Steam And Hydrogen Peroxide Biological Indicators Market Size and Share
Steam And Hydrogen Peroxide Biological Indicators Market Analysis by Mordor Intelligence
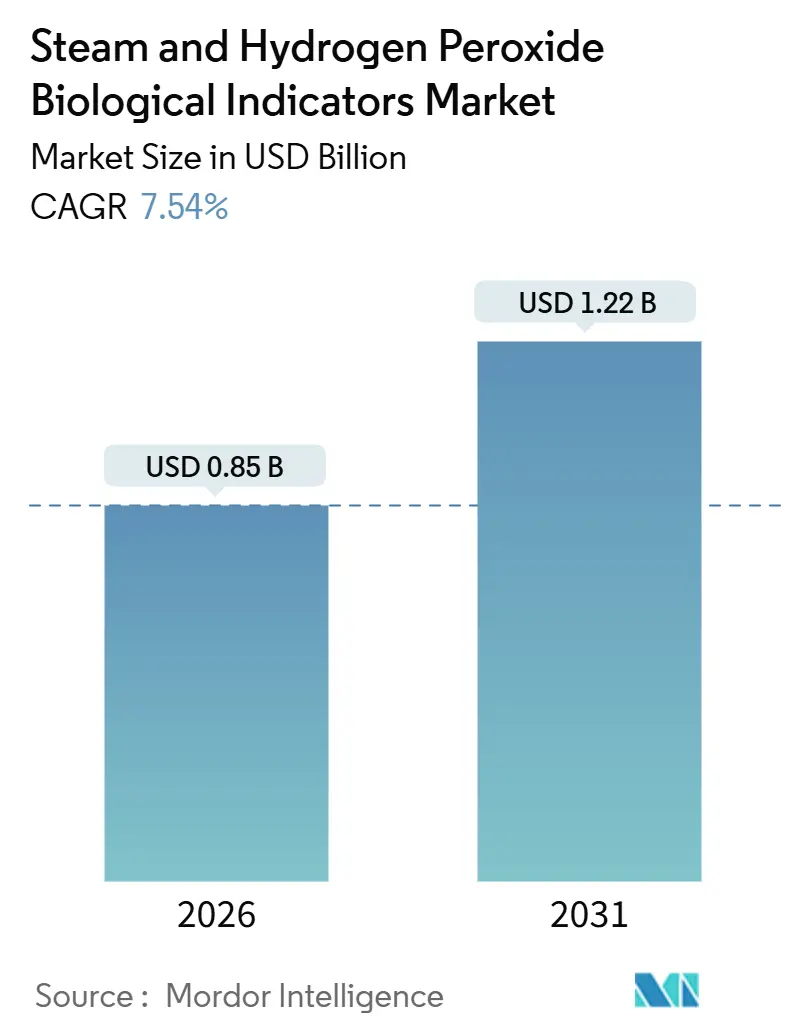
The Steam And Hydrogen Peroxide Biological Indicators Market size is estimated at USD 0.85 billion in 2026, and is expected to reach USD 1.22 billion by 2031, at a CAGR of 7.54% during the forecast period (2026-2031).
Tightening global sterilization rules, growing surgical volumes, and wider use of low-temperature sterilization in pharmaceuticals and complex medical devices are reinforcing demand. Hospitals rely on self-contained indicators to document every implant load, while rapid-read units shorten reprocessing cycles, freeing scarce instrument sets for back-to-back surgeries. Vaporized hydrogen peroxide (VHP) indicators are gaining ground as single-use bioprocess systems and robotic instruments expand. Regional manufacturers, especially in Latin America and China, compete on price, prompting incumbents to bundle indicators with autoclaves and traceability software. Cloud-based data platforms now link indicator results with autoclave parameters so that multi-site hospital systems can flag failures in real time and prove compliance during audits.
Key Report Takeaways
- By product type, self-contained biological indicators led with 46.23% revenue share in 2025; rapid-read variants are advancing at a 10.35% CAGR through 2031.
- By sterilization method, steam retained 52.56% of steam and hydrogen peroxide biological indicators market share in 2025, while VHP is forecast to grow at 10.44% CAGR to 2031.
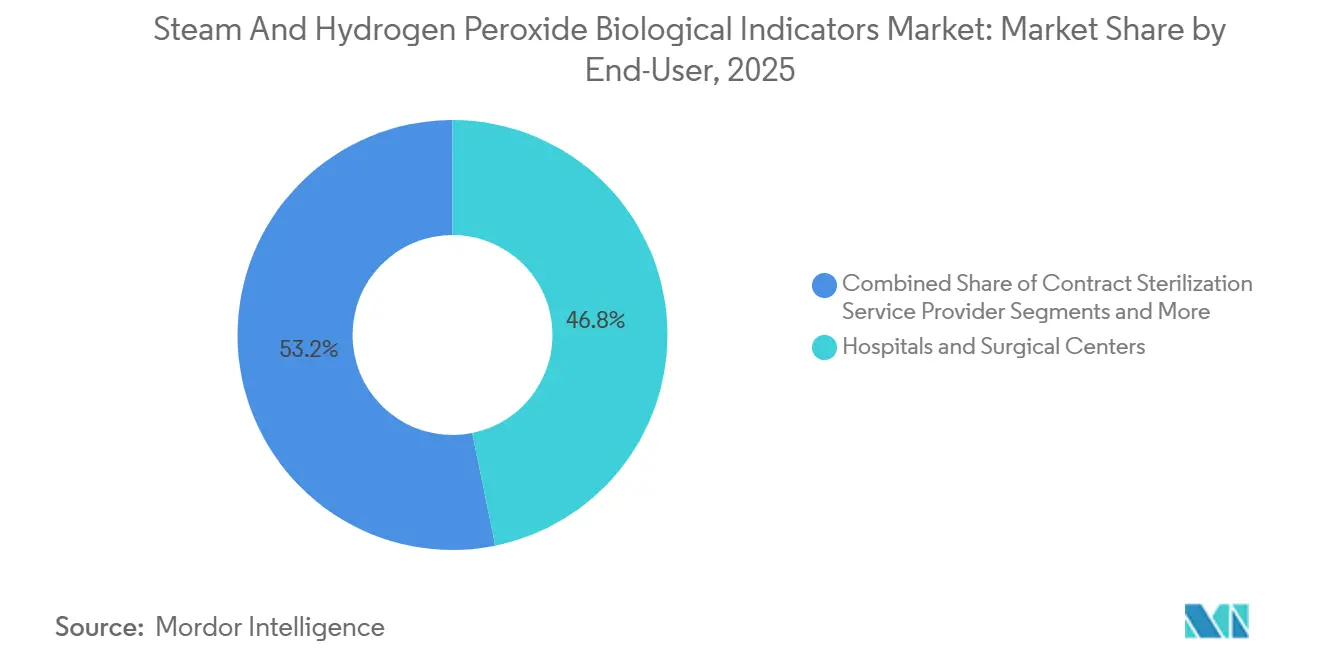
- By end-user, hospitals and surgical centers accounted for 46.82% of the steam and hydrogen peroxide biological indicators market size in 2025, whereas contract sterilizers post the fastest 11.57% CAGR through 2031.
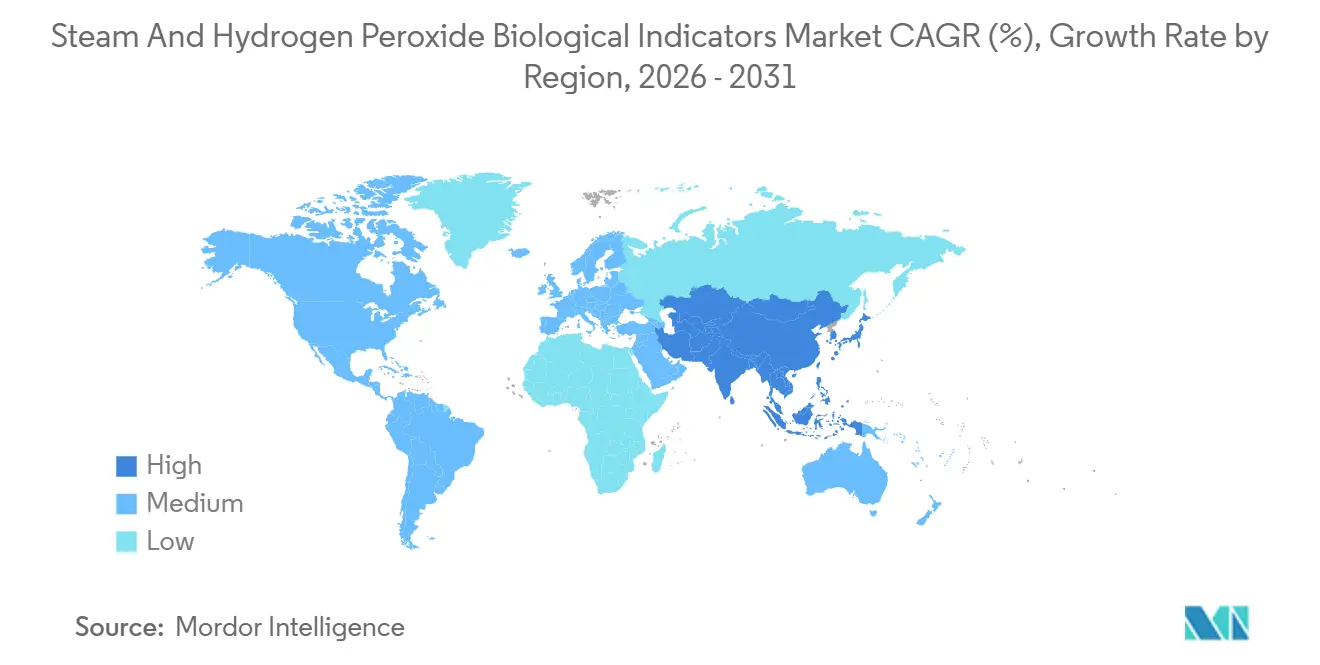
- By geography, North America captured 41.33% of 2025 revenue, yet Asia-Pacific is on track for a 9.49% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Steam And Hydrogen Peroxide Biological Indicators Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Surgical Volumes and HAI-Prevention | +1.5% | North America, Western Europe, global influence | Medium term (2-4 years) |
| Stricter ISO 11138 and FDA Mandates | +1.3% | North America, European Union, China | Long term (≥4 years) |
| Adoption of Rapid-Read Self-Contained BIs | +1.2% | North America, European Union, urban Asia-Pacific | Short term (≤2 years) |
| Growth in Contract Sterilization Outsourcing | +1.4% | North America, emerging Asia-Pacific | Medium term (2-4 years) |
| Surge in Low-Temperature Device Sterilization | +1.1% | United States, Germany, Switzerland, India | Medium term (2-4 years) |
| Cloud-Connected BI Traceability Platforms | +0.9% | North America, European Union, GCC nations | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
Rising Surgical Volumes and HAI-Prevention Focus
Elective orthopedic and cardiovascular procedures climbed 8% year over year across OECD hospitals in 2025, reversing pandemic deferrals.[1]Centers for Disease Control and Prevention, “National Healthcare Safety Network 2023 Manual,” cdc.gov At the same time, surgical site infections in several categories ticked upward, prompting facilities to adopt per-load biological indicator (BI) testing for implant trays. U.S. Joint Commission rules effective January 2025 require BI documentation within 48 hours of surgery, so departments are shifting from weekly checks to rapid-read units that turn results around in under an hour.[2]The Joint Commission, “Comprehensive Standards Manual 2025,” jointcommission.org The change is most evident in ambulatory surgery centers that must reprocess instruments between morning and afternoon cases without growing their tray inventory. Higher BI consumption lifts recurring revenue for suppliers and underpins a steady growth baseline for the steam and hydrogen peroxide biological indicators market.
Stricter ISO 11138 and FDA Mandates
ISO 11138-8:2021 cut standard incubation times from 24 hours to as little as 1 hour, bringing fluorescence-based readouts into mainstream practice.[3]International Organization for Standardization, “ISO 11138-8:2021,” iso.org The FDA endorsed this approach by clearing Solventum’s Attest 1493 rapid BI with a 30-minute protocol in November 2024, cementing rapid-read technology as an acceptable validation path. Updated FDA guidance now demands six consecutive successful BI challenges before manufacturers may rely solely on physical parameters. China’s NMPA mirrored the requirement in July 2024 for contract sterilizers serving export markets. Tighter oversight compels hospitals and device makers to purchase larger BI batches, supporting mid-term growth even as some facilities pursue parametric release.
Adoption of Rapid-Read Self-Contained BIs
Rapid-read units use enzyme or pH-shift chemistry to compress validation from 24 hours to under 1 hour. STERIS reported 13.1% organic Healthcare-segment growth in FY2025, driven largely by rapid-read consumable contracts. A 2025 IAHCSMM survey found that 42% of U.S. ambulatory surgery centers had adopted rapid BIs, up from 28% in 2023, citing labor savings and reduced idle inventory. Price premiums of 40%–60% remain a barrier for low-volume clinics, yet high-throughput sites justify the spend with faster room turns. The cascading effect boosts overall penetration, aligning with the forward momentum of the steam and hydrogen peroxide biological indicators market.
Growth in Contract Sterilization Outsourcing
Device makers increasingly outsource terminal sterilization to avoid USD 10 million-plus capital outlays for ethylene oxide suites. STERIS’s Applied Sterilization Technologies division grew 9.3% organically in FY2025 as clients shifted validation work off-site. Contract sterilizers buy BIs in bulk and negotiate better pricing, but they also mandate best-practice validation, raising total volume. A temporary FDA shutdown of a large Midwest contract sterilizer in 2024 halted shipments from 14 device manufacturers, spotlighting systemic supply-chain risk. The episode underlines the importance of diversified sterilization capacity and fuels interest in additional third-party providers.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Cost and Supply-Chain Volatility for Rapid BIs | -0.8% | Global, acute in emerging markets | Short term (≤2 years) |
| Skills Gap Causing Improper BI Usage | -0.6% | South Asia, Sub-Saharan Africa, Latin America | Medium term (2-4 years) |
| Consumable-Averse Reimbursement in Emerging Markets | -0.7% | Latin America, South Asia, Southeast Asia | Medium term (2-4 years) |
| Sustainability Scrutiny of Disposable Spore Carriers | -0.4% | European Union, North America, nascent in Asia-Pacific | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
High Cost and Supply-Chain Volatility for Rapid BIs
Rapid-read units depend on enzyme substrates that require refrigeration, raising shipping costs 30%–50%. Mesa Laboratories cited a 120-basis-point gross-margin hit in Q1 FY2025 after substrate inflation. A 2024 fire at a European enzyme plant delayed launches for several BI makers, as reformulation needed fresh FDA clearance. Smaller hospitals in emerging economies often default to 24-hour strips when rapid units are unavailable, creating unpredictable demand swings in the steam and hydrogen peroxide biological indicators market.
Skills Gap Causing Improper BI Usage
Turnover among sterile processing staff runs 20%–30% annually, leaving gaps in expertise. An FDA inspection in 2024 logged 17 instances where BIs were placed in low-challenge spots, voiding the test. Certification programs require 400 supervised hours, yet many facilities cannot spare staff for extended training. Misuse undermines rapid-read benefits, so suppliers now embed QR-linked videos and placement guides in packaging.
*Our forecasts treat driver/restraint impacts as directional, not additive. The impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Self-Contained Systems Face Rapid-Read Disruption
Self-contained indicators held a 46.23% share in 2025, reflecting integrated spore vials and media that cut handling errors. Rapid-read units, though costlier, are forecast to expand at a 10.35% CAGR as high-volume sites pay for faster turnover, underscoring the competitive thrust of the steam and hydrogen peroxide biological indicators market. Spore strips remain a low-cost staple in price-sensitive regions, while spore suspensions support aseptic pharmaceutical production where each batch may consume multiple BIs.
Rising demand for rapid-read technology reshapes margins. Tuttnauer’s 30-minute fluorescence BI pairs exclusively with its autoclaves, anchoring customers to proprietary consumables. Chinese firms such as Zhejiang Tailin ship ISO-compliant strips at half Western prices, pushing incumbents to focus on training and digital support rather than price cuts.


By Sterilization Method: VHP Gains as Device Complexity Rises
Steam accounted for 52.56% of 2025 demand, favored for stainless-steel tools and low per-cycle cost. Yet VHP units are growing 10.44% annually as polymer devices, robotic arms, and single-use bioprocess bags proliferate. This shift directly lifts VHP indicator consumption and widens the steam and hydrogen peroxide biological indicators market size for low-temperature modalities.
Geographically, VHP adoption centers on U.S., German, Swiss, and Indian pharmaceutical hubs. STERIS’s STERRAD base exceeds 25,000 units, each using multiple BIs per cycle. An ISO 11138-6 draft due in 2026 should normalize VHP BI performance criteria and accelerate global uptake.
By End-User: Contract Sterilizers Capture Outsourcing Wave
Hospitals and surgery centers held 46.82% of 2025 revenue, driven by rules that mandate a BI for every implant load. Contract sterilizers, however, are growing 11.57% a year as device makers outsource validation to avoid eight-figure capital costs. They leverage bulk purchases that trim per-unit BI prices 20%–30%, yet their strict protocols drive higher absolute volumes, enlarging the steam and hydrogen peroxide biological indicators market share addressed by third-party processors.
Pharmaceutical and device manufacturers form the next largest user block, and they face tighter FDA rules requiring six consecutive successful BI challenges before parametric release, boosting consumption even after scale-up. Research labs remain niche but are adopting quarterly BI checks for BSL-2 and above.

Geography Analysis
North America represented 41.33% of global revenue in 2025, supported by FDA regulations classifying BIs as Class II devices and by more than 6,100 U.S. ambulatory surgery centers that must verify every implant cycle. Canadian provinces require four-hour turnaround, effectively mandating rapid-read systems, while Mexico’s burgeoning device exports spur demand for VHP indicators.
Asia-Pacific is on track for a 9.49% CAGR through 2031. China’s 14th Five-Year Plan funds 1,200 new tertiary hospitals, each needing autoclave fleets and indicators. India’s expanding surgical access and Japan’s aging population further lift volumes. Fragmented regulatory pathways across NMPA, CDSCO, and PMDA slow product launches, but the region’s sheer procedure growth outweighs delays.
Europe’s Medical Device Regulation requires exhaustive validation, prompting German manufacturers to adopt dual-modality protocols that double BI usage. The United Kingdom pilots centralized sterilization hubs to cut costs, while the GCC invests in state-of-the-art suites. South America remains cost-sensitive, with Brazil prioritizing low-cost strips and Argentina’s Terragene exporting competitively priced products.
Competitive Landscape
The analysis indicates moderate concentration within the steam and hydrogen peroxide biological indicators industry. STERIS’s February 2024 takeover of Getinge’s infection-control division broadened its portfolio into end-to-end bundles, deepening customer lock-in. Vertical integration lets incumbents capture margin from autoclaves, indicators, and software, while price-focused regional firms chip away at legacy spore-strip lines.
Mesa Laboratories invests in enzyme-linked substrates protected by multiple U.S. patents. Solventum, spun out from 3M in April 2024, now markets Attest as a standalone platform but faces margin pressure as Chinese generics undercut tenders in Southeast Asia. Digital-native startups pursue AI-driven traceability, yet hospitals hesitate to rip out established systems, limiting disruptive traction.
Steam And Hydrogen Peroxide Biological Indicators Industry Leaders
Getinge AB
STERIS plc
Terragene S.A.
Solventum
Mesa Laboratories
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- August 2025: Advanced Sterilization Products launched BIOTRACE Instant Read Steam, delivering seven-second results to cut sterile processing downtime.
- June 2025: Solventum introduced the Attest Super Rapid VHP Clear Challenge Pack, integrating BI and chemical indicators in one disposable unit.
- November 2024: FDA cleared Solventum’s Attest 1493 rapid BI with a 30-minute protocol, reinforcing rapid-read adoption momentum.
Global Steam And Hydrogen Peroxide Biological Indicators Market Report Scope
Biological Indicators (BIs) using Steam and Hydrogen Peroxide (H2O2) are defined as devices containing resilient bacterial spores, typically Geobacillus stearothermophilus, that are designed to validate the success of sterilization cycles. The death of these spores during the process confirms effective sterilization.
The Steam and Hydrogen Peroxide Biological Indicators Market Report is segmented by Product Type, Sterilization Method, End User, and Geography. By Product Type, the market is segmented into Self-Contained Biological Indicators, Spore Strips, Spore Suspension, and Rapid-Read Biological Indicators. By Sterilization Method, the market is segmented into Steam, Vaporized Hydrogen Peroxide, and Hybrid. By End User, the market is segmented into Hospitals & Surgical Centers, Pharmaceutical & Medical-Device Manufacturers, Contract Sterilization Service Providers, and Research & Academic Laboratories. By Geography, the market is segmented into North America, Europe, Asia-Pacific, Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends across major regions globally. Market Forecasts are Provided in Terms of Value (USD).
| Self-Contained Biological Indicators |
| Spore Strips |
| Spore Suspension |
| Rapid-Read Biological Indicators |
| Steam |
| Vaporized Hydrogen Peroxide (VHP) |
| Hybrid |
| Hospitals & Surgical Centers |
| Pharmaceutical & Medical-Device Manufacturers |
| Contract Sterilization Service Providers |
| Research & Academic Laboratories |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| France | |
| United Kingdom | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Self-Contained Biological Indicators | |
| Spore Strips | ||
| Spore Suspension | ||
| Rapid-Read Biological Indicators | ||
| By Sterilization Method | Steam | |
| Vaporized Hydrogen Peroxide (VHP) | ||
| Hybrid | ||
| By End-User | Hospitals & Surgical Centers | |
| Pharmaceutical & Medical-Device Manufacturers | ||
| Contract Sterilization Service Providers | ||
| Research & Academic Laboratories | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| France | ||
| United Kingdom | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large is the steam and hydrogen peroxide biological indicators market in 2026?
The market is valued at USD 0.846 billion in 2026 and is projected to rise to USD 1.218 billion by 2031.
What is the expected CAGR for steam and hydrogen peroxide indicators through 2031?
The compound annual growth rate is forecast at 7.54%.
Which product type is growing fastest?
Rapid-read biological indicators are advancing at a 10.35% CAGR, reflecting demand for quicker cycle validation.
Why is vaporized hydrogen peroxide gaining share?
Low-temperature sterilization suits complex polymer devices and single-use bioprocess bags, driving a 10.44% CAGR for VHP indicators.
Page last updated on:









