Ultrasonic Nebulizers Market Size and Share
Ultrasonic Nebulizers Market Analysis by Mordor Intelligence
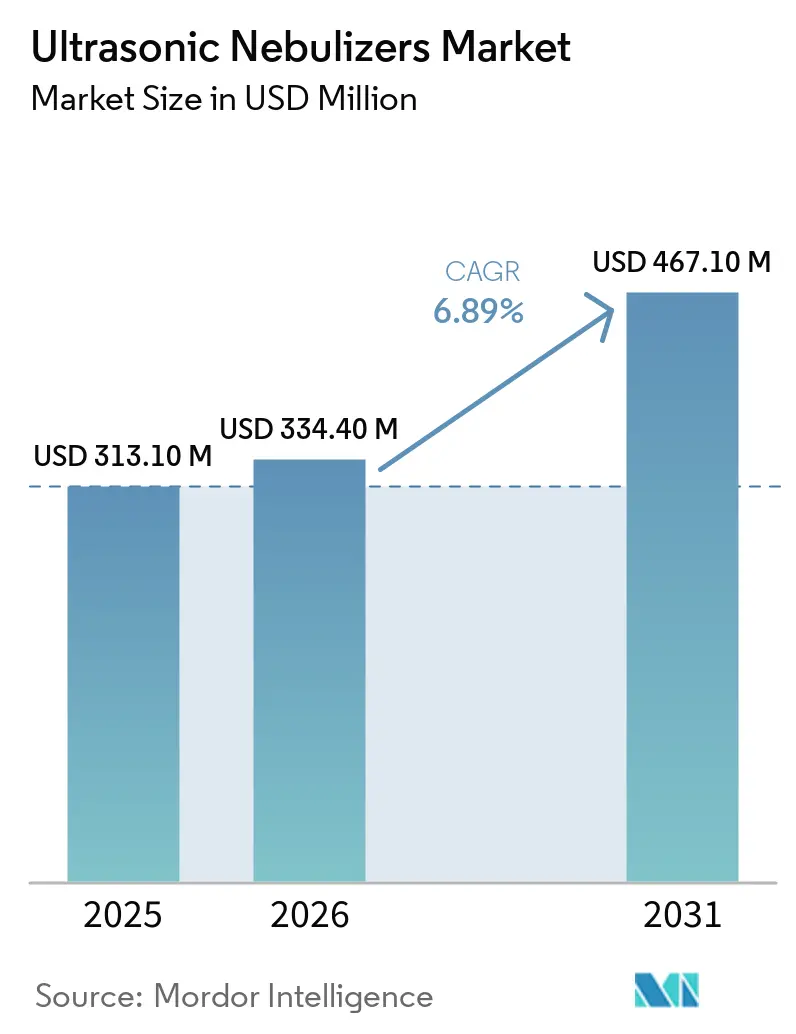
The Ultrasonic Nebulizers Market size was valued at USD 313.10 million in 2025 and is estimated to grow from USD 334.40 million in 2026 to reach USD 467.10 million by 2031, at a CAGR of 6.89% during the forecast period (2026-2031).
Home-care demand and pediatric comfort continue to generate unit volumes, but hospital buyers are steering high-value biologics toward vibrating-mesh systems, leaving ultrasonic models to compete on silent operation and entry-level pricing. Small-volume devices dominate everyday use, yet large-volume platforms are carving a foothold in intensive-care units that require continuous bronchodilator delivery. Portable, battery-driven handhelds are narrowing the performance gap with tabletop designs, enabling active patients to complete therapy in under six minutes. Competitive intensity is escalating as low-cost Asian OEMs pair vertical integration with aggressive Western partnerships, accelerating price pressure across the ultrasonic nebulizers market.
Key Report Takeaways
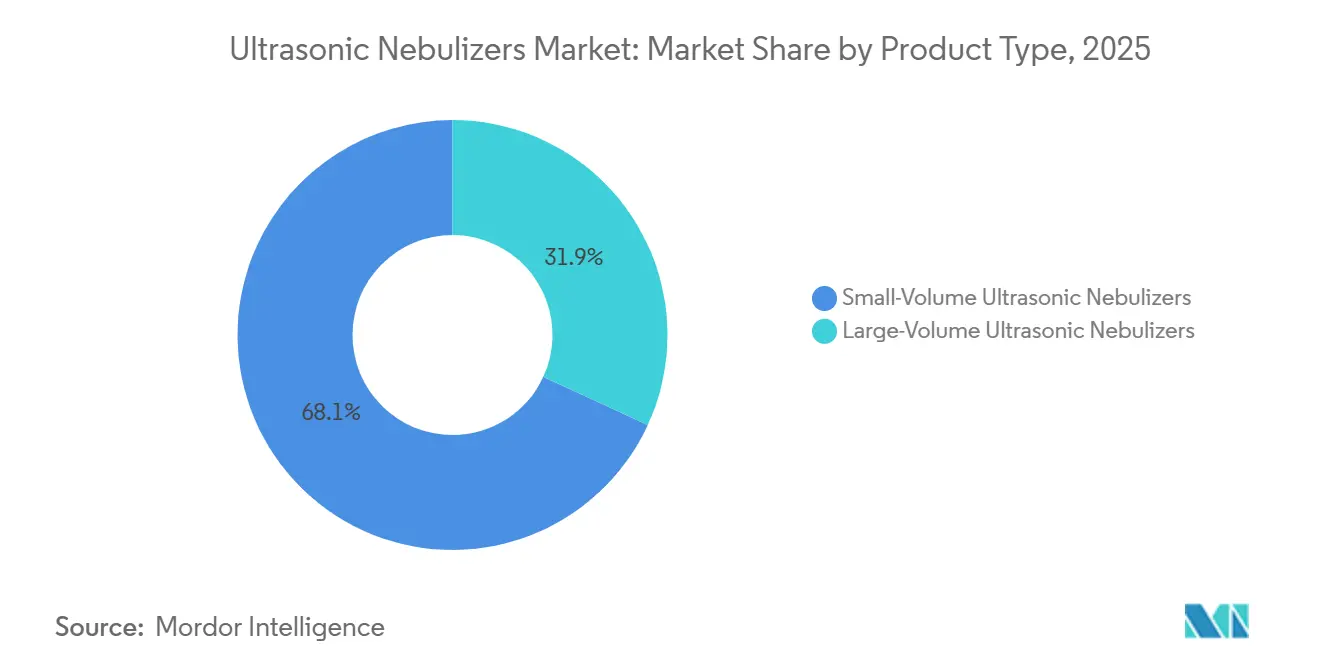
- By product type, small-volume devices led with 68.13% of the ultrasonic nebulizers market share in 2025, while large-volume units are forecast to post a 7.90% CAGR through 2031.
- By portability, tabletop configurations accounted for 65.76% of the ultrasonic nebulizers market size in 2025, yet handheld models are projected to expand at an 8.12% CAGR to 2031.
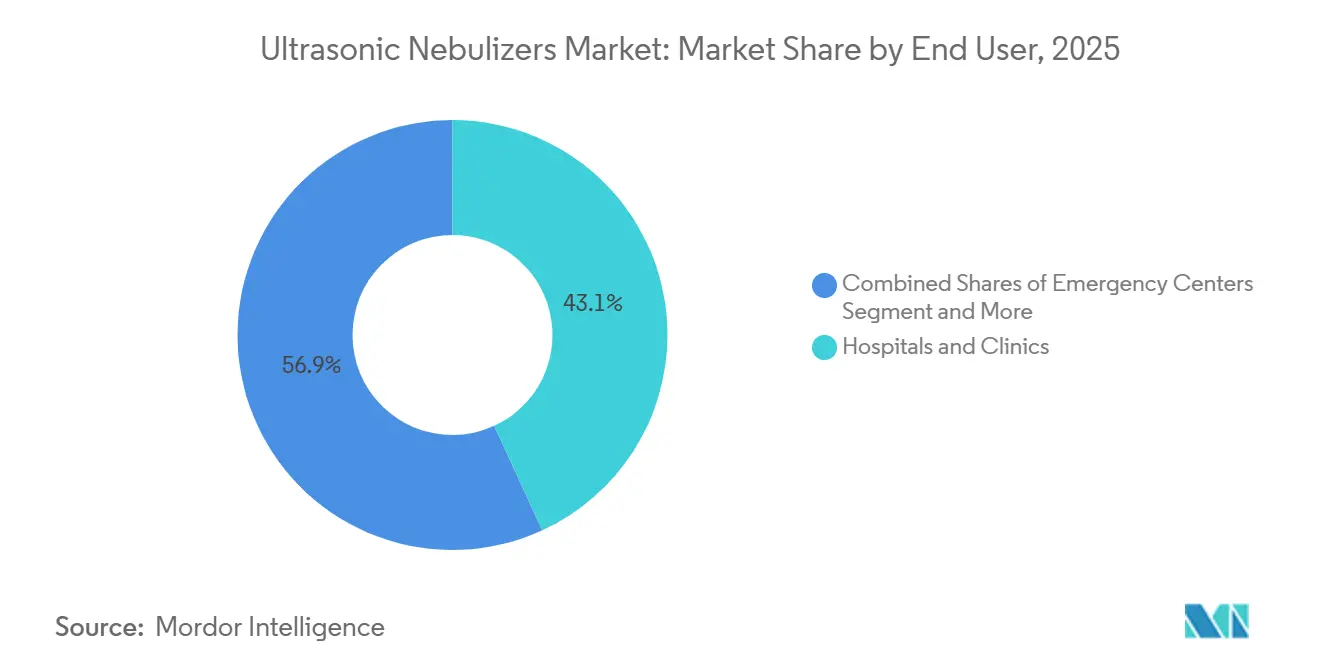
- By end user, hospitals and clinics held 43.15% share of the ultrasonic nebulizers market in 2025; the home-healthcare channel is advancing at an 8.34% CAGR through 2031.
- By application, COPD captured 38.15% share in 2025, whereas the asthma segment is on track for the fastest 7.88% CAGR to 2031.
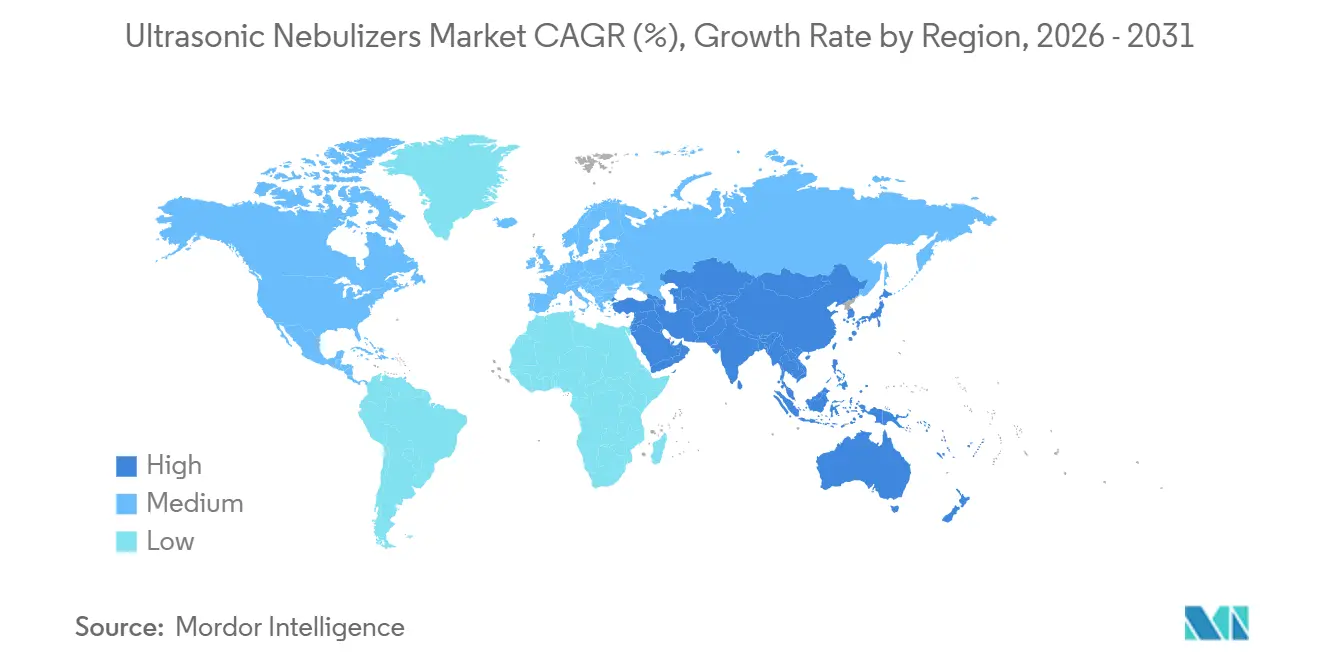
- By geography, North America commanded 42.15% of the ultrasonic nebulizers market share in 2025, while Asia-Pacific is set to grow at 8.06% CAGR over 2026-2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Ultrasonic Nebulizers Market Trends and Insights
Driver Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising home-care adoption and e-commerce availability for respiratory devices | +1.2% | Global, with early gains in North America, Western Europe, urban Asia-Pacific | Medium term (2-4 years) |
| Silent operation and faster treatments vs. jet nebulizers improve adherence | +0.9% | Global, especially pediatric and geriatric niches | Short term (≤ 2 years) |
| Pediatric comfort and tolerance due to low-noise operation | +0.7% | North America, Europe, urban Asia-Pacific | Short term (≤ 2 years) |
| Growing chronic respiratory disease burden requiring inhalation therapy in home settings | +1.5% | Global, strongest absolute growth in Asia-Pacific and aging Europe, Japan | Long term (≥ 4 years) |
| Veterinary respiratory-care adoption (equine and companion animals) | +0.3% | North America, Western Europe, Australia | Medium term (2-4 years) |
| Emerging low-cost Asian OEMs expanding access | +1.1% | Asia-Pacific core, Middle East & Africa, Latin America | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Home-Care Adoption and E-Commerce Availability for Respiratory Devices
Durable-medical-equipment reimbursement is tilting toward at-home therapy, and online storefronts now bypass pharmacy mark-ups by shipping ultrasonic nebulizers within 48 hours and at 40-60% discounts[1]Centers for Medicare & Medicaid Services, “2024 Improper Payment Data,” cms.gov. Medicare documentation lapses caused a 53.9% improper-payment rate in 2024, nudging patients toward self-pay e-commerce channels. Manufacturers that embed prescription validation and automated claims filing directly into web check-out flows can capture displaced demand. The shift underpins a broader re-allocation of value: as remote-patient-monitoring codes proliferate, device makers that offer cloud dashboards and telehealth APIs will strengthen recurring-revenue streams. Consequently, the ultrasonic nebulizers market is transitioning from hardware sales to integrated digital-health subscriptions.
Silent Operation and Faster Treatments vs. Jet Nebulizers Improve Adherence
Piezoelectric frequencies between 100 kHz and 500 kHz generate fine droplets at sub-45 dBA, roughly half the acoustic footprint of jet models and a critical parameter for user comfort[2]Journal of Evaluation in Clinical Practice, “Pediatric Anxiety Reduction Via Soundproof Nebulizers,” wiley.com. In pediatric cohorts, anxiety scores fell 30% when soundproof housings and music playback were added to ultrasonic units, translating into visibly higher treatment completion rates. Ultrasonic nebulizers also cut dose time for a 2.5 ml solution to 4–6 minutes versus 10–15 minutes for pneumatic systems, further limiting distraction. Heat buildup, however, remains a design caveat: temperature rises of 10–15 °C can degrade protein biologics, requiring hybrid ultrasonic-mesh designs to preserve medication potency. Vendors that master such dual-mode platforms will protect their share against hospital migration to pure mesh.
Pediatric Comfort and Tolerance Due to Low-Noise Operation
Around one fourth of preschoolers reach high adherence on nebulizer regimens, and device noise is a leading deterrent. Ultrasonic units mitigate this barrier through whisper-quiet output and child-friendly form factors that demedicalize therapy. Omron’s 100-gram MicroAir NE-U100 qualifies for U.S. HCPCS E0574 reimbursement and neatly exemplifies the pull of quiet hardware paired with reimbursement access. Yet studies show that caregiver coaching and app-based reminders explain more adherence variance than acoustics alone, implying that silent operation must be backed by digital-engagement layers to sustain pediatric gains across the ultrasonic nebulizers market.
Growing Chronic Respiratory Disease Burden Requiring Inhalation Therapy in Home Settings
In the Asia-Pacific region, household solid-fuel combustion and ambient PM2.5 pollution now outweigh smoking as dominant risk factors. In China, 99% of residents breathe air exceeding the 5 µg/m³ WHO guideline. Because hospital capacity cannot scale at the same pace, payers and clinicians are prescribing ultrasonic devices for long-term inhalation therapy at home. That macro-demand, though, increases the infection-control burden on lay caregivers, creating a parallel opportunity for self-sterilizing chambers and antimicrobial coatings.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Medication incompatibility (suspensions/proteins); heat-related instability | -1.3% | Global, high-value hospital and specialty channels | Short term (≤ 2 years) |
| Hospital shift to vibrating-mesh systems cannibalizes ultrasonic demand | -0.9% | North America, Western Europe, developed Asia-Pacific | Medium term (2-4 years) |
| Infection-control and cleaning complexity in home settings | -0.6% | Global, more severe where health literacy is low | Medium term (2-4 years) |
| Added compliance burden under EU MDR and select LMIC rules | -0.5% | Europe, emerging markets with tightening standards | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Medication Incompatibility (Suspensions/Proteins); Heat-Related Instability
High-frequency vibration generates shear stress that cuts biologic stability by 22% in recombinant DNase compared with mesh delivery. Thick suspensions likewise risk particle agglomeration, forcing formulators to exclude ultrasonic platforms from liposomal or antibody therapies. Medicare’s non-coverage of large-volume ultrasonic codes reflects this pharmacological constraint, effectively limiting the ultrasonic nebulizers market size for high-cost drugs. Until transducers with real-time temperature control mature, the segment will concentrate on small-molecule bronchodilators where heat sensitivity is minimal.
Hospital Shift to Vibrating-Mesh Systems Cannibalizes Ultrasonic Demand
Health-system purchasers are trading up to mesh devices that blend silent operation with superior medication compatibility. Philips InnoSpire Go delivers a 2.5 ml salbutamol dose in four minutes and sustains 30 treatments per charge. Such performance, and formulary inclusion for biologics, has shortened emergency-department stays and swayed protocol committees. Ultrasonic units that lack hybrid capability are being relegated to cost-driven generic bronchodilator use, dampening institutional sales growth within the ultrasonic nebulizers market.
Segment Analysis
By Product Type: ICU Demand Elevates Large-Volume Systems
Large-volume ultrasonic platforms are posting a 7.90% CAGR through 2031, eclipsing the broader ultrasonic nebulizers market growth as emergency and intensive-care units deploy them for uninterrupted 6–15 ml/min bronchodilator streams. Yet small-volume devices still held a commanding 68.13% ultrasonic nebulizers market share in 2025, anchored by out-patient maintenance regimens. Over the forecast horizon, dual-mode transducers that toggle flow rates could shrink performance gaps and let suppliers cross-sell large-volume functionality to home users requiring overnight ventilation.
Second-order effects revolve around materials science. Lead-free potassium-sodium-niobate piezoceramics match PZT performance while satisfying RoHS rules, lowering EU regulatory friction and potentially trimming the component cost curve for both volume classes. Early adopters could thus defend margins even as price wars intensify.
By Portability: Miniaturization Spurs Wearable Adoption
Tabletop units dominated value in 2025, yet handheld devices are growing at 8.12% CAGR as lithium-polymer cells deliver 90-minute runtimes in sub-50-gram bodies. Portable devices now achieve ISO 27427 aerosol consistency, allowing payers to view them as therapeutic equivalents and widening the ultrasonic nebulizers market size for wearables. Brands such as Medel’s 200-gram SMART nebulizer illustrate how USB-charged designs can balance flow rate and acoustic comfort.
Marketing strategies increasingly position wearables as lifestyle enablers rather than clinical aids. Nuvita’s 35-gram collar-style mesh model incorporates self-cleaning chambers, essentially inserting a compliance feature that mitigates caregiver burden in pediatric segments. As a result, portability is shifting from a premium to a default expectation inside the ultrasonic nebulizers market.
By End User: Connected Home Devices Capture Share
Institutional channels retained 43.15% of revenue in 2025, but home healthcare is on an 8.34% CAGR trajectory that will redefine purchasing criteria. Home use leans on reimbursement codes that now reward remote-physiologic-monitoring uploads, a capability embedded in AI-powered NebSync cartridges. The ultrasonic nebulizers market size for hospitals is stabilizing as value-based-care models pressure administrators to discharge faster and keep chronic-care patients outside expensive beds.
Remote data capture also reshapes clinician workflow: adherence dashboards flag missed doses in real time, enabling nurses to intervene before exacerbations require re-admission. Such digital reinforcement makes at-home ultrasonic units more than commodity hardware; instead, they become nodes in a telehealth continuum, unlocking subscription revenues.

By Application: Asthma Outpaces COPD on Pediatric Gains
COPD retained a 38.15% share in 2025, aided by an aging global population. Yet asthma prescriptions are expanding at 7.88% CAGR, granted sub-45 dBA operation that lowers treatment anxiety and improves pediatric compliance. Ultrasonic nebulizers market size for cystic-fibrosis medications stays niche because aztreonam and amikacin liposomes still require mesh delivery to prevent heat-induced degradation.
Market education is another differentiator. Parental familiarity with connected games and smartphone alerts dovetails with adherence-gamification apps bundled by leading vendors. This cultural fit accelerates replacement cycles in the pediatric asthma subsegment, sustaining double-digit unit growth.
Geography Analysis
North America generated 42.15% of 2025 revenue thanks to Medicare Part B coverage of small-volume treprostinil nebulizers, yet improper-payment audits underscore tightening documentation hurdles that may slow future uptake. Hospitals are simultaneously migrating to vibrating-mesh systems, restraining institutional ultrasonic volume. Canadian provincial formularies mirror U.S. reimbursement but impose lower paperwork overhead, cushioning regional demand. Europe’s outlook remains tethered to the EU MDR. Mandatory EUDAMED uploads and EUR 50,000-150,000 Class IIa/IIb certification fees lengthen launch timelines, favoring incumbents over start-ups. Nevertheless, Germany’s outpatient budget reforms now allocate separate device line items, potentially offsetting regulatory drag by expanding reimbursable SKUs.
Asia-Pacific is the growth engine, accelerating at 8.06% CAGR to 2031 as local OEMs flood the ultrasonic nebulizers market with sub-USD 50 bill-of-materials devices. China’s omnipresent air pollution and India’s dense urban smog raise baseline respiratory morbidity, driving absolute device demand higher. Telehealth penetration in Metro Manila and Jakarta adds distribution leverage because online pharmacies can clear customs faster than brick-and-mortar channels.
Middle East & Africa and South America trail in absolute size but showcase the highest e-commerce headroom. Saudi Arabia’s e-pharmacy regulations enacted in 2025 legalized direct-to-consumer inhalation-therapy shipments. Brazil’s ANVISA fast-track for connected medical devices positions cloud-linked ultrasonic models to leapfrog older jet systems.
Competitive Landscape
First-tier incumbents Omron, PARI GmbH and Koninklijke Philips N.V. leverage brand equity and published clinical evidence to preserve margins. Omron crossed the 50-million-unit milestone in November 2025 and is rolling out app-linked nebulizers for India’s variable grid conditions[3]Omron Healthcare, “Global Sales Milestone Press Release,” omronhealthcare.com. Philips’ InnoSpire Go capitalizes on its hospital footprint, bundling staff training and consumables to lock in formulary preference.
Asian challengers deploy joint ventures to vault trade barriers. Yuwell’s 9.9% stake in Inogen secures U.S. distribution while giving Inogen low-cost production in Jiangsu. Contec Medical Systems mirrors the template with Gulf distributors that localize labeling under fast-track import licenses. Innovation hotspots include AI analytics. IVPMED’s NebSync scrapes real-time flow data, encrypts it on-chain, and surfaces dashboards that satisfy CPT remote-monitoring billing. Adherium’s smart inhaler, although metered-dose rather than ultrasonic, validates reimbursement viability for connected respiratory devices. Veterinary niches function as pilot grounds: Hippomed’s equine Air One platform sells at premium prices and allows rapid iteration on antimicrobial coatings before migrating technology into human devices.
Competitive strategy is tilting toward hybrid ultrasonic-mesh architectures to defend hospital share. Suppliers able to certify ISO 18562 particulate emissions alongside IEC 62366-1 usability will widen tender eligibility and set higher entry barriers for low-cost imitators. Overall, differentiation is moving downstream from the transducer to the software layer, making cloud ecosystems the next arena of value capture within the ultrasonic nebulizers market.
Ultrasonic Nebulizers Industry Leaders
Omron Healthcare
Koninklijke Philips N.V.
JIANGSU YUYUE MEDICAL EQUIPMENT & SUPPLY CO., LTD.
PARI GmbH
GF Health Products
- *Disclaimer: Major Players sorted in no particular order



Recent Industry Developments
- February 2026: CMS has updated its Nebulizer Policy Article (A52466), requiring specific modifiers (KX, GA, or GZ) for all claims to ensure compliance with coverage rules.
- November 2025: OMRON Healthcare crossed an important threshold, confirming that global nebulizer sales have exceeded 50 million units. The achievement highlighted the company’s decades-long focus on giving people reliable tools to manage respiratory illnesses and maintain better day-to-day health.
Global Ultrasonic Nebulizers Market Report Scope
As per the scope of the report, ultrasonic nebulizers represent a second generation of nebulizer technology designed to convert liquid medication into a fine mist for inhalation. Unlike traditional jet nebulizers that rely on compressed air, these devices use a piezoelectric crystal that vibrates at high frequencies (typically 1–3 MHz) to break down the liquid into tiny aerosol particles. This mechanism allows for virtually silent operation and typically faster treatment times compared to compressor-based models.
The ultrasonic nebulizers market is segmented by product type, portability, end-user, application, and geography. Based on product type, the market is segmented into large-volume ultrasonic nebulizers and small-volume ultrasonic nebulizers. By portability, the market is segmented into tabletop/benchtop and handheld/portable. By end users, the market is segmented into hospitals & clinics, emergency centers, and home healthcare. Geographically, the market is segmented into North America, Europe, Asia-Pacific, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Large-volume ultrasonic nebulizers |
| Small-volume ultrasonic nebulizers |
| Tabletop/Benchtop |
| Handheld/Portable |
| Hospitals & Clinics |
| Emergency Centers |
| Home Healthcare |
| COPD |
| Asthma |
| Cystic Fibrosis |
| Bronchitis & Lower Respiratory Infections |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Large-volume ultrasonic nebulizers | |
| Small-volume ultrasonic nebulizers | ||
| By Portability | Tabletop/Benchtop | |
| Handheld/Portable | ||
| By End-user | Hospitals & Clinics | |
| Emergency Centers | ||
| Home Healthcare | ||
| By Application | COPD | |
| Asthma | ||
| Cystic Fibrosis | ||
| Bronchitis & Lower Respiratory Infections | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large is the ultrasonic nebulizer market in 2026?
The ultrasonic nebulizers market size stands at about USD 313.1 million in 2026 and is projected to reach USD 467.1 million by 2031, expanding at a 6.89% CAGR
Which product category holds the biggest share?
Small-volume units accounted for 68.13% of ultrasonic nebulizers market share in 2025, driven by home-care use.
Which segment is growing fastest?
Handheld portable devices are set to advance at an 8.12% CAGR through 2031 on the strength of battery miniaturization.
What region leads to global demand?
North America generated 42.15% of 2025 revenue, yet Asia-Pacific is expected to post the highest 8.06% CAGR to 2031.
Page last updated on:









