||


Key Questions Answered in the Report
How big is the Europe Artificial Organs And Bionic Implants Market?
The Europe Artificial Organs And Bionic Implants Market size is expected to reach USD 9.29 billion in 2025 and grow at a CAGR of 7.35% to reach USD 13.24 billion by 2030.
What is the current Europe Artificial Organs And Bionic Implants Market size?
In 2025, the Europe Artificial Organs And Bionic Implants Market size is expected to reach USD 9.29 billion.
Who are the key players in Europe Artificial Organs And Bionic Implants Market?
Medtronic plc, Zimmer Biomet, Ottobock, Sonova and Ossur are the major companies operating in the Europe Artificial Organs And Bionic Implants Market.
What years does this Europe Artificial Organs And Bionic Implants Market cover, and what was the market size in 2024?
In 2024, the Europe Artificial Organs And Bionic Implants Market size was estimated at USD 8.61 billion. The report covers the Europe Artificial Organs And Bionic Implants Market historical market size for years: 2021, 2022, 2023 and 2024. The report also forecasts the Europe Artificial Organs And Bionic Implants Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Europe Artificial Organs And Bionic Implants Market Report
Statistics for the 2025 Europe Artificial Organs And Bionic Implants market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Europe Artificial Organs And Bionic Implants analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.



