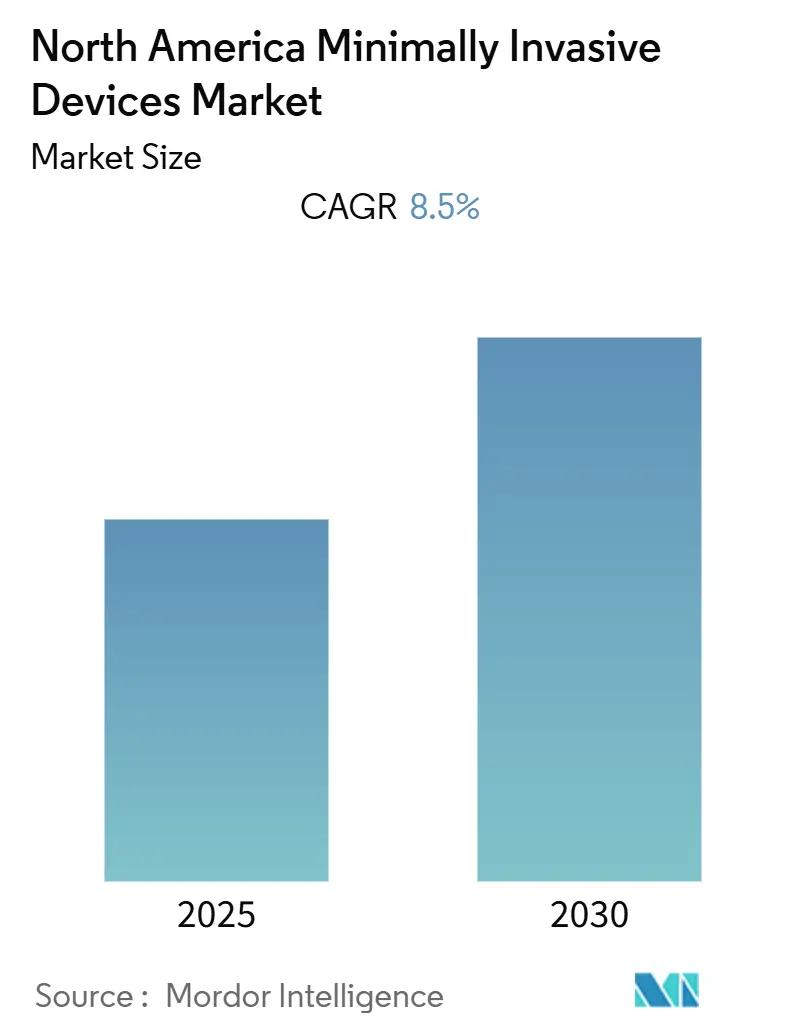
North America Minimally Invasive Devices Market Analysis by Mordor Intelligence
The North America Minimally Invasive Devices Market is expected to register a CAGR of 8.5% during the forecast period.
North America minimally invasive surgical devices market is being driven by the growing burden of chronic diseases leading to the surgical interventions and high adoption of minimally invasive devices.
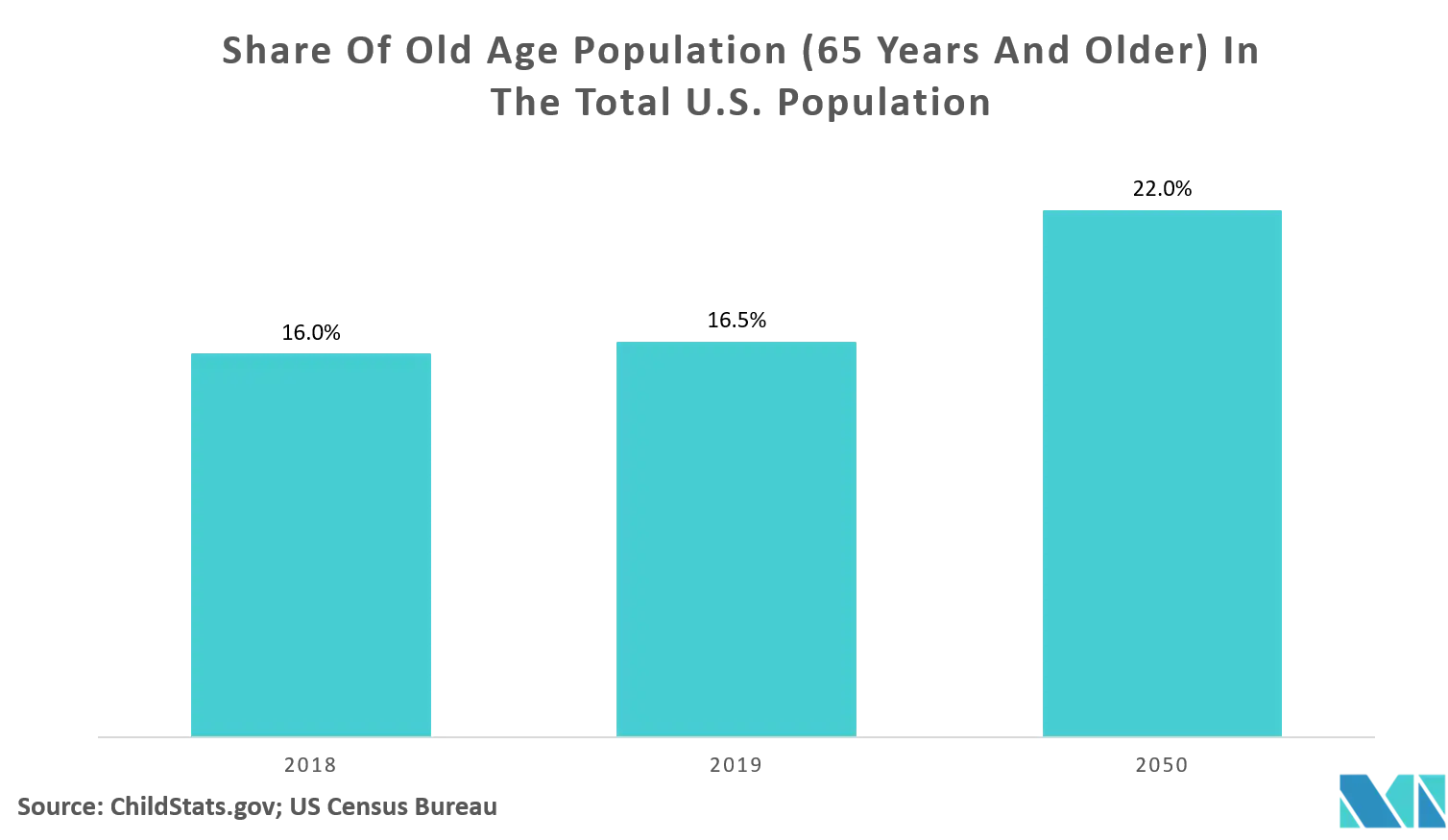
According to the data published by Centers for Disease Control and Prevention (CDC, 2018), there were 32.3 million of visits to physician offices, with diseases of the digestive system as the primary diagnosis in the United States. With the increase in the government's continuous effort to improve the health of people, the number of gastrointestinal surgeries is bound to rise, which, in turn, may improve the scope for minimally-invasive devices meant for gastrointestinal surgery. Similarly, about Nearly 18 million people in the United States underwent cosmetic surgical and minimally invasive procedures in 2018, states the ASPS. There has been a rise in the number of procedures in recent years, with over a quarter more million procedures performed in 2018 than 2017.
North America Minimally Invasive Devices Market Trends and Insights
Gastrointestinal Segment Expected to Have Notable Growth Over the Period.
The main factors driving the growth of the gastrointestinal segment is owing to the rising incidence of gastrointestinal diseases in the young, adult, and geriatric groups, in the United States. Dyspepsia, irritable-bowel syndrome and constipation are some of the examples of gastrointestinal disorders.
According to the International Foundation for Gastrointestinal Disorders, estimated that are approximately 2.4 to 3.5 million annual physician visits for irritable bowel syndrome (IBS) in the United States alone. The irritable bowel syndrome (IBS) is the most common disorder diagnosed by gastroenterologists, which accounts for up to 12% of total visits to primary care providers.
Moreover, with the increase in the government’s continuous effort to improve the health of people, the number of gastrointestinal surgeries is bound to rise, which, in turn, may improve the scope for minimally-invasive devices meant for gastrointestinal surgery.
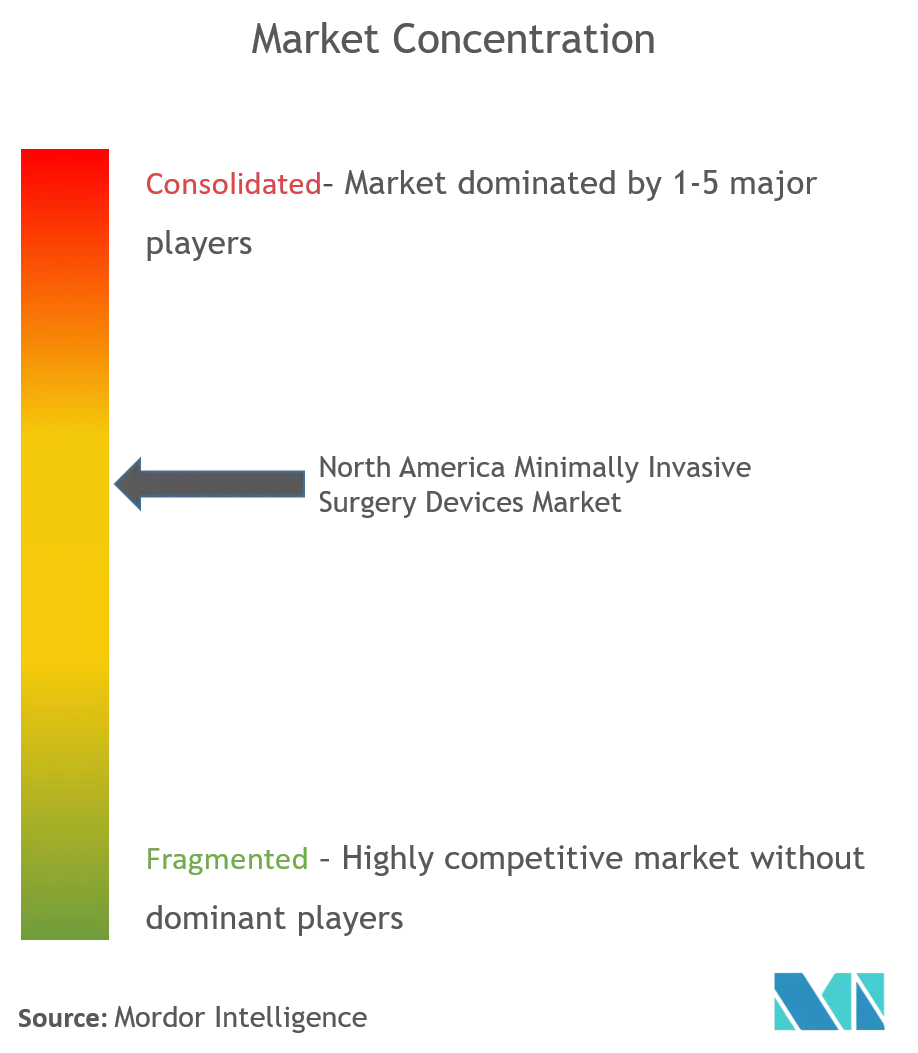
Competitive Landscape
The market studied is moderately consolidated, owing to the presence of a few key players in the market. The companies are focusing on the technological advancement of devices, in order to gain significant market share. Some of the market players are Abbott Laboratories, GE Healthcare, Intuitive Surgical Inc., Koninklijke Philips NV, Medtronic PLC, Olympus Corporation, Siemens Healthineers, Smith & Nephew, Stryker Corporation, and Zimmer Biomet.
North America Minimally Invasive Devices Industry Leaders
Medtronic Plc
Smith & Nephew
Zimmer Biomet
Abbott Laboratories
Intuitive Surgical Inc.
- *Disclaimer: Major Players sorted in no particular order


North America Minimally Invasive Devices Market Report Scope
As per the scope of this report, minimally-invasive surgical devices refer to electrosurgical systems, robot-assisted surgical systems, and surgical equipment, which are used during the minimally-invasive procedure. The minimally-invasive neurosurgery device market is growing at a significant rate, due to various benefits associated with minimally-invasive procedures, such as less recovery time, minimal pain to the patients, shortened hospital stay, and high accuracy.
| Handheld Instruments | |
| Guiding Devices | Guiding Catheters |
| Guidewires | |
| Electrosurgical Devices | |
| Endoscopic Devices | |
| Laproscopic Devices | |
| Monitoring and Visualization Devices | |
| Robotic Assisted Surgical Systems | |
| Ablation Devices | |
| Laser Based Devices | |
| Other MIS Devices |
| Aesthetic |
| Cardiovascular |
| Gastrointestinal |
| Gynecological |
| Orthopedic |
| Urological |
| Other Applications |
| North America | United States |
| Canada | |
| Mexico |
| By Products | Handheld Instruments | |
| Guiding Devices | Guiding Catheters | |
| Guidewires | ||
| Electrosurgical Devices | ||
| Endoscopic Devices | ||
| Laproscopic Devices | ||
| Monitoring and Visualization Devices | ||
| Robotic Assisted Surgical Systems | ||
| Ablation Devices | ||
| Laser Based Devices | ||
| Other MIS Devices | ||
| By Application | Aesthetic | |
| Cardiovascular | ||
| Gastrointestinal | ||
| Gynecological | ||
| Orthopedic | ||
| Urological | ||
| Other Applications | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||


Key Questions Answered in the Report
What is the current North America Minimally Invasive Devices Market size?
The North America Minimally Invasive Devices Market is projected to register a CAGR of 8.5% during the forecast period (2025-2030)
Who are the key players in North America Minimally Invasive Devices Market?
Medtronic Plc, Smith & Nephew, Zimmer Biomet, Abbott Laboratories and Intuitive Surgical Inc. are the major companies operating in the North America Minimally Invasive Surgery Devices Market.
What years does this North America Minimally Invasive Devices Market cover?
The report covers the North America Minimally Invasive Devices Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the North America Minimally Invasive Devices Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:








