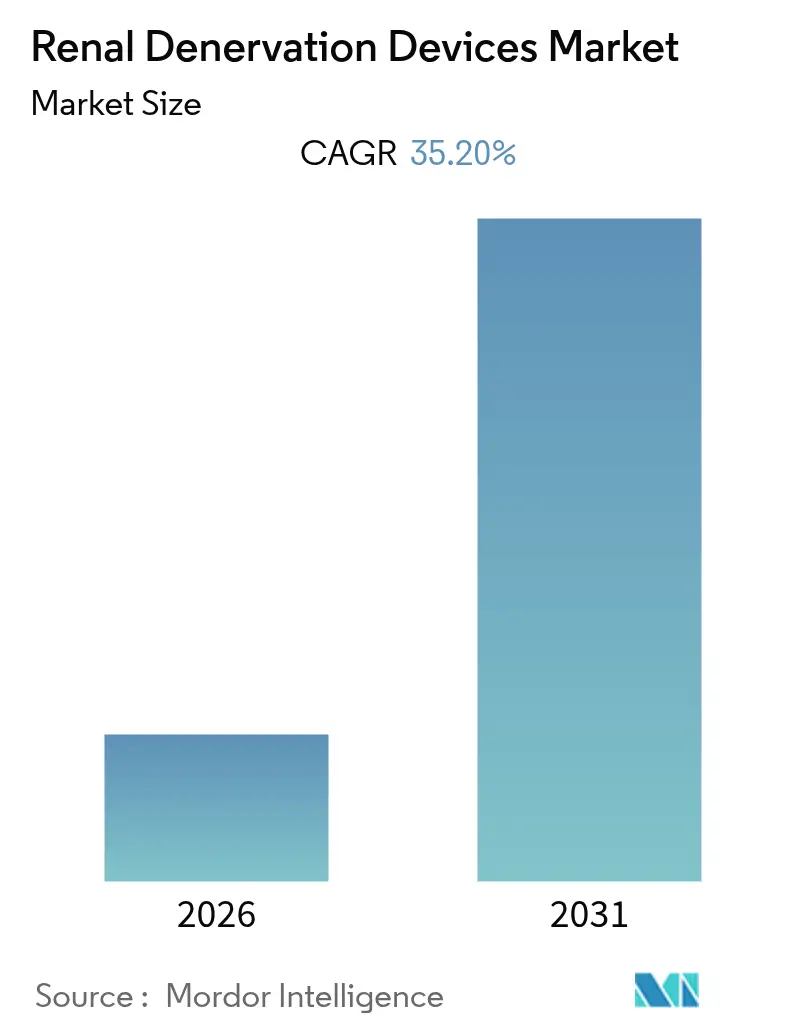
Renal Denervation Devices Market Analysis by Mordor Intelligence
The Renal Denervation Devices Market size is expected to register a CAGR of 35.20% during the forecast period.
The renal denervation devices industry is experiencing significant technological advancement and innovation in device development, particularly in the areas of microengineering, advanced robotics, microfabrication, microfluidics, and advanced materials. These developments are enabling manufacturers to create more precise and effective devices for treating hypertension through minimally invasive procedures. The integration of these technologies has led to improved patient outcomes and increased adoption of renal sympathetic denervation procedures. According to the World Health Organization's 2021 data, approximately 1.28 billion adults aged 30-79 years worldwide are affected by hypertension, highlighting the substantial market potential for these innovative devices.
The industry is witnessing a surge in breakthrough device designations and regulatory approvals, indicating growing confidence in renal denervation devices technology. Major medical device companies are actively pursuing clinical trials and research studies to validate the effectiveness of their devices. The field has seen particular advancement in ultrasound-based technologies, with multiple companies receiving breakthrough device designations from regulatory authorities. These developments are creating new opportunities for market expansion and improving treatment options for patients with resistant hypertension.
The market is experiencing a shift toward more sophisticated treatment approaches, particularly in addressing obesity-related hypertension. According to WHO statistics from 2021, over 39 million children under the age of 5 were overweight, indicating a growing potential patient pool as these individuals age. This demographic trend is driving innovation in device design and treatment protocols, as manufacturers work to develop solutions that can effectively address hypertension in various patient populations, including those with comorbid conditions.
Clinical evidence supporting the efficacy of renal sympathetic denervation procedures continues to strengthen, enhancing market credibility and adoption. A notable study published in the Nature journal in April 2022 revealed that more than 80% of Thai patients with resistant hypertension experienced successful outcomes from renal nerve denervation, with sustained results over extended follow-up periods and no intervention-related side effects. This growing body of evidence is crucial in establishing renal denervation devices as a mainstream treatment option and is encouraging healthcare providers to incorporate these devices into their treatment protocols for resistant hypertension.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Renal Denervation Devices Market Trends and Insights
Rising Prevalence of Hypertension
The burden of hypertension-induced diseases and chronic disorders continues to grow significantly worldwide, creating an urgent need for effective treatment options. According to the World Health Organization's June 2022 update, at least one in four adults in India has hypertension, with only about 12% having their blood pressure under control. This concerning trend is further evidenced by recent European statistics, where a 2021 Italian survey revealed that 42.3% of participants reported being diagnosed with hypertension, with prevalence being notably higher among men compared to women. The increasing prevalence can be attributed to various factors, including high sodium intake, low potassium consumption, rising obesity rates, increased alcohol consumption in developing nations, physical inactivity, and poor dietary habits.
The aging global population represents another significant factor contributing to hypertension's rising prevalence. As people age, they experience arterial stiffness, hemodynamic changes, neurohormonal and autonomic dysregulation, and declining renal function, all of which contribute to hypertension development. This demographic shift, combined with increasingly sedentary lifestyles and dietary changes, has led to a substantial increase in hypertension cases worldwide. Healthcare systems are particularly challenged by the fact that many individuals remain unaware of their condition, as demonstrated by recent studies showing that a significant portion of hypertensive patients are either undiagnosed or inadequately treated, highlighting the critical need for more effective treatment approaches.
Long Lasting Effect of the Procedure
Renal sympathetic denervation procedures have demonstrated remarkable long-term positive impacts on blood pressure control, particularly in patients who have undergone the procedure and those who had suffered from hemorrhagic stroke. Clinical studies have consistently shown that renal sympathetic denervation can provide sustained blood pressure reduction benefits, with follow-up data indicating maintained efficacy over extended periods. The procedure's ability to normalize blood pressure may contribute to better outcomes in various clinical situations, including cases of intracranial bleeding, making it an increasingly attractive treatment option for healthcare providers and patients alike.
The procedure's enduring effectiveness is particularly noteworthy in the context of treatment resistance and patient compliance. Research has demonstrated that systolic blood pressure reduction in patients undergoing the procedure is sustained for over three years, including significant reductions in both office and 24-hour ambulatory systolic blood pressure measurements. This long-term efficacy, combined with a favorable safety profile, represents a significant advantage over traditional medication-based approaches, which often require continuous adjustment and may face challenges with patient adherence. The procedure's ability to provide sustained results without requiring daily medication management makes it a particularly valuable option for patients who struggle with medication compliance or experience adverse effects from traditional antihypertensive medications. Additionally, the use of renal denervation devices and renal denervation systems in these procedures further enhances their effectiveness and appeal within the field of interventional cardiology devices.
Segment Analysis: Technology
Radiofrequency-Based Segment in Renal Denervation Devices Market
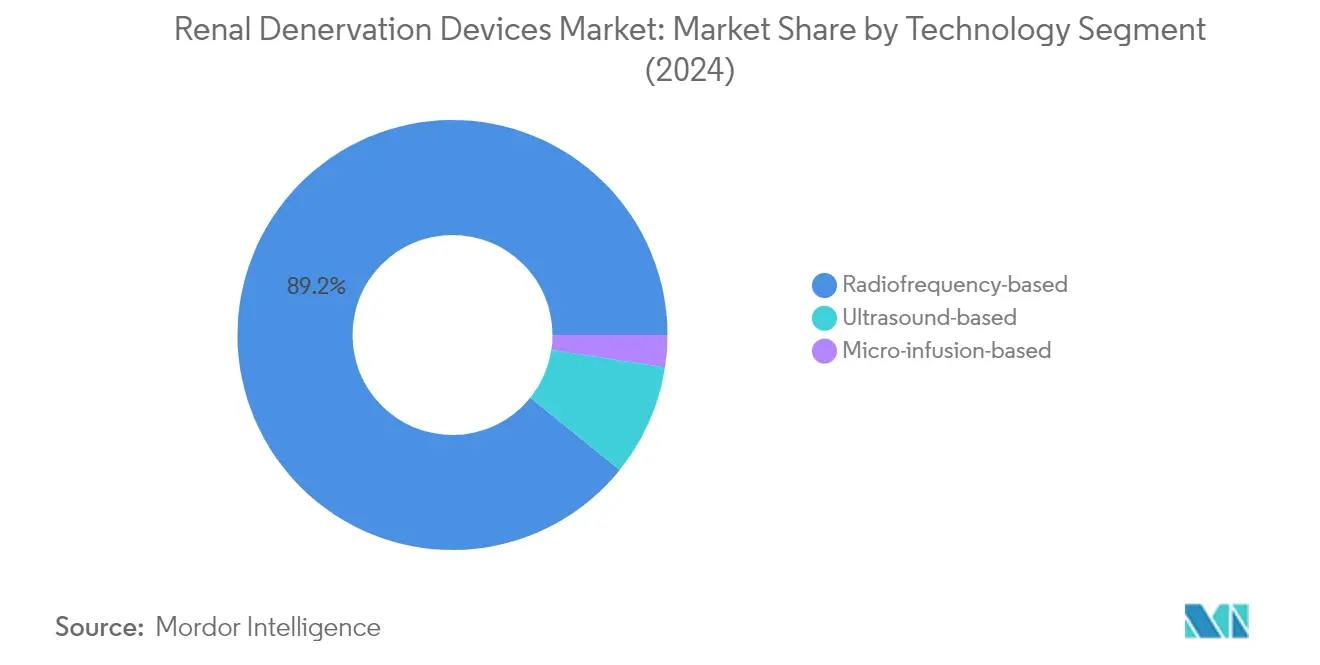
The radiofrequency ablation devices market size segment dominates the global renal denervation devices market, accounting for approximately 88.40% of the market share in 2025. This significant market position can be attributed to the segment's proven efficacy in treating resistant hypertension through radiofrequency ablation of nerves in the renal arteries. The technology's ability to provide precise and controlled energy delivery during the denervation procedure has made it the preferred choice among healthcare providers. Recent clinical trials have demonstrated that radiofrequency-based renal denervation devices effectively reduce blood pressure in patients with uncontrolled hypertension, both with and without concurrent antihypertensive medication. The segment's leadership is further strengthened by continuous technological advancements in catheter design and energy delivery systems, enhancing procedural safety and effectiveness.
Ultrasound-Based Segment in Renal Denervation Devices Market
The ultrasound-based segment is emerging as the fastest-growing technology in the renal denervation devices market, with projections indicating robust growth from 2025 to 2031. This remarkable growth trajectory is driven by the technology's unique ability to deliver precise, circumferential ablation patterns while minimizing procedural complications. The non-focused ultrasound energy offers deeper penetration into the arterial wall, potentially leading to more complete denervation. Recent breakthrough device designations from regulatory authorities have further accelerated the adoption of ultrasound-based systems. The technology's growing acceptance among healthcare providers is supported by promising clinical outcomes demonstrating consistent blood pressure reductions in resistant hypertension patients.
Remaining Segments in Technology
The micro-infusion-based segment represents an innovative approach in the renal denervation system, offering a unique mechanism of action through the delivery of neurolytic agents directly to the perivascular space. This technology provides an alternative to energy-based ablation methods, particularly beneficial for patients who may not be suitable candidates for radiofrequency or ultrasound-based procedures. The segment's development continues to evolve with improvements in catheter design and delivery systems, focusing on enhanced precision and safety profiles. The technology's ability to provide targeted drug delivery while maintaining the structural integrity of the arterial wall positions it as a promising option in the expanding renal denervation system landscape.
Renal Denervation Devices Market Geography Segment Analysis
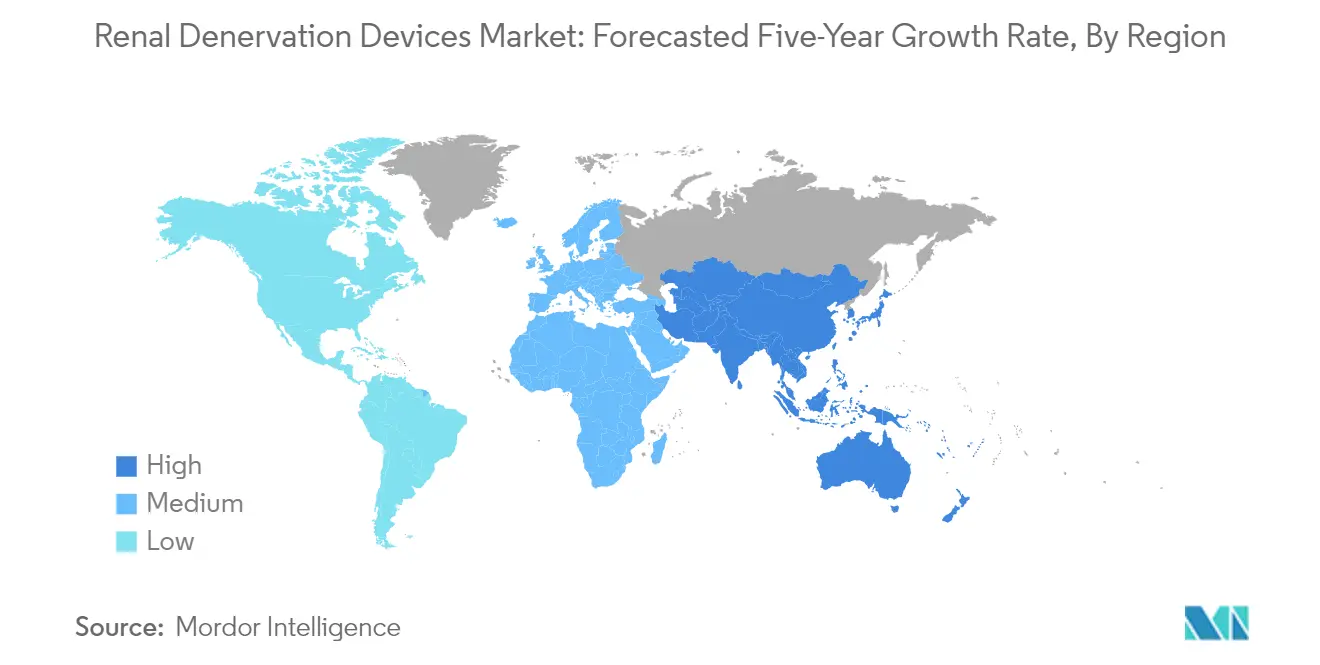
The North American renal denervation devices market demonstrates significant potential, driven by advanced healthcare infrastructure and the increasing prevalence of hypertension across the United States, Canada, and Mexico. The region benefits from the early adoption of innovative medical technologies and the presence of major market players. Healthcare providers in these countries are increasingly recognizing renal denervation devices as an effective treatment option for resistant hypertension. The regulatory environment, particularly in the United States and Canada, continues to evolve favorably for renal denervation technologies, while Mexico shows promising adoption rates in specialized cardiac care centers.
Canada emerges as the dominant force in the North American renal denervation devices market, holding approximately 61.30% market share in the region. The country's healthcare system provides comprehensive coverage for innovative medical procedures, contributing to higher adoption rates of renal denervation technologies. Canadian healthcare institutions have demonstrated a strong commitment to treating resistant hypertension through advanced interventional procedures. The country's robust clinical research environment and well-established healthcare infrastructure support the widespread implementation of renal denervation procedures. Additionally, increasing awareness among healthcare providers and patients about the benefits of renal denervation has contributed to market growth.
Canada continues to demonstrate exceptional growth potential in the renal denervation devices market, with an expected CAGR of approximately 38.90% from 2025 to 2031. The country's healthcare system actively promotes innovative treatment options for resistant hypertension, supporting the adoption of renal denervation procedures. Canadian medical institutions are increasingly incorporating these devices into their treatment protocols, backed by positive clinical outcomes. The growing elderly population and rising prevalence of hypertension further drive market expansion. Additionally, strong collaboration between healthcare providers and device manufacturers facilitates market development and technology adoption.
The European renal denervation devices market showcases robust development across Germany, the United Kingdom, France, Italy, and Spain. The region benefits from well-established healthcare systems and favorable reimbursement policies supporting innovative medical procedures. European medical institutions demonstrate strong acceptance of renal denervation technologies, supported by extensive clinical research and development activities. The presence of major market players and ongoing technological advancements further strengthens the market landscape. Collaborative efforts between healthcare providers and medical device manufacturers contribute to market expansion across the region.
Germany maintains its position as the largest market for renal denervation devices in Europe, commanding approximately 16.65% of the regional market share. The country's advanced healthcare infrastructure and strong focus on innovative medical technologies drive market growth. German medical institutions demonstrate high adoption rates of renal denervation procedures, supported by comprehensive healthcare coverage. The country's robust research and development activities in cardiovascular interventions contribute to market expansion. Additionally, the increasing prevalence of resistant hypertension and growing awareness among healthcare providers fuel market development.
Spain exhibits the highest growth potential in the European renal denervation devices market, with an anticipated CAGR of approximately 36.70% from 2025 to 2031. The Spanish healthcare system's increasing focus on minimally invasive procedures for treating resistant hypertension drives market expansion. Medical institutions across Spain are rapidly adopting renal denervation technologies, supported by positive clinical outcomes. The country's growing elderly population and rising incidence of cardiovascular diseases contribute to market growth. Additionally, increasing awareness among healthcare providers and patients about the benefits of renal denervation procedures supports market development.
The Asia-Pacific renal denervation devices market demonstrates significant growth potential across Japan, China, India, Australia, and South Korea. The region experiences increasing adoption of advanced medical technologies, supported by improving healthcare infrastructure and rising healthcare expenditure. Growing awareness about minimally invasive procedures and the increasing prevalence of hypertension drive market expansion. The presence of large patient populations and improving access to healthcare services create substantial opportunities for market growth. Regional governments' initiatives to enhance healthcare accessibility further support market development.
India emerges as the largest market for renal denervation devices in the Asia-Pacific region. The country's large patient population and increasing adoption of advanced medical technologies drive market growth. Indian healthcare institutions are rapidly incorporating renal denervation procedures into their treatment protocols. The country's improving healthcare infrastructure and growing focus on minimally invasive procedures support market expansion. Additionally, increasing awareness about hypertension management and rising healthcare expenditure contribute to market development.
India demonstrates exceptional growth potential in the renal denervation devices market. The country's healthcare system shows increasing acceptance of innovative treatment options for resistant hypertension. Medical institutions across India are rapidly adopting renal denervation technologies, supported by positive clinical outcomes. The growing prevalence of hypertension and increasing healthcare awareness drive market expansion. Additionally, government initiatives to improve healthcare accessibility and rising private sector investments support market growth.
The Middle East and African renal denervation devices market encompasses diverse healthcare landscapes across GCC countries and South Africa. The region demonstrates growing adoption of advanced medical technologies, particularly in major urban centers. Healthcare systems in GCC countries lead the market development, supported by substantial healthcare investments and modern medical infrastructure. South Africa emerges as a key market, driven by improving healthcare accessibility and increasing awareness about advanced treatment options. The region's growing focus on cardiovascular health and increasing healthcare expenditure support market expansion, with GCC countries representing the largest market and South Africa showing the fastest growth potential.
The South American renal denervation devices market, primarily driven by developments in Brazil and Argentina, shows promising growth potential. The region's healthcare systems demonstrate increasing acceptance of advanced medical technologies for treating resistant hypertension. Brazil leads the market development with its extensive healthcare infrastructure and large patient population, while Argentina shows the fastest growth potential. The region's growing focus on minimally invasive procedures and increasing healthcare expenditure support market expansion. Additionally, rising awareness about hypertension management and improving healthcare accessibility contribute to market development across South America.
Competitive Landscape
Top Companies in Renal Denervation Devices Market
The renal denervation devices market is characterized by intense innovation and strategic positioning among key players, including Medtronic, Abbott Laboratories, Boston Scientific, and Terumo Corporation. These companies are heavily investing in research and development to advance their technological capabilities, particularly in radiofrequency-based and ultrasound-based denervation systems. Market leaders are focusing on expanding their geographical presence through strategic partnerships and distribution agreements, especially in emerging markets across Asia-Pacific and Latin America. Companies are also prioritizing clinical trials and safety data collection to strengthen their regulatory submissions and gain approvals in major markets. The competitive landscape shows a clear trend toward developing integrated solutions that combine device efficacy with improved patient outcomes while simultaneously building comprehensive portfolios through strategic acquisitions and licensing agreements.
Consolidated Market with Strong Growth Potential
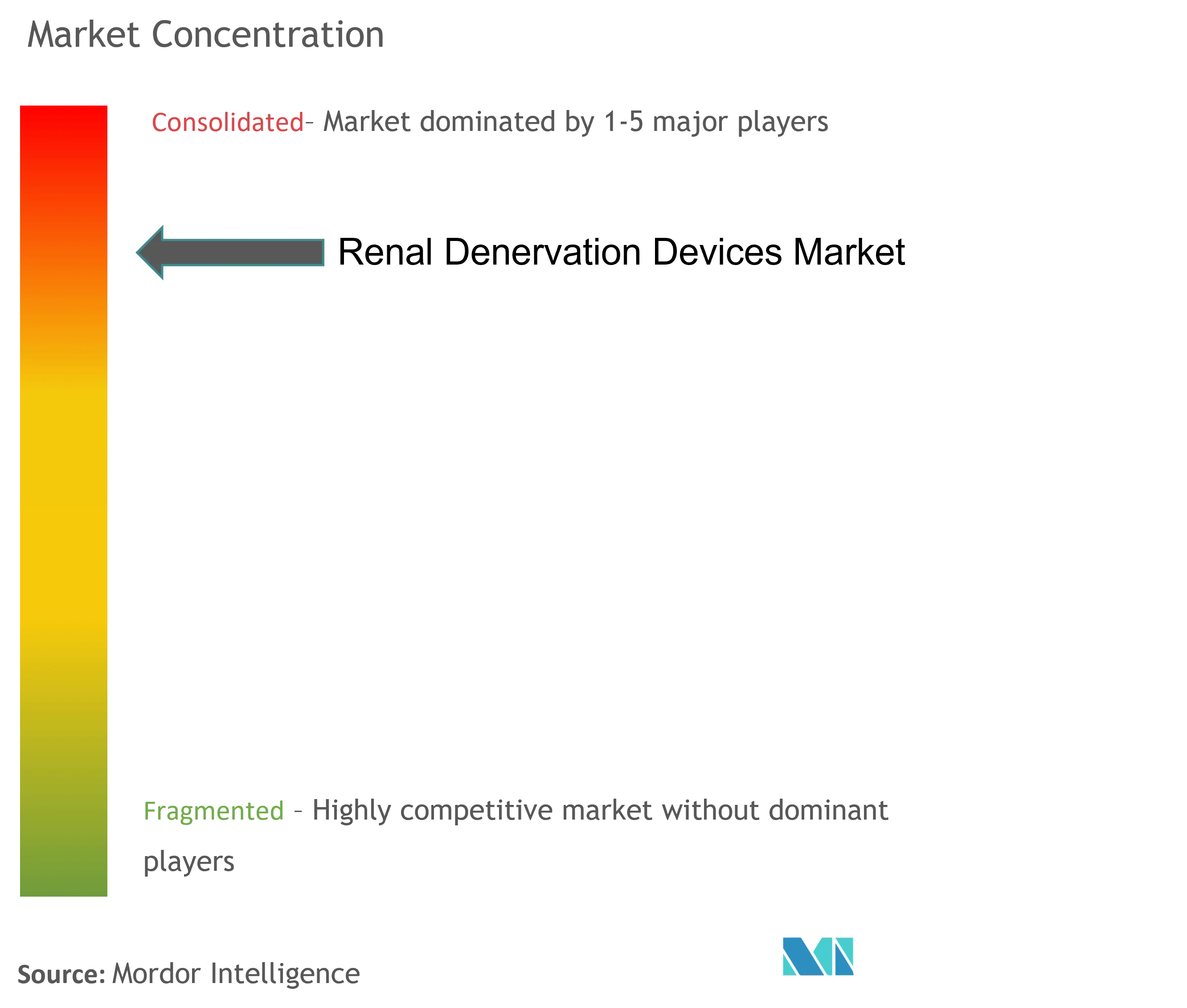
The renal denervation devices market exhibits a relatively consolidated structure dominated by large medical device conglomerates with established global presence and substantial research capabilities. These major players leverage their extensive distribution networks, strong financial resources, and existing relationships with healthcare providers to maintain their market positions. The market is characterized by significant barriers to entry, including high capital requirements, stringent regulatory standards, and the need for extensive clinical validation, which favors established players. Merger and acquisition activities are primarily focused on acquiring innovative technologies and expanding therapeutic applications, with larger companies actively seeking smaller firms with promising technological platforms.
The competitive dynamics are shaped by the presence of specialized players who focus exclusively on renal denervation devices technology, alongside diversified medical device manufacturers who incorporate these devices into their broader cardiovascular portfolios. Regional market penetration varies significantly, with Europe leading in terms of product adoption and clinical implementation, followed by Asia-Pacific and North America. The market structure encourages collaborative approaches, with companies forming strategic alliances to combine complementary technologies and share development costs while accelerating market access and expanding geographical reach.
Innovation and Market Access Drive Success
Success in the renal denervation devices market requires a multi-faceted approach combining technological innovation with strategic market access initiatives. Incumbent companies must focus on developing next-generation devices with improved efficacy and safety profiles while simultaneously building robust clinical evidence bases to support adoption. Market leaders need to invest in physician education and training programs to drive procedure adoption while also working closely with healthcare providers to demonstrate the economic value proposition of their technologies. Companies must also navigate the complex reimbursement landscape and work with payers to establish favorable coverage policies.
For emerging players and contenders, differentiation through technological innovation and specialized therapeutic applications presents the most viable path to market success. These companies need to focus on developing unique features or therapeutic approaches that address specific unmet needs in the treatment of resistant hypertension. The competitive landscape is further influenced by the high concentration of buyers, primarily hospitals and specialized cardiac centers, which necessitates strong relationship building and comprehensive support services. Regulatory requirements continue to evolve, particularly regarding clinical evidence requirements and safety demonstrations, making regulatory expertise and compliance capabilities critical success factors for all market participants. Additionally, the integration of interventional cardiology devices within the broader cardiovascular treatment landscape enhances the value proposition of a comprehensive renal denervation system.
Renal Denervation Devices Industry Leaders
Boston Scientific Corporation
Medtronic PLC
ReCor Medical
Symple Surgical
Mercator MedSystems
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- May 2022: Medtronic PLC released data showing the benefits of the Symplicity Blood Pressure Procedure with the Symplicity Spyral Renal Denervation (RDN) system on achieving target blood pressure level ranges. As part of the SPYRAL HTN Global Clinical Program, three-year data from SPYRAL HTN-ON MED and GSR-DEFINE were presented at EuroPCR 2022 as late-breaking clinical trials.
- March 2022: ReCor Medical Inc., a fully owned subsidiary of Otsuka Medical Devices Co., Ltd, completed the enrollment in their RADIANCE-II pivotal trial of the Paradise Ultrasound Renal Denervation (uRDN) System for the treatment of uncontrolled hypertension.
Global Renal Denervation Devices Market Report Scope
As per the scope of the report, renal denervation devices are one of several innovative device-based interventional therapies that help control blood pressure. It is safe, minimally invasive, and does not leave any implants behind. The renal denervation procedure uses radiofrequency ablation to burn the nerves in the renal arteries. This process causes a reduction in nerve activity, which decreases blood pressure.
The Renal Denervation Devices Market is segmented by technology (radiofrequency-based, ultrasound-based, and micro-infusion-based) and geography (North America, Europe, Asia-Pacific, and Rest of the World). The market sizes and forecasts are provided in terms of value (USD million) for all the above segments.
| Radiofrequency-based |
| Ultrasound-based |
| Micro-infusion-based |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Rest of the World |
| By Technology | Radiofrequency-based | |
| Ultrasound-based | ||
| Micro-infusion-based | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Rest of the World | ||


Key Questions Answered in the Report
What is the current Renal Denervation Devices Market size?
The Renal Denervation Devices Market is projected to register a CAGR of 35.20% during the forecast period (2026-2031)
Who are the key players in Renal Denervation Devices Market?
Boston Scientific Corporation, Medtronic PLC, ReCor Medical, Symple Surgical and Mercator MedSystems are the major companies operating in the Renal Denervation Devices Market.
Which is the fastest growing region in Renal Denervation Devices Market?
North America is estimated to grow at the highest CAGR over the forecast period (2026-2031).
Which region has the biggest share in Renal Denervation Devices Market?
In 2025, the Europe accounts for the largest market share in Renal Denervation Devices Market.
What years does this Renal Denervation Devices Market cover?
The report covers the Renal Denervation Devices Market historical market size for years: 2021, 2022, 2023, 2024 and 2025. The report also forecasts the Renal Denervation Devices Market size for years: 2026, 2027, 2028, 2029, 2030 and 2031.
Page last updated on:







