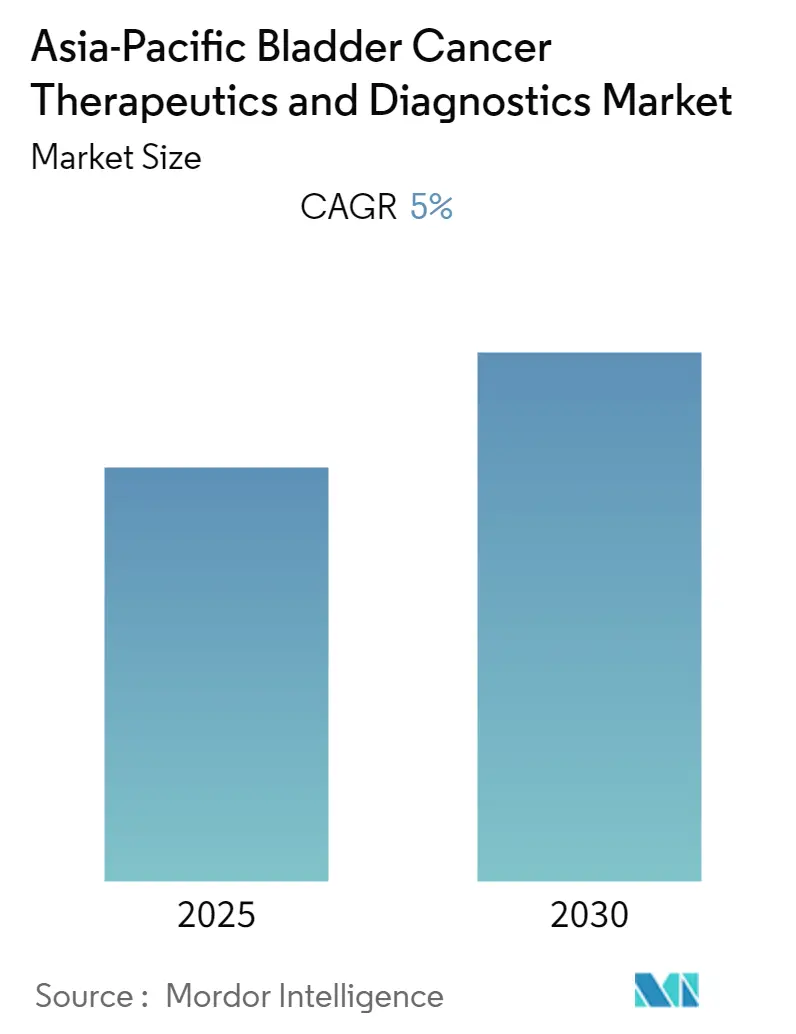
Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market Analysis by Mordor Intelligence
The Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market is expected to register a CAGR of 5% during the forecast period.
Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market Trends and Insights
Bladder Ultrasound is Expected to Show a Steady Growth During the Forecast Period
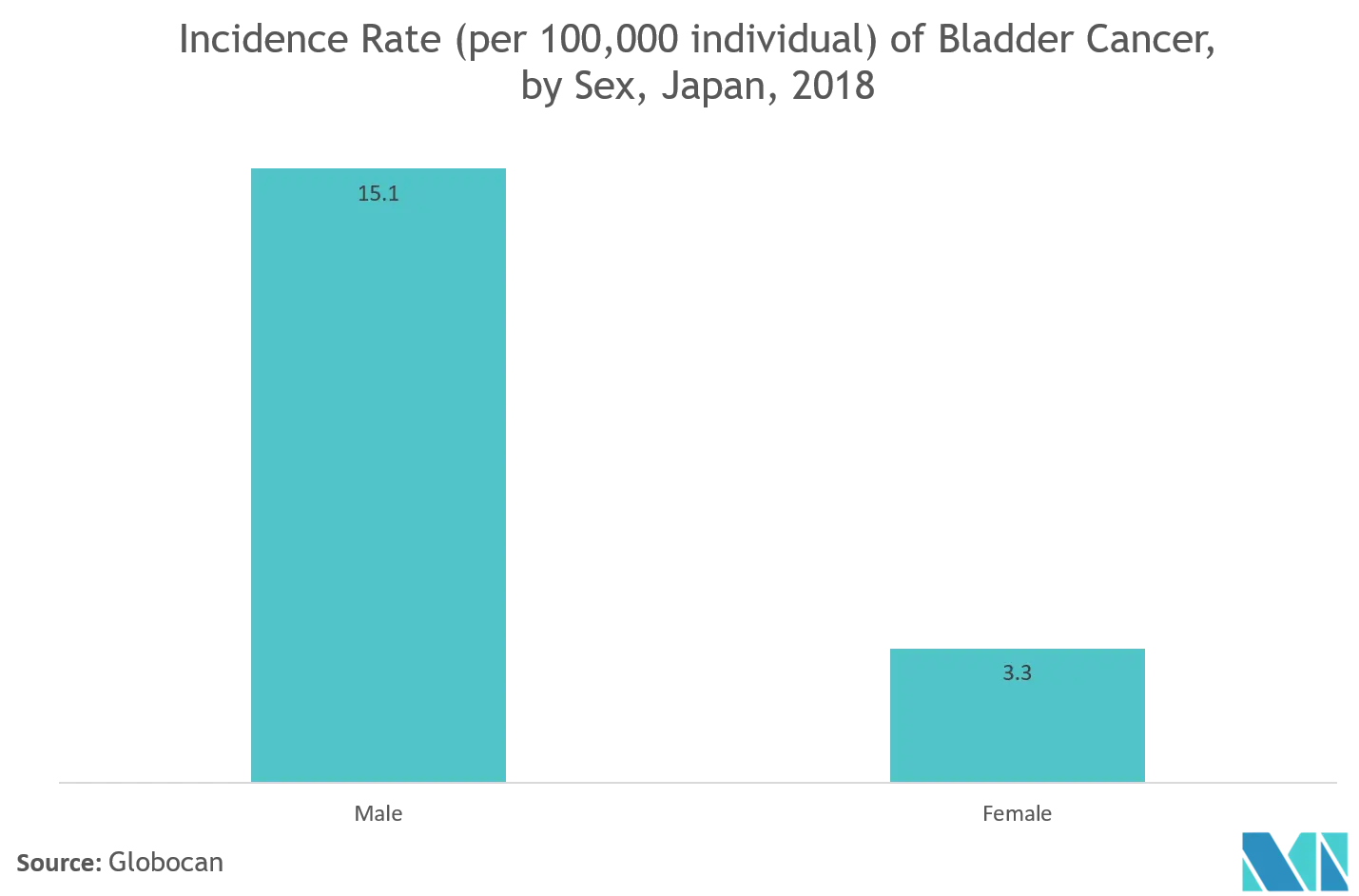
Bladder Ultrasound is one of the type of diagnostic device which uses sound waves to detect and analyze the cancer present in the bladder. The growing incidence of bladder cancer and new devices are being developed for the diagnosis of cancer are expected to stimulate the growth in this segment. According to the Globocan, 2018, almost 4,249 incidences of bladder cancer were observed in the Korea Republic of with around 1,712 deaths. Ultrasound is the most common method used in diagnosing the early stages of cancer due to its specificity. Hence, it is expected to boost the demand for ultrasound devices in the market during the forecast period.


Competitive Landscape
Some of the key players in the market are AstraZeneca Plc, Bristol-Myers Squibb Company, Eli Lilly & Company, F. Hoffmann-La Roche Ltd, GlaxoSmithKline, Novartis, Pfizer, Sanofi, Endo Pharmaceuticals Inc. and Johnson & Johnson. Companies are constantly indulged in developing the products related to diagnosis and treatment of the bladder cancer to grow their presence in the market. In 2018, Merck recieved an approval from Japanese Ministry of Health, Labor and Welfare (MHLW) for it's product Keytruda (MSD) which is used for the treatment of urothelial cancer patients.
Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Industry Leaders
F. Hoffmann-La Roche Ltd
Johnson & Johnson
Endo Pharmaceutical Inc.
AstraZeneca Plc
Bristol-Myers Squibb Company
- *Disclaimer: Major Players sorted in no particular order




Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market Report Scope
Bladder cancer is a type of tumor that occurs in the cells of the bladder. It grows rapidly and shows uncontrolled growth in the urinary bladder lining with epithelial cells. These cancerous cells may even spread through the lining into the muscular wall of the bladder. The Asia-Pacific Bladder Cancer Therapeutics and Diagnostics market is segmented by Product, by Cancer Type, and Geography.
| By Therapeutics | Chemotherapy |
| Immunotherapy | |
| Others | |
| By Diagnostics | Cystoscopy |
| Bladder Ultrasound | |
| Urinalysis | |
| Others |
| Transitional Cell Bladder Cancer |
| Squamous Cell Bladder Cancer |
| Others |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific |
| By Product | By Therapeutics | Chemotherapy |
| Immunotherapy | ||
| Others | ||
| By Diagnostics | Cystoscopy | |
| Bladder Ultrasound | ||
| Urinalysis | ||
| Others | ||
| By Cancer Type | Transitional Cell Bladder Cancer | |
| Squamous Cell Bladder Cancer | ||
| Others | ||
| Geography | Asia-Pacific | China |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||


Key Questions Answered in the Report
What is the current Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market size?
The Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market is projected to register a CAGR of 5% during the forecast period (2025-2030)
Who are the key players in Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market?
F. Hoffmann-La Roche Ltd, Johnson & Johnson, Endo Pharmaceutical Inc., AstraZeneca Plc and Bristol-Myers Squibb Company are the major companies operating in the Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market.
What years does this Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market cover?
The report covers the Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Asia-Pacific Bladder Cancer Therapeutics and Diagnostics Market Report
Statistics for the 2025 Asia-Pacific Bladder Cancer Therapeutics and Diagnostics market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Asia-Pacific Bladder Cancer Therapeutics and Diagnostics analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





