Electronic Trial Master File (ETMF) Market Size and Share
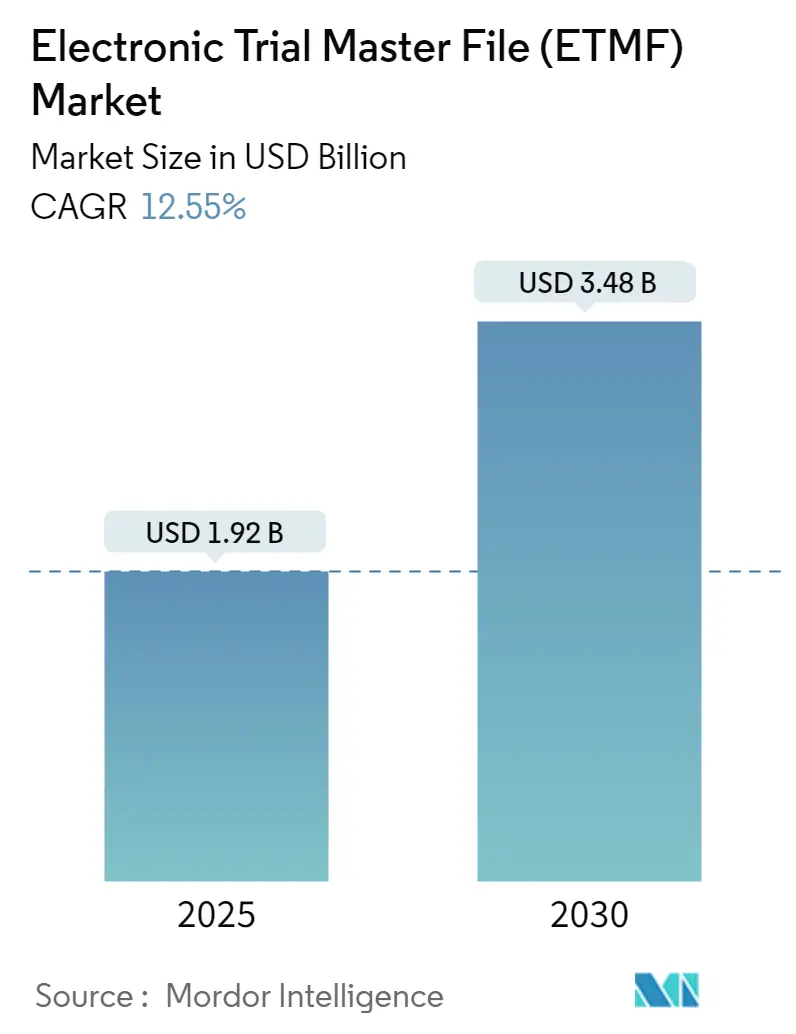
Electronic Trial Master File (ETMF) Market Analysis by Mordor Intelligence
The Electronic Trial Master File Market size is estimated at USD 1.92 billion in 2025, and is expected to reach USD 3.48 billion by 2030, at a CAGR of 12.55% during the forecast period (2025-2030).
- With the COVID-19 pandemic, there was a surge in the number of clinical trials across the globe. Major companies focused on developing effective vaccines and therapies for COVID-19 disease prevention and treatment. As per the 2022 data from clinicaltrials.gov, there were more than 8,283 studies from 150 countries registered on clinicaltrials.gov to date. The above-mentioned studies included around 777 vaccine studies and 2,155 drug studies. This indicated the hike in data generated during the COVID-19 pandemic regarding clinical trials. Hence, the utilization of the electronic trial master file was increased to keep track of clinical trial data generated during the pandemic, which helped the market grow significantly. However, initiatives by key market players were expected to increase market growth.
- For instance, in April 2021, Phlexglobal stated that an increasing number of pharmaceutical companies, including five of the global Top 10, utilized Phlexglobal's innovative TMF Quality Review solution to identify and mitigate potential regulatory risk for mergers & acquisitions and improve inspection readiness.
- The electronic trial master file is a novel application that leverages software and server technology to assist in the setup, collection, storage, tracking, and archival of essential clinical study documents. With increasing clinical trials across the globe, it is essential to keep clinical data secure and in a form that can be easily accessed and understood. In addition, to cope with increasing clinical trials, companies are focusing more on bringing advanced and easy-to-use software that can reduce the burden of huge data generated through clinical trials. In addition, the healthcare industry is witnessing a transformation in terms of digitalization across the developing and developed regions, which is accelerating the adoption of software such as an electronic trial master file.
- For instance, as per the 2022 data published by Clinicaltrials.gov, there are around 423,514 clinical trials listed with locations in all 50 States and 221 countries to date. With the increasing adoption of information technology in the healthcare industry, ETMF use is increasing. There are several benefits of using electronic trial master file, as it just needs to be installed once and reused multiple times. Additionally, as the clinical trial progresses and expands, the burden of huge data is easily managed with the electronic trial master file application.
- Initiatives by key organizations are expected to provide a favorable environment for the growth of such technologies. For instance, on March 15, 2023, the Food and Drug Administration (FDA or the Agency) issued a draft guidance entitled Electronic Systems, Electronic Records, and Electronic Signatures in Clinical Investigations: Questions and Answers (2023 Draft Guidance). FDA has grouped these questions and answers into five sections: (A) electronic records, (B) electronic systems owned or controlled by sponsors or other regulated entities, (C) information technology service providers and services, (D) digital health technologies, and (E) electronic signatures.
- FDA continues to take a narrow and practical interpretation of Part 11, but it has augmented its risk-based approach to validation through additional considerations and recommendations. Thus, such guidelines are expected to provide better support for such platforms, which is expected to increase market growth over the forecast period. However, budget constraints are expected to hinder market growth.
- Therefore, due to the growing adoption of electronic trial master files (ETMF) owing to the increasing number of clinical trials globally and the increasing number of research and development activities, the demand for electronic trial master files (ETMF) is projected to increase over the forecast period. However, budget constraints of healthcare providers and companies are expected to restrain the growth of the market during the forecast period.
Global Electronic Trial Master File (ETMF) Market Trends and Insights
Pharmaceutical & Biotechnology Companies Segment is Expected to Exhibit a Significant Growth
- Pharmaceutical & biotechnology companies are expected to show significant growth in the market, primarily due to the increasing investments in research and development activities. As per the 2021 report from the India Brand Equity Foundation (IBEF), around USD 1.206 billion was invested in the pharmaceutical industry during the year 2021. Such huge investments are propelling the number of clinical trials being performed, which is acting as a fueling factor for the market studied.
- Furthermore, the number of trials is increasing, which requires the ETMF system for the management of the data concerning the patients and drugs. According to the European Medicine Agency, In 2021, there were 37,293 clinical trials registered in Europe. Hence, these factors are expected to boost the demand for ETMF systems in pharmaceutical and biopharmaceutical companies.
- Additionally, as per the report published by Biogen in July 2022, Eisai and Biogen developed Alzheimer's Disease drugs and are asking for accelerated approval based on their ability to clear toxic plaques of a protein called amyloid. Similarly, there are several clinical trials planned in the years 2022-2023 by various pharmaceutical and biotechnology companies, which is a fueling factor for the market studied.
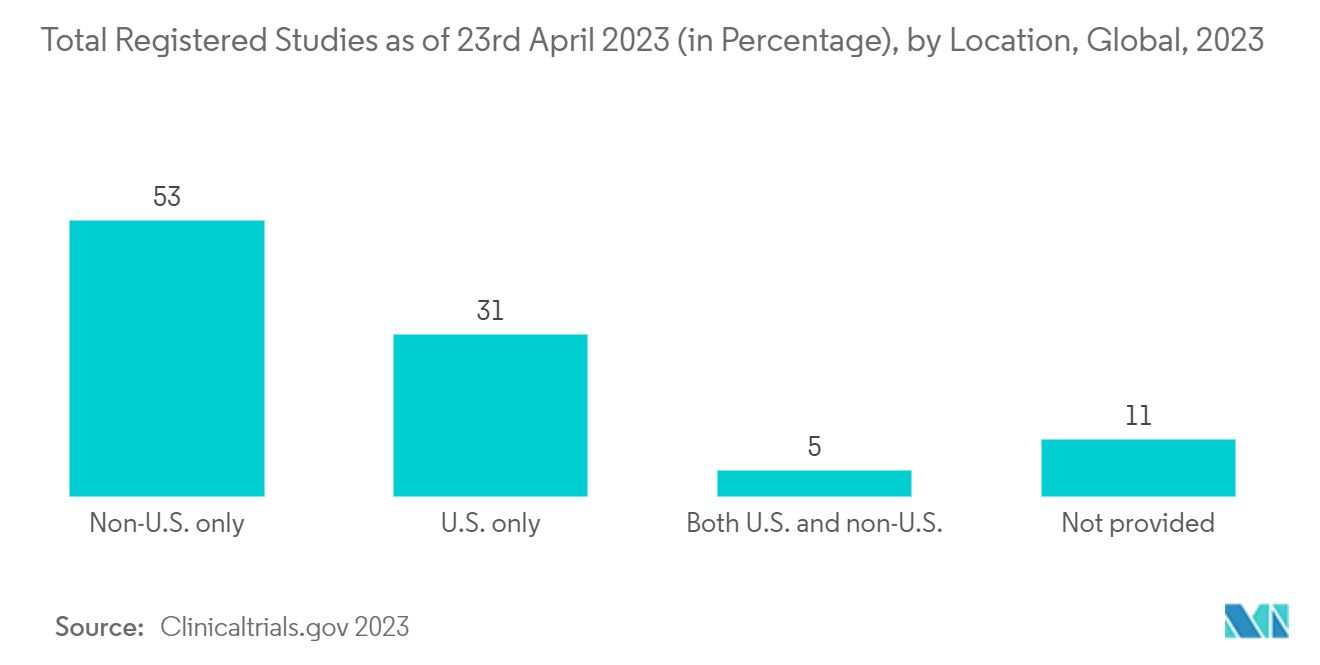
North America is Expected to Hold a Significant Share in the Market and Expected to do Same in the Forecast Period
- North America is expected to be a dominant region in the Electronic Trial Master File (ETMF) market owing to the increasing investments and several studies related to research and development.
- According to the Pharmaceutical Researchers and Manufacturers of America (PhRMA), the United States had the highest share of around 58% in terms of R&D expenditure during the year 2021. In addition, the R&D expenditure by biopharmaceutical companies was around 102.3 billion USD in the United States, as per the same source.
- Furthermore, as of 23rd April 2023, around 139,632 studies are registered in the United States, which will grow by the end of April 2023. The United States alone accounts for 31% of the registered study locations. This indicates that there is a huge number of clinical trials taking place in the country, which is beneficial for the market studied as there will be a need for a suitable tool that can help researchers keep track of data and store the data effectively.
- Additionally, the adoption of healthcare information technology is higher in the United States, which is augmenting the demand for software that can ease the work of huge data generated through clinical trials every year. Hence, it is believed that the demand for ETMF software in the country will boost as the number of trials increases in the United States.
- Therefore, owing to the above-mentioned factors, it is believed that the market studied will hold a strong share.
Competitive Landscape
The market for electronic trial master files (ETMF) is moderately competitive, and several companies that are well-known in the market are Veeva Systems, Aris Global LLC, Master Control, Inc., Clinevo Technology, Oracle Corporation, Phlexglobal, TransPerfect, Aurea Software, Labcorp (Covance, Inc.) and Sureclinical, among others.
Electronic Trial Master File (ETMF) Industry Leaders
Oracle Corporation
Clinevo Technologies
Master Control Inc
Veeva Systems
Aris Global LLC
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- In February 2023, Vial, a clinical research organization (CRO), signed a partnership agreement with Egnyte for the integration of the Life Sciences eTMF (electronic trial master file) solution into Vial's platform. As per the company, Egnyte eTMF is a 21 CFR Part 11 compliant platform that lowers the processing time for documents and data, maintains compliance, and enhances audit readiness for life sciences companies.
- In September 2022, Montrium launched its expert-led trial master file (TMF) services and its new TMF maturity educational training. These innovative offerings are designed to support clinical operations and TMF teams at every stage of the clinical development journey.
Global Electronic Trial Master File (ETMF) Market Report Scope
The electronic trial master file is a managing system that provides a structured way of organizing, storing, and managing documents, images, and other contents related to clinical trials.
The electronic trial master file market is segmented by product type (software, services), end-user (pharmaceutical & biotechnology companies, contract research organizations, other end-users), and geography (North America, Europe, Asia-Pacific, and the Rest of the World).
The report offers the value (in USD) for the above segments.
| Services |
| Software |
| Pharmaceutical & Biotechnology Companies |
| Contract Research Organizations |
| Other End-Users |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Rest of the World |
| By Product Type | Services | |
| Software | ||
| By End-User | Pharmaceutical & Biotechnology Companies | |
| Contract Research Organizations | ||
| Other End-Users | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Rest of the World | ||
Key Questions Answered in the Report
How big is the Electronic Trial Master File Market?
The Electronic Trial Master File Market size is expected to reach USD 1.92 billion in 2025 and grow at a CAGR of 12.55% to reach USD 3.48 billion by 2030.
What is the current Electronic Trial Master File Market size?
In 2025, the Electronic Trial Master File Market size is expected to reach USD 1.92 billion.
Who are the key players in Electronic Trial Master File Market?
Oracle Corporation, Clinevo Technologies, Master Control Inc, Veeva Systems and Aris Global LLC are the major companies operating in the Electronic Trial Master File Market.
Which is the fastest growing region in Electronic Trial Master File Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Electronic Trial Master File Market?
In 2025, the North America accounts for the largest market share in Electronic Trial Master File Market.
What years does this Electronic Trial Master File Market cover, and what was the market size in 2024?
In 2024, the Electronic Trial Master File Market size was estimated at USD 1.68 billion. The report covers the Electronic Trial Master File Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Electronic Trial Master File Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Electronic Trial Master File (ETMF) Market Report
Statistics for the 2025 Electronic Trial Master File (ETMF) market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Electronic Trial Master File (ETMF) analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.




