Artificial Heart Market Size
Artificial Heart Market Analysis
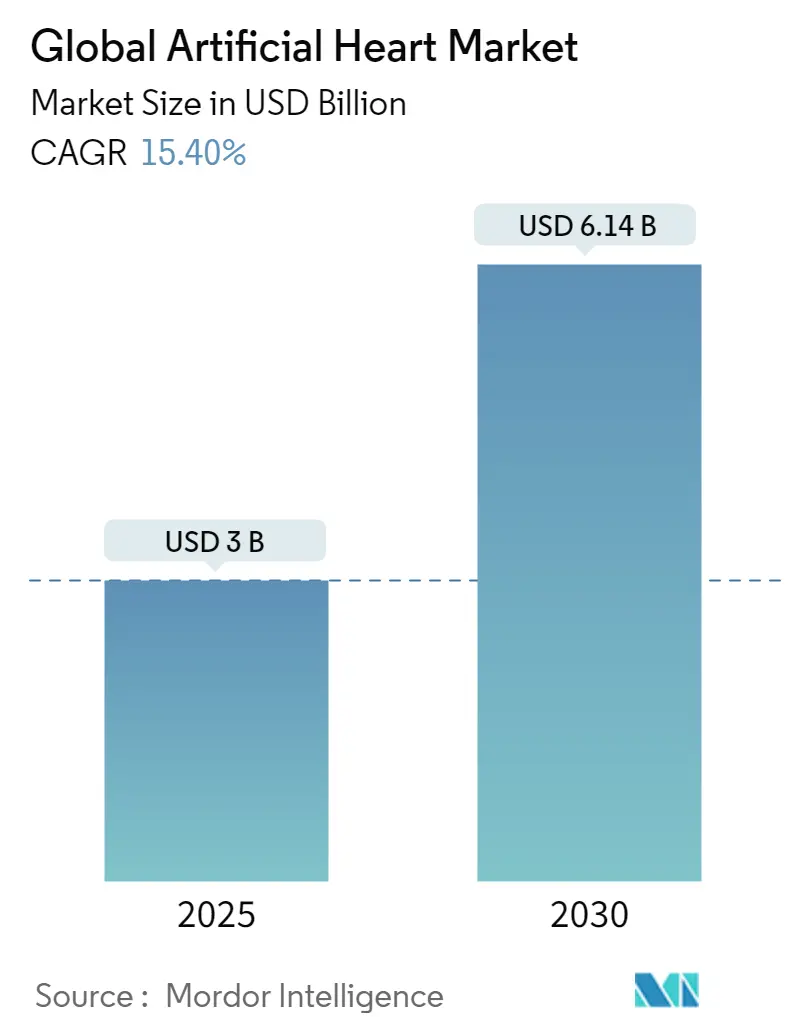
The Global Artificial Heart Market size is estimated at USD 3.00 billion in 2025, and is expected to reach USD 6.14 billion by 2030, at a CAGR of 15.4% during the forecast period (2025-2030).
The growing burden of cardiovascular diseases and technological advancements are the major factors driving the growth of the artificial heart market. The growth of the geriatric population and sedentary lifestyles are increasing the risk of cardiovascular diseases. Rising research and development activities are also expected to boost the market.
- According to the factsheet published by the British Heart Foundation (BHF) in January 2024, approximately 7.6 million people in the United Kingdom were suffering from heart disease. Approximately 100,000 patients suffering from heart disease are admitted to hospitals in the United Kingdom every year.
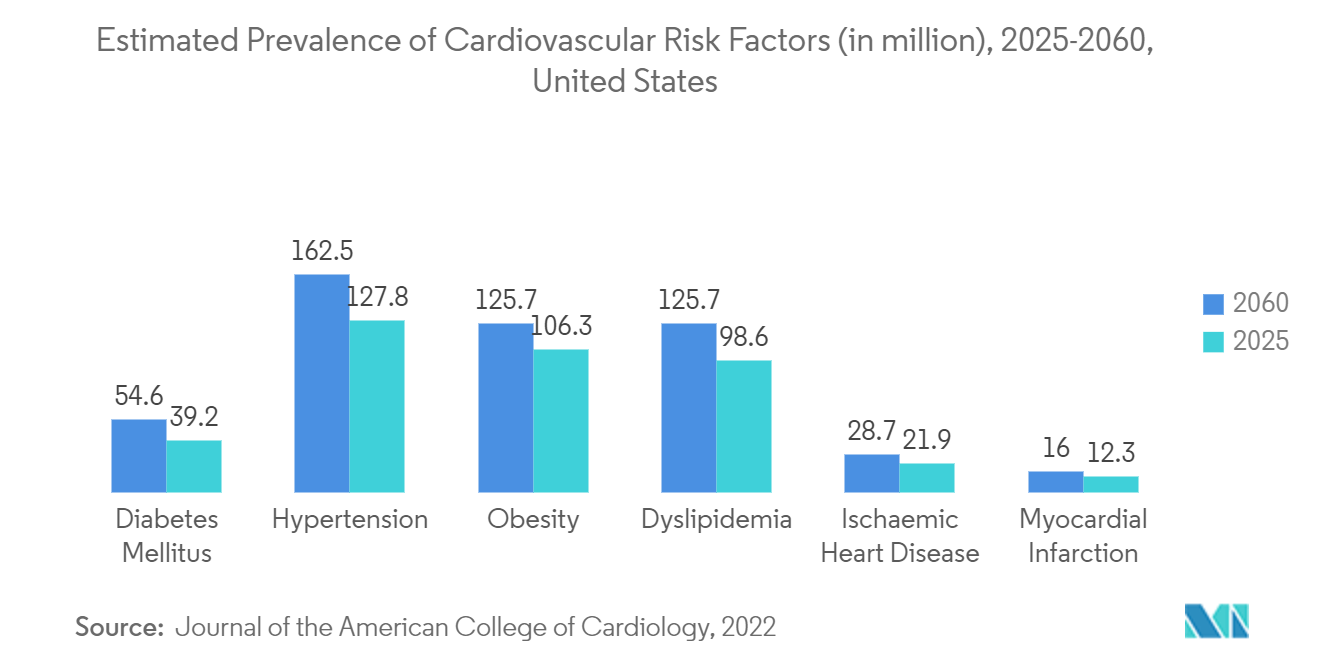
- Around 620 million people are currently living with heart and circulatory diseases worldwide. The prevalence of heart disease is highest in Asia, followed by Europe and the Middle East, Turkey, and Africa.
- Due to the rising burden of cardiovascular diseases, there is an increasing demand for innovative technologies that can decrease the mortality rates associated with heart failure, thereby increasing the demand for artificial hearts.
- All regions and areas worldwide are experiencing population aging, and it is projected to continue to do so over the next several decades. For instance, according to the United Nations World Population Ageing 2023, developed countries are expected to move to a more advanced stage of population aging, with the proportion of older persons rising from 20% in 2023 to 28% in 2050. For other developing countries, this proportion also rises from 9 to 17%, while for least developed countries, it is expected to rise from 4% to over 6% by 2050. Such an increase in the aging population poses a high risk for cardiovascular diseases and thus drives the growth of the market.
- Therefore, owing to the rising prevalence of heart diseases and the surging number of research studies, the market is expected to grow significantly over the coming years. However, the high cost of artificial hearts and related surgeries may hinder the market's growth during the forecast period.
Artificial Heart Market Trends
Ventricular Assist Devices are Expected to Account for a Large Share
- A ventricular assist device (VAD) helps pump blood from the lower chambers of the heart (ventricles) to the rest of the body. VAD is essential for patients with weakened hearts or heart failures. They temporarily maintain the heart function until the heart recovers or before the heart transplant. In some chronic heart conditions, they are used as permanent support therapy, as they are mechanical pumps surgically implanted in the heart to pump blood into the whole body.
- Factors such as increasing product launches, rising research and development activities, and strategies adopted by key market players will likely drive the growth of the market. For instance,
- In July 2022, China's National Medical Products Administration issued marketing approval for a domestically developed artificial heart for severe heart failure. It is an implantable ferrofluids-driven left ventricular assist device.
- In March 2022, Penn State Health Milton S. Hershey Medical Center became the second hospital in the United States to implant a newly designed EvaHeart2 Left Ventricular Assist System (LVAS) in a patient with severe heart failure. The procedure was part of the COMPETENCE Trial, a multi-center clinical study to evaluate EvaHeart2, including 40 clinical sites and 399 patients with severe heart failure through 2022.
- Furthermore, the prominent players are actively participating in the development of advanced VAD, which is projected to drive the growth of the segment during the forecast period.
- For instance, in June 2023, Magenta Medical commenced a clinical trial study for its smallest heart pump, known as elevated percutaneous Left Ventricular Assist Device (VAD), for high-risk percutaneous coronary intervention (HR-PCI). The aim of the study is to reduce vascular access complications during heart surgery.
- Thus, the studied segment is expected to contribute to significant growth due to the increasing product launches, rising research and development activities, and strategies adopted by key market players.
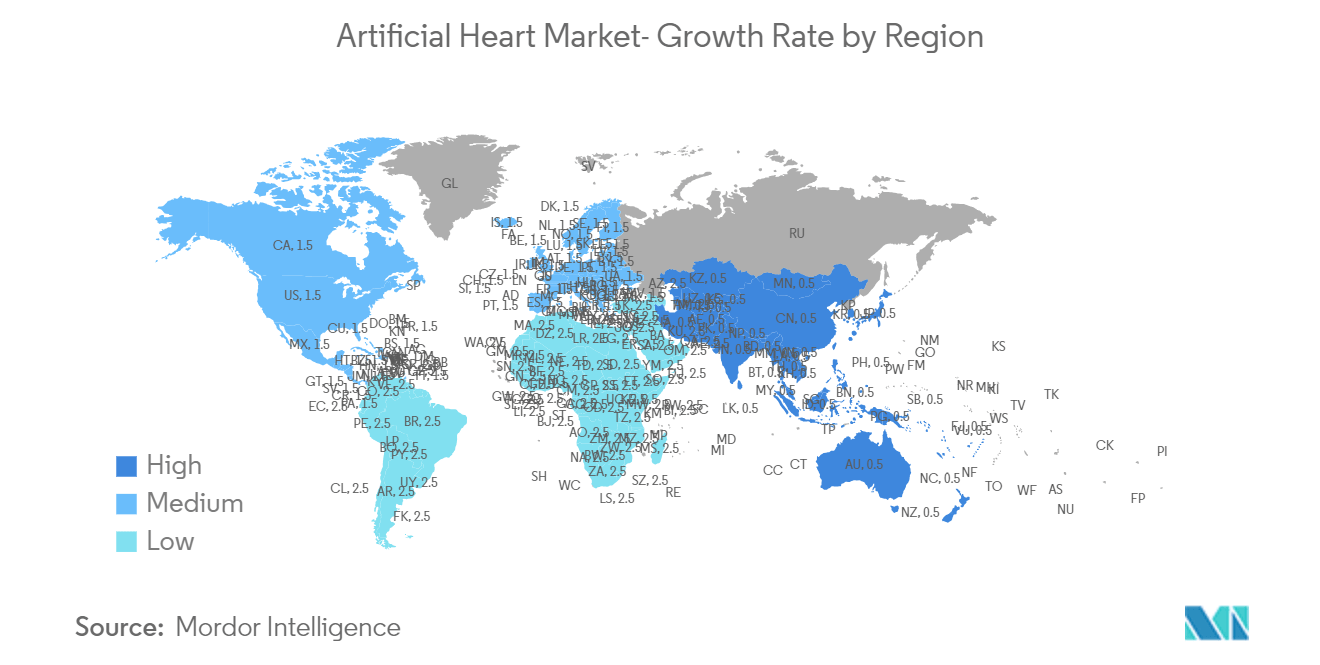
North America is Expected to Hold a Significant Share in the Market
- The geographical analysis of the artificial heart market shows that North America holds a significant market share in the global market. The primary driving factors for the growth of the North American artificial heart market are the growing burden of cardiovascular diseases, rising adoption of technologies, increasing product approvals, increasing investments, and key initiatives taken by the key market players.
- The rising burden of cardiovascular diseases leading to heart failure is expected to boost the advancements in the artificial heart market in the region. For instance, according to the article published by the CDC in May 2023, about 659,000 Americans have a heart attack every year. Hence, due to the high prevalence of heart attacks in the region, there is a continuous need for devices that can increase the efficiency of hearts.
- The increase in the launch of products and services in the region is also anticipated to drive the market significantly. For instance, in February 2022, Carmat received FDA approval for its new version of its artificial heart in the United States. This new device version includes improvements in the prosthesis and the wearable system based on clinical experience gained in the PIVOTAL study.
- The rising research and development activities for developing technologies supporting heart functioning are another major factor likely to drive growth. For instance, in February 2023, Researchers from Harvard University engineered a school of fish that uses the contractions of human heart cells to swim autonomously. This experiment could advance pacemaker technology and improve the development of artificial hearts for humans.
- Therefore, due to the high prevalence of heart diseases and the number of product launches by prominent players, the market studied is expected to witness lucrative growth in North America.
Artificial Heart Industry Overview
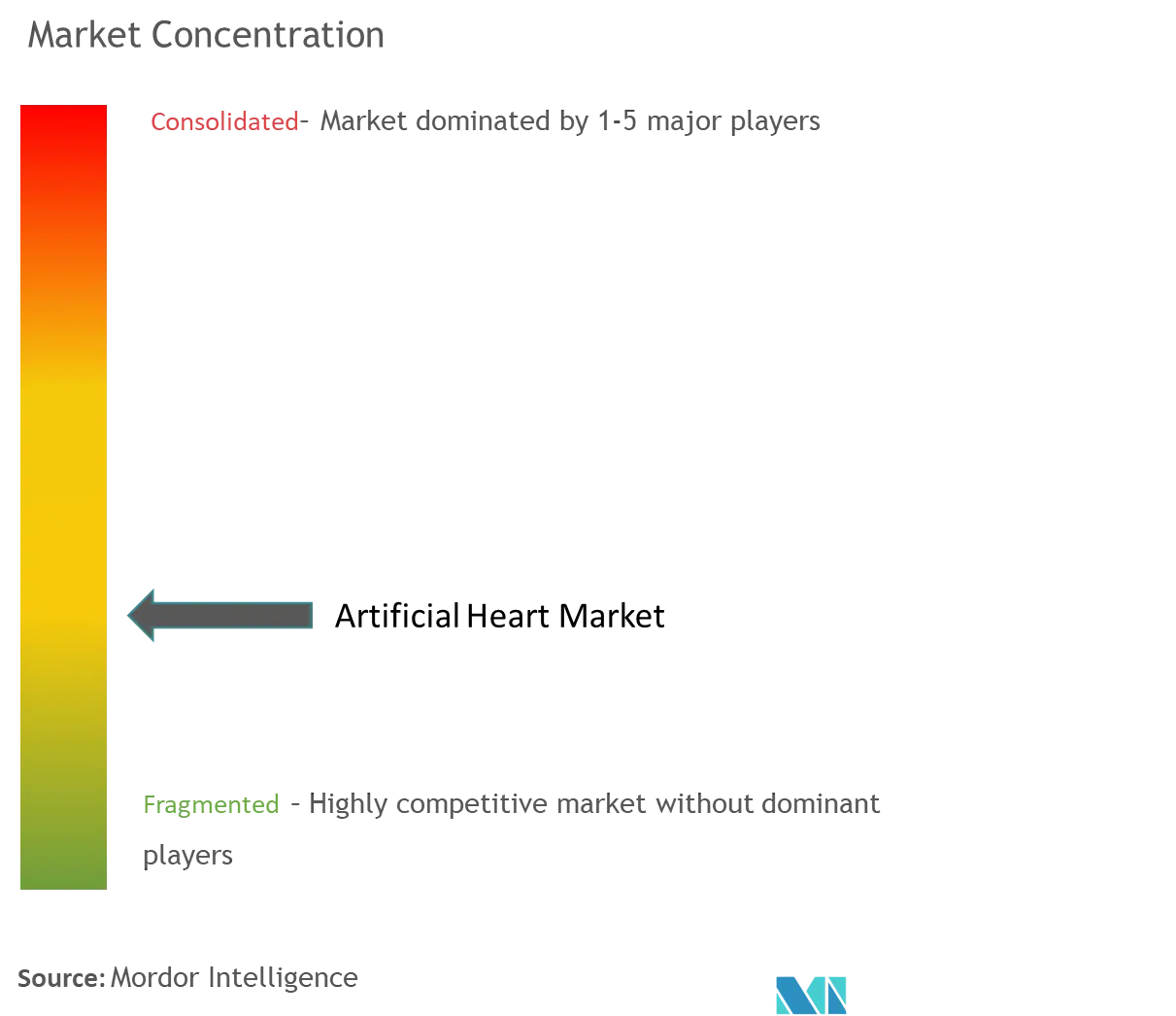
The global artificial heart market is semi-consolidated. The adoption of key strategies such as mergers, acquisitions, research activities, partnerships, and others is expected to boost the growth of the market. Major players in the market include SynCardia Systems LLC, CryoLife Inc., Abiomed, Carmat, and Abbott Laboratories, among others.
Global Artificial Heart Market Leaders
-
SynCardia Systems, LLC
-
CryoLife, Inc
-
Abiomed
-
Carmat
-
Abbott
- *Disclaimer: Major Players sorted in no particular order
Global Artificial Heart Market News
- March 2023: LivaNova PLC received US-FDA approval for its Essenz Heart-Lung Machine (HLM), including the Essenz Perfusion System, which enables a patient-tailored perfusion approach that improves both clinical workflows and the quality of patient care during cardiopulmonary bypass (CPB) procedures.
- January 2022: In India, the School of Medical Research and Technology (SMRT), IIT Kanpur, launched Hridyantra, a grand challenge-based program to develop an advanced artificial heart or a left ventricular assist device.
Artificial Heart Industry Segmentation
As per the scope of the report, an artificial heart is a device that maintains blood circulation and oxygenation in the human body for varying periods of time. Artificial hearts are typically used to bridge the time to heart transplantation or to replace the heart if a heart transplant is impossible.
The artificial heart market is segmented by type, power source, end user, and geography. By type, the market is segmented into ventricular assist devices, total artificial hearts, heart-lung machines, and others. Others include temporary assist devices and pulsatile flow pumps. The market is segmented by power source into internal and external batteries. By end user, the market is segmented into hospitals and clinics and specialty clinics. By geography, the market is segmented into North America, Europe, Asia-Pacific, Middle East and Africa, and South America. For each segment, the market size and forecasts are provided on the basis of value (in USD).
| By Type | Ventricular Assist Device | ||
| Total Artificial Heart | |||
| Heart-Lung Machine | |||
| Other Types | |||
| By Power Source | Internal Battery | ||
| External Battery | |||
| By End User | Hospitals and Clinics | ||
| Specialty Clinics | |||
| By Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| Japan | |||
| India | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||
| Ventricular Assist Device |
| Total Artificial Heart |
| Heart-Lung Machine |
| Other Types |
| Internal Battery |
| External Battery |
| Hospitals and Clinics |
| Specialty Clinics |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
Global Artificial Heart Market Research Faqs
How big is the Global Artificial Heart Market?
The Global Artificial Heart Market size is expected to reach USD 3.00 billion in 2025 and grow at a CAGR of 15.40% to reach USD 6.14 billion by 2030.
What is the current Global Artificial Heart Market size?
In 2025, the Global Artificial Heart Market size is expected to reach USD 3.00 billion.
Who are the key players in Global Artificial Heart Market?
SynCardia Systems, LLC, CryoLife, Inc, Abiomed, Carmat and Abbott are the major companies operating in the Global Artificial Heart Market.
Which is the fastest growing region in Global Artificial Heart Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Global Artificial Heart Market?
In 2025, the North America accounts for the largest market share in Global Artificial Heart Market.
What years does this Global Artificial Heart Market cover, and what was the market size in 2024?
In 2024, the Global Artificial Heart Market size was estimated at USD 2.54 billion. The report covers the Global Artificial Heart Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Global Artificial Heart Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on: May 20, 2024
Global Artificial Heart Industry Report
Statistics for the 2025 Global Artificial Heart market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Global Artificial Heart analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.