Smart Fertility Trackers Market Size and Share
Smart Fertility Trackers Market Analysis by Mordor Intelligence
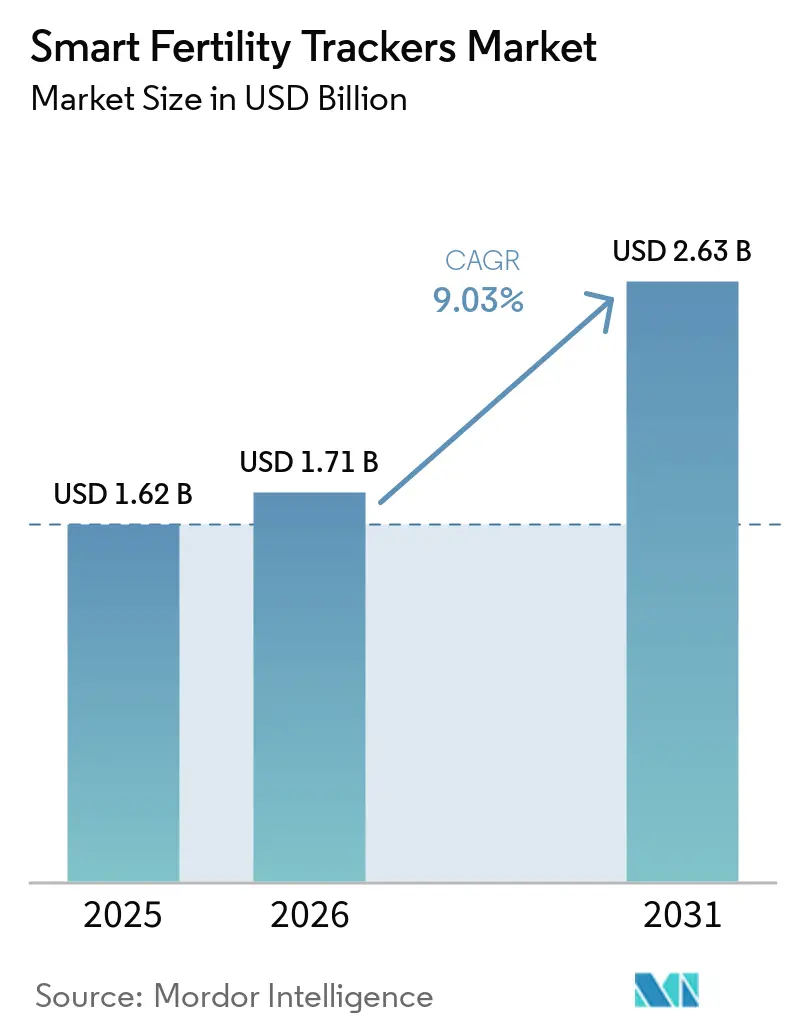
The Smart Fertility Trackers Market size is expected to increase from USD 1.62 billion in 2025 to USD 1.71 billion in 2026 and reach USD 2.63 billion by 2031, growing at a CAGR of 9.03% over 2026-2031.
Adoption strengthens as couples delay childbearing and seek accurate, non-invasive tools that fit daily routines. Platform strategies that link proprietary devices and apps with algorithms remain central, while open integrations with third-party wearables widen reach among existing smartwatch users and reduce hardware barriers[1]Garmin International, Inc., “Natural Cycles Integration Unlocks FDA-Cleared Birth Control App for Users With Select Garmin Smartwatches,” PR Newswire, prnewswire.com. Quantitative at-home hormone testing gains traction as new devices provide lab-like readouts in minutes and sync to apps that support coaching and care pathways. Reimbursement and employer benefits expansion supports uptake as digital contraceptives secure standardized billing codes that simplify claims processing and make coverage easier to administer. Privacy expectations and the uneven quality of wellness products shape purchase criteria, which in turn sustains demand for clinically validated features and clear data controls.
Key Report Takeaways
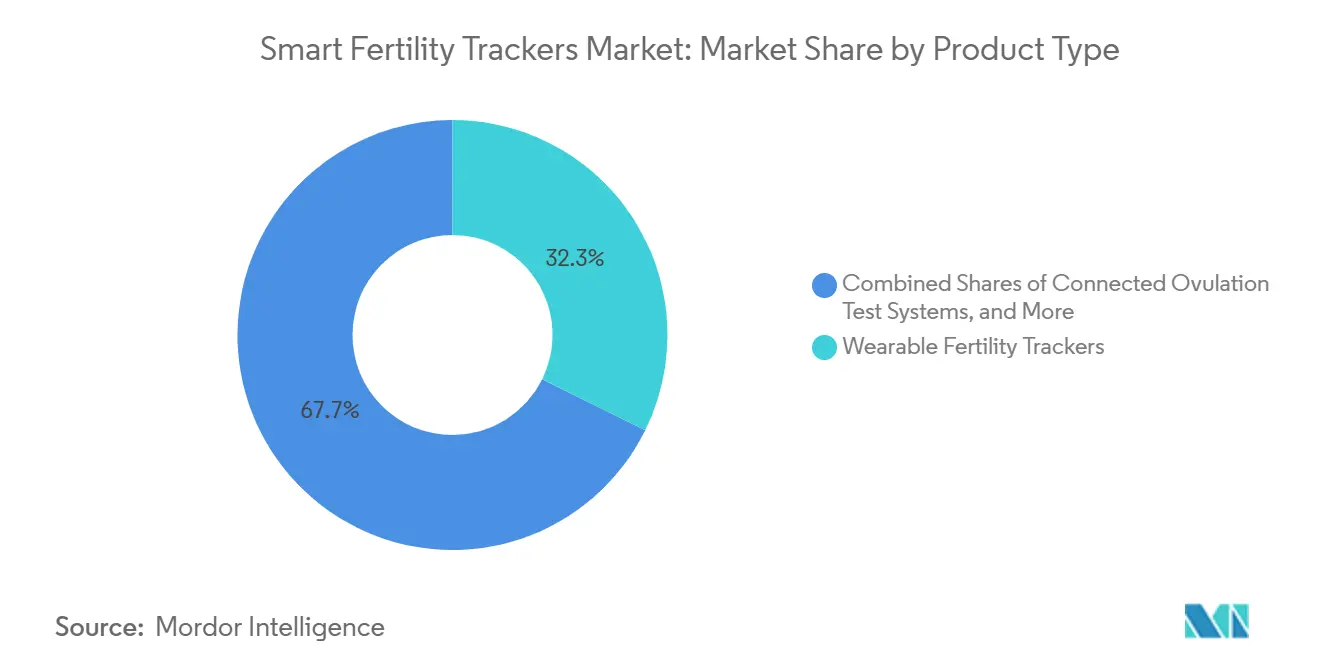
- By product type, wearable fertility trackers led with 32.34% revenue share in 2025, while at-home multi-hormone analyzers are projected to expand at an 11.32% CAGR through 2031.
- By biomarker, luteinizing hormone held a 35.67% share in 2025, and progesterone-metabolite tracking is forecast to grow at a 10.87% CAGR through 2031.
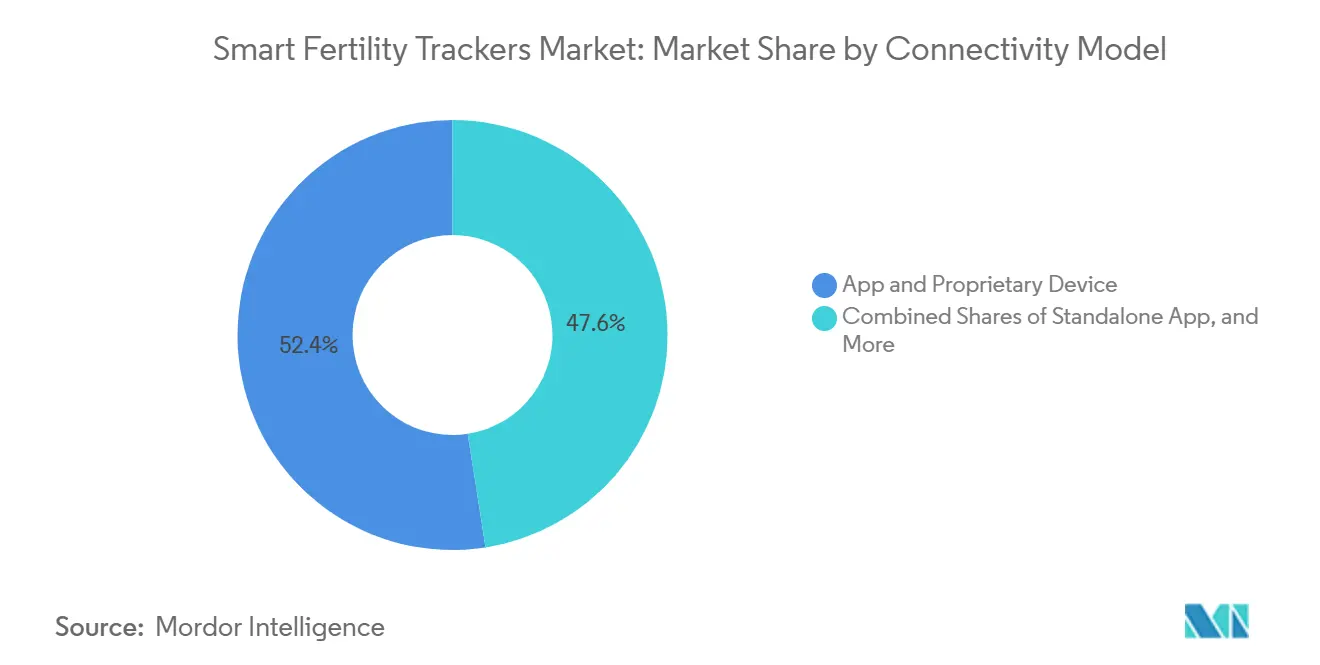
- By connectivity model, app-and-proprietary-device bundles captured a 52.45% share in 2025, while app-and-third-party-wearable integration is set to advance at an 11.46% CAGR through 2031.
- By end use, trying to conceive represented 47.89% of revenue in 2025, and natural contraception is the fastest-growing use case at a 12.12% CAGR through 2031.
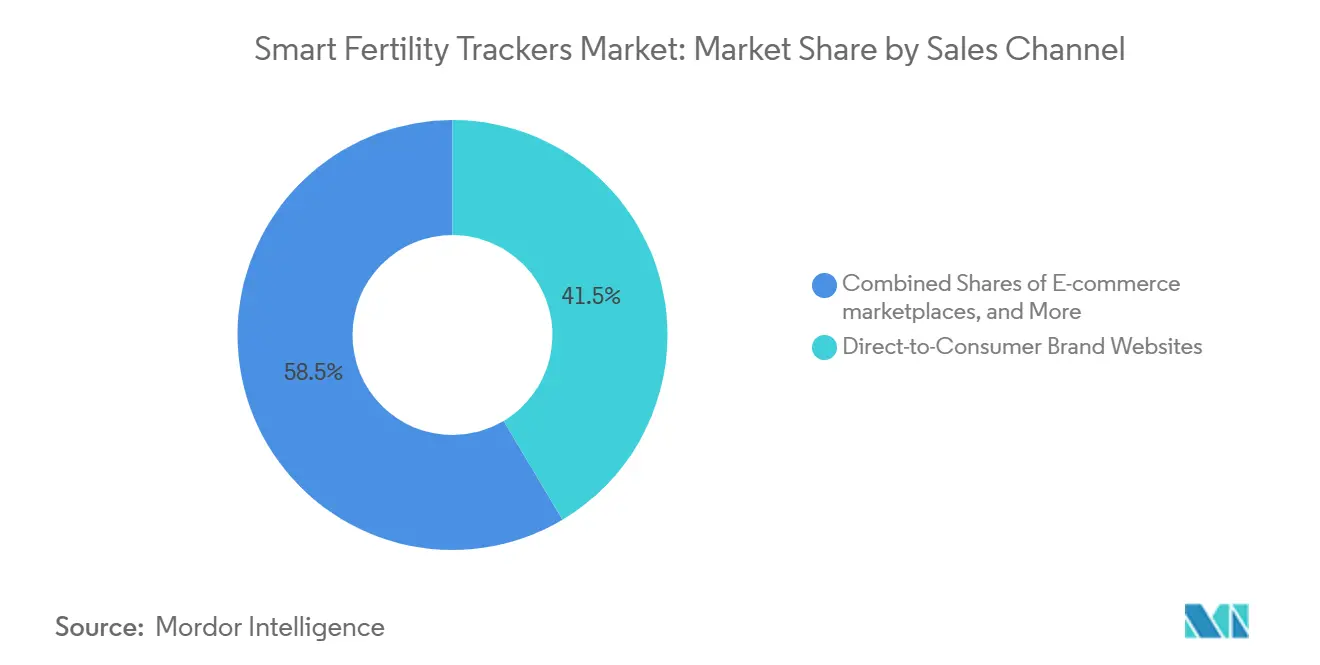
- By sales channel, direct-to-consumer brand websites accounted for a 41.45% share in 2025, and e-commerce marketplaces are on track for an 11.65% CAGR through 2031.
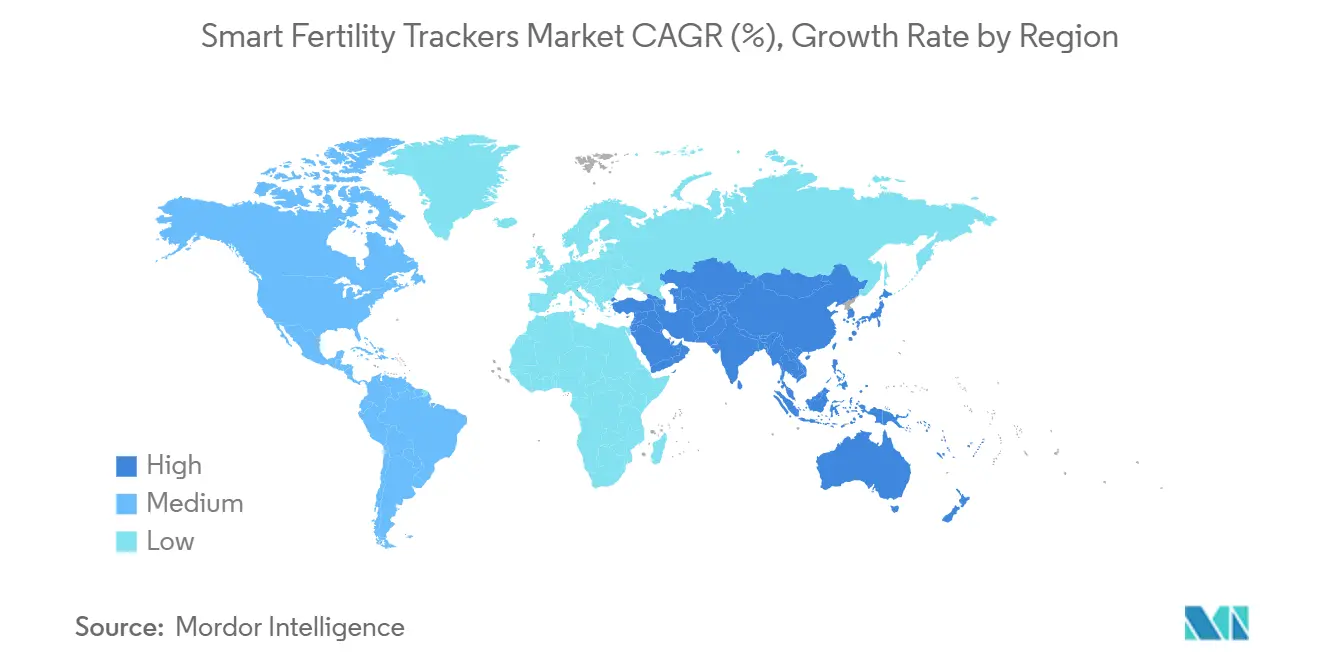
- By geography, North America commanded 43.45% of revenue in 2025, and Asia-Pacific is expected to be the fastest-growing region at a 12.82% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Smart Fertility Trackers Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising infertility prevalence and delayed parenthood | +2.1% | Global, concentrated in developed markets (North America, Western Europe, East Asia) | Medium term (2-4 years) |
| Rapid shift to at-home, app-connected fertility testing | +1.8% | Global, early gains in North America and Europe, spill-over to Asia-Pacific | Short term (≤ 2 years) |
| Wearable integrations enabling overnight temperature capture | +1.5% | North America, Europe, and affluent APAC markets (Japan, South Korea, Australia) | Medium term (2-4 years) |
| Employer fertility benefits and HSA/FSA eligibility broaden access | +1.3% | United States, Canada, early adoption in Northern Europe | Short term (≤ 2 years) |
| AI-powered multi-parameter tracking improves accuracy and usability | +1.2% | Global, with faster adoption in tech-forward regions (Silicon Valley, Shanghai, Tel Aviv) | Long term (≥ 4 years) |
| D2C and e-commerce scale global reach for smart trackers | +0.9% | Asia-Pacific core, spill-over to Latin America and MEA | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Infertility Prevalence And Delayed Parenthood
Wider use of digital tracking aligns with later family planning timelines, which increases the need to pinpoint fertile windows with greater certainty. In the smart fertility trackers market, regulatory progress and platform expansion help convert intent into adoption by reducing friction across devices and care pathways. Cross-wearable integrations extend access to clinically cleared contraceptive algorithms for smartwatch owners, creating a conversion funnel without added hardware purchases. At-home multi-hormone systems that generate numeric readouts promote more informed cycle management and allow users and clinicians to track trends over multiple cycles[2]Mira, “Mira Named CES Innovation Awards Honoree for At-Home Lab-Quality Hormone Testing,” PR Newswire, prnewswire.com. Standardized billing codes for digital contraception lower hurdles to reimbursement and employer coverage, which supports continued category growth as benefits teams look for cost-effective options.
Rapid Shift To At-Home, App-Connected Fertility Testing
The smart fertility trackers market continues to shift toward quantitative, at-home panels that deliver lab-like results within minutes. One example is a wireless multi-hormone reader that uses dual fluorescence and colorimetric sensing to produce numerical concentrations that correlate closely with blood serum and confirm ovulation with high precision, with results syncing directly to mobile apps[3]Team Inito, “Introducing Inito InSight Wireless Reader,” Inito Blog, inito.com. Recognition from major consumer technology forums further validates user demand for integrated systems that measure multiple hormones in a single session and fit within daily routines. App ecosystems increasingly combine device data with education and coaching, and they now link to broader wellness platforms, which support continuous engagement across cycle phases. As the smart fertility trackers market integrates hormones, biometrics, and software services, user expectations evolve toward clear, numeric insights and actionable guidance rather than simple calendar estimates.
Wearable Integrations Enabling Overnight Temperature Capture
Continuous overnight sensing removes the need for strict measurement routines and reduces user error, which makes it well-suited to daily life. In the smart fertility trackers market, watch and ring integrations connect passive temperature and related biometrics to cleared algorithms that provide fertility status and contraception guidance for users across supported regions. A 2026 systematic review reported that wearable digital technology achieved 88% pooled accuracy for detecting fertility windows across tens of thousands of cycles[4]Yue Shi et al., “The Diagnostic Accuracy of Wearable Digital Technology in Detecting Fertility Window and Menstrual Cycles,” npj Digital Medicine, nature.com, which outperformed self-reported basal body temperature and calendar-based methods. Ring-based systems that add more stable skin contact and personalized models also show strong cycle-detection performance in field use, including narrower ovulation-detection error margins than legacy methods. These developments help the smart fertility trackers market move from manual logs to automated, passive measurement linked to validated software.
Employer Fertility Benefits And HSA/FSA Eligibility Broaden Access
Coverage innovations make digital options easier to fund at scale, which supports steady adoption. A key milestone was a standardized HCPCS Level II code for a digital contraceptive, which streamlines claims handling and paths to coverage within employer plans and public programs. New entrants are also expanding care models around advanced diagnostics and automation, which adds complementary services and channels to reach users through clinical networks and payers. In the smart fertility trackers market, this benefits-led adoption mixes with direct-to-consumer channels and e-commerce to support hybrid growth that balances unit economics with user access. As more employers recognize the burden of out-of-pocket costs for fertility journeys, digital tools that offer measurable value and clear regulatory status stand to gain placement within benefits portfolios. The resulting subsidy effect directs more users toward clinically validated options and stabilizes demand.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Privacy/regulatory scrutiny of fertility apps and data handling | -0.8% | North America (especially post-Dobbs U.S. states), EU under GDPR | Short term (≤ 2 years) |
| Clinical validity limits of single-parameter approaches | -0.5% | Global | Medium term (2-4 years) |
| Subscription and device lock-in raise total cost of ownership | -0.4% | Global, particularly price-sensitive emerging markets | Medium term (2-4 years) |
| Platform/regulatory limits on contraceptive claims | -0.3% | United States (FDA 510(k) pathway), EU (MDR Class IIa/IIb) | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Privacy/Regulatory Scrutiny Of Fertility Apps And Data Handling
Data handling remains a visible concern for users who expect strong controls on reproductive health information. The smart fertility trackers market is sensitive to privacy signals, which influences trust and churn across paid and free offerings. Regional frameworks differ on consent and scope, so vendors that operate in multiple jurisdictions often design toward stricter baselines to avoid rework and build user confidence. Product claims also interact with oversight because contraception, diagnosis, and wellness have different regulatory definitions that determine evidence standards and marketing limits. These dynamics make clear disclosures and consent models an important part of product design, app onboarding, and customer support in 2026. Vendors that address privacy up front tend to see stronger retention and advocacy, which supports sustainable growth in the category.
Clinical Validity Limits Of Single-Parameter Approaches
Single-parameter methods can miss or misclassify fertile days, especially in irregular cycles, which reduces confidence in day-to-day decisions. In the smart fertility trackers market, multi-parameter fusion that combines hormones and biometrics has shown higher pooled accuracy than temperature-only approaches in peer-reviewed analyses. Device makers that quantify multiple hormones create trend lines that better reflect follicular and luteal dynamics, which helps users and clinicians avoid false positives and confirm cycle phases. New readers and cartridges that deliver numeric results in minutes support timely, app-based guidance without waiting for lab work. These features reduce ambiguity and align with professional use cases, which support continued migration away from single-signal kits. Over time, higher-accuracy systems reinforce the value proposition for paid subscriptions and premium devices.
Segment Analysis
By Product Type: Multi-Hormone Analyzers Surge as Quantitative Demand Rises
Wearable fertility trackers held a 32.34% share in 2025, reflecting strong user preference for passive, overnight sensing tied to cycle insights. The smart fertility trackers market continues to benefit from integrations that bring cleared fertility algorithms to popular smartwatch platforms, which convert existing device owners into app subscribers without requiring a new dedicated tracker purchase. At-home multi-hormone analyzers are projected to grow at an 11.32% CAGR through 2031 as users and clinicians prioritize numeric hormone data for more precise timing and cycle assessment. One platform recognized at a global consumer electronics forum highlighted at-home lab-quality testing for multiple hormones in a single 16-minute session, with early users reporting a stronger understanding of symptoms that map to their hormone curves. As quantitative systems mature, connected ovulation tests and connected basal thermometers keep a mid-tier position, although margin pressure rises as fully integrated solutions cut manual data entry and bundle insights with coaching.
The smart fertility trackers market also reflects growing diversity across devices and software. A new wireless reader introduced in 2026 uses dual fluorescence and colorimetric sensing to deliver high-precision hormone results that correlate well with venous assays, with built-in connectivity for seamless app sync. App-only options remain a lower-cost entry point for cycle tracking and education, yet they face higher churn when accuracy or personalization falls short of user expectations. Vendors that pair hardware with software strengthen recurring revenue via test-strip refills and premium subscriptions, while open wearable integrations offer a lower-cost path to scale by tapping into large installed bases. With clearer evidence and more robust user support, quantitative analyzers are positioned to lead premium adoption, while wearables define convenience at scale. These dynamics shape growth paths for both vertical bundles and open-platform strategies in the smart fertility trackers market.

By Biomarker: Progesterone Metabolite Tracking Gains Clinical Traction
Luteinizing hormone represented a 35.67% biomarker share in 2025, supported by its role as the most familiar signal for imminent ovulation. Progesterone-metabolite tracking is the fastest-growing biomarker with a 10.87% CAGR through 2031 as users and clinicians look to confirm ovulation and assess luteal sufficiency with quantitative signals. In the smart fertility trackers market, multi-hormone systems present a fuller picture by tracking estrogenic rise, LH surge, and post-ovulation progesterone changes as a single curve, which enables more targeted timing and clearer diagnostics. Readers that combine fluorescence and colorimetric imaging produce stable numeric baselines and trend lines that reduce interpretation errors common in yes or no strips. As more platforms link hormone results to algorithmic insights, users gain clearer, day-by-day guidance across the follicular and luteal phases.
Wearables that monitor temperature and related biometrics add value by passively capturing ovulatory patterns between at-home tests, and peer-reviewed analyses support higher pooled accuracy for multi-parameter approaches over temperature-only methods. In practice, the smart fertility trackers market is moving toward biomarker fusion, where hormone panels anchor confirmation and wearables fill gaps with continuous context. Vendors that present hormone and biometric data together in an app workflow make it easier for users to interpret signals and for clinicians to review longitudinal trends. This integrated approach helps reduce false positives and missed windows, which supports conversion to paid tiers. As quantitative accuracy and user experience improve, biomarker strategies that balance depth and convenience will continue to gain share in the smart fertility trackers market.
By Connectivity Model: Third-Party Wearable Integrations Unlock Installed Base
App-and-proprietary-device bundles captured a 52.45% share in 2025, reflecting the appeal of integrated ecosystems that align hardware, software, and algorithms. App-and-third-party-wearable integration is the fastest-growing model with an 11.46% CAGR through 2031 because it converts smartwatch owners into subscribers without new device purchases. In the smart fertility trackers market, cleared contraceptive algorithms that run on supported watch platforms extend functionality to millions of wearers across eligible regions, which broadens access and simplifies onboarding. Ring and band ecosystems add differentiated insights and accuracy through stable skin contact and personalized models, which further strengthen the open-integration thesis. Standalone apps remain a meaningful entry point, but face commoditization pressure as calendar tracking saturates and users expect validated performance.
Regulatory positioning and privacy practices shape adoption within each model. Vendors that pursue clinical validations for contraceptive use build a premium brand posture that can justify subscription pricing and support paths to reimbursement. Open integrations require robust data-sharing pipelines and consent handling that meet user expectations and regional rules. With growing support across watch and ring platforms, the smart fertility trackers market is likely to see continued outperformance from integrations that minimize hardware friction and maximize convenience. As these models mature, performance transparency and clinical claims will be key signals for users choosing between open and closed ecosystems.

By End Use: Natural Contraception Outpaces TTC Growth
Trying to conceive accounted for 47.89% of revenue in 2025 as delayed parenthood and cycle complexity drove demand for higher-accuracy tools. Natural contraception is projected to grow at a 12.12% CAGR through 2031 as users seek hormone-free options that blend daily guidance with clear evidence standards. In the smart fertility trackers market, regulatory momentum helps widen reach as a digital contraceptive gained a standardized billing code in 2024 and expanded smartwatch integrations in 2026, which reinforced both coverage and convenience. Health Canada’s 2024 approval opened access to over 10 million women, which added a sizable audience for non-hormonal options across Canada. As end users balance conception, contraception, and cycle health goals, integrated platforms that serve multiple needs within one app lower churn risk and extend lifetime value.
Cycle health tracking and insights form a steady use case for users managing PCOS, perimenopause, or general hormone literacy. Platforms that combine hormone panels with wearable data enable coaching that addresses sleep, recovery, and metabolic factors tied to cycle quality. For natural contraception, open-wearable models reduce hardware barriers by leveraging installed bases while keeping efficacy anchored in validated algorithms. TTC users benefit from numeric confirmation that shortens time to targeted attempts, while contraception users need clear daily status and audit-ready records for peace of mind. These distinct needs are converging within unified app experiences that guide users across goals, which supports durable growth in the smart fertility trackers market.
By Sales Channel: E-Commerce Marketplaces Capture Emerging-Market Share
Direct-to-consumer brand websites held a 41.45% share in 2025 because they enable full-funnel control, subscriptions, and higher-margin bundles. E-commerce marketplaces are set to expand at an 11.65% CAGR through 2031, supported by price transparency, high-velocity fulfillment, and wide discovery. Cross-border platforms provide verified-supplier programs and logistics assurance, which lower friction for manufacturers and streamline access for buyers across 190 or more countries. In the smart fertility trackers market, these dynamics make marketplaces attractive for emerging markets where shoppers compare brands across feature sets and total cost of ownership. Clinics and retail pharmacies continue to play a supporting role where professional guidance or point-of-care placement drives adoption.
Brand websites remain vital in premium segments because they support education-heavy journeys and complex bundles that need more context than a listing page can provide. Marketplaces are gaining share in entry and mid tiers where reviews and quick delivery outweigh longer education cycles. As marketplace policies evolve on wellness and contraceptive claims, vendors align messaging to regional rules while protecting category positioning. The smart fertility trackers market will likely see ongoing channel mixing where D2C supports premium users and marketplaces scale breadth through convenience and price.

Geography Analysis
North America holds a leading position with a 43.45% revenue share in 2025, supported by coverage progress and third-party wearable integrations that allow scale without new hardware. The smart fertility trackers market size reflects that base and continues to grow in 2026 as cleared contraceptive algorithms connect to watch platforms and gain billing pathways for simpler reimbursement. Canada’s 2024 approval extended access to a large audience for non-hormonal digital contraception, which pairs well with the region’s high smartphone and wearable penetration. In 2026, user expectations center on accuracy, privacy, and ecosystem fit, which favor platforms that provide numeric hormone data or validated biometric fusion. Clinic-linked models also emerge as complements where specialist guidance supports TTC users and complex cases.
Asia-Pacific is the fastest-growing region at a 12.82% CAGR through 2031, powered by mobile-first behavior and expanding marketplace infrastructure that makes cross-border fulfillment easier for consumers and sellers. The smart fertility trackers market gains from verified-supplier programs and logistics support that reduce import friction and elevate trust for first-time buyers. As price sensitivity remains a factor in many APAC markets, open wearable integrations provide a path to adoption using devices users already own. In 2026, vendors are tailoring SKUs, language packs, and education content for local contexts to raise conversion and retention. Hybrid strategies that combine marketplace reach with owned app communities are becoming standard as brands scale regionally.
Europe holds steady with emphasis on privacy-by-design and clinical clarity. The smart fertility trackers market share for companies that secure regional approvals and maintain transparent data practices is rising as users weigh accuracy claims alongside privacy expectations. Integrations that enable contraception and conception support across major watch ecosystems continue to raise awareness and lower setup friction. The Middle East and Africa, along with South America, are early in the adoption curve yet show meaningful momentum where marketplaces improve delivery reliability and customer support. In 2026, region-specific partnerships with clinics and women’s health providers are helping align products with local standards and care pathways, which supports durable expansion in the smart fertility trackers market.
Competitive Landscape
The smart fertility trackers market shows fragmented to moderate concentration, with leading vendors spanning device makers, app platforms, and open wearable ecosystems. Product differentiation centers on algorithmic validation, multi-hormone quantification, and user experience that reduces friction across measurement, interpretation, and action. Natural Cycles extended its FDA-cleared algorithm to select Garmin smartwatches in 2026, connecting overnight temperature signals to fertility status and contraception guidance across multiple countries. Oura introduced a proprietary AI model in 2026 to deliver personalized women’s health guidance by blending biometric signals with curated medical research and clinician input. Mira’s multi-hormone platform earned recognition for at-home lab-quality testing and later integrated ring data to connect sleep, readiness, and temperature with quantitative hormone profiles.
New entrants are pushing sensing boundaries to reduce consumables and manual steps. OTO Fertility’s Cira platform couples a wrist-worn biosensor that tracks dozens of biometric markers with a clinician-facing system to deliver predictive insights and is available through partner clinics in North America. FertilAI secured the first CE mark under EU MDR for predictive AI aimed at ovulation prediction and IVF trigger timing and embedded those capabilities into a SaaS platform trained on a large cycle dataset. An at-home reader launched in 2026 moves quantitative sensing to the point of need with high correlation to blood assays and built-in connectivity for fast interpretation. These advances help vendors translate clinical signals into daily guidance, which improves retention and word-of-mouth.
Smart Fertility Trackers Industry Leaders
SPD Swiss Precision Diagnostics
NaturalCycles Nordic AB
Mira
Premom (Easy@Home)
Femometer
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2026: FertilAI secured the world’s first CE mark under EU MDR for predictive AI in fertility treatment cycle optimization, covering both IVF trigger timing and natural-cycle ovulation prediction.
- March 2026: Garmin enabled Natural Cycles integration on select smartwatches, expanding FDA-cleared birth control and fertility insights to supported regions including Australia, Brazil, Canada, the EU, Singapore, Switzerland, the UK, and the US.
Global Smart Fertility Trackers Market Report Scope
The smart fertility trackers market comprises digital devices, connected diagnostics, and software applications designed to monitor physiological and hormonal indicators of the menstrual cycle to support fertility planning, contraception, and reproductive health insights. These solutions leverage biomarkers such as hormones and temperature, combined with data analytics and mobile connectivity, to provide personalized predictions and guidance.
The smart fertility trackers market is segmented by product type including wearable fertility trackers, connected ovulation test systems, connected basal thermometers, at-home multi-hormone analyzers, and app-only fertility or contraceptive solutions; by biomarker including luteinizing hormone, estrogen metabolites, progesterone metabolite, basal or skin temperature, and others; by connectivity model including app with proprietary device, app with third-party wearable integration, and standalone app; by end use including trying to conceive, natural contraception, and cycle health tracking and insights; by sales channel including direct-to-consumer brand websites, e-commerce marketplaces, pharmacies and retail, and clinics; and by geography including North America, Europe, Asia-Pacific, the Middle East and Africa, and South America, with market forecasts provided in terms of value in USD. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally.
| Wearable Fertility Trackers |
| Connected Ovulation Test Systems |
| Connected Basal Thermometers |
| At-home Multi-hormone Analyzers |
| App-only Fertility/Contraceptive Solutions |
| Luteinizing Hormone |
| Estrogen Metabolites |
| Progesterone Metabolite |
| Basal/Skin Temperature |
| Others (HRV and respiratory rate, Multi-parameter fusion) |
| App & Proprietary Device |
| App & Third-party Wearable Integration |
| Standalone App |
| Trying to conceive (TTC) |
| Natural contraception |
| Cycle Health Tracking and Insights |
| Direct-to-Consumer Brand Websites |
| E-commerce marketplaces |
| Pharmacies and Retail |
| Clinics |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Wearable Fertility Trackers | |
| Connected Ovulation Test Systems | ||
| Connected Basal Thermometers | ||
| At-home Multi-hormone Analyzers | ||
| App-only Fertility/Contraceptive Solutions | ||
| By Biomarker | Luteinizing Hormone | |
| Estrogen Metabolites | ||
| Progesterone Metabolite | ||
| Basal/Skin Temperature | ||
| Others (HRV and respiratory rate, Multi-parameter fusion) | ||
| By Connectivity Model | App & Proprietary Device | |
| App & Third-party Wearable Integration | ||
| Standalone App | ||
| By End Use | Trying to conceive (TTC) | |
| Natural contraception | ||
| Cycle Health Tracking and Insights | ||
| By Sales Channel | Direct-to-Consumer Brand Websites | |
| E-commerce marketplaces | ||
| Pharmacies and Retail | ||
| Clinics | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the outlook for the smart fertility trackers market through 2031?
The smart fertility trackers market size is set to grow from USD 1.71 billion in 2026 to USD 2.63 billion by 2031 at an 9.03% CAGR, supported by cross-wearable integrations, quantitative at-home testing, and expanding coverage pathways.
Which regions will lead growth in smart fertility tracking over the next five years?
Asia-Pacific is the fastest-growing region at a 12.82% CAGR through 2031, while North America retains the largest revenue share based on strong device penetration and regulatory clarity.
Which product categories are gaining the most traction in smart fertility tracking?
At-home multi-hormone analyzers are the fastest-growing at an 11.32% CAGR through 2031, and wearable trackers held the highest share in 2025, reflecting user preference for passive sensing and numeric insights.
How are integrations with third-party wearables changing adoption?
App-and-third-party-wearable integrations are growing at an 11.46% CAGR through 2031, by tapping large installed bases of watches and rings, which reduces hardware barriers and speeds onboarding for cleared algorithms.
What end uses are expanding fastest in 2026?
Natural contraception is the fastest-growing end use at a 12.12% CAGR through 2031, while trying to conceive remains the largest use case given delayed parenthood trends and demand for more accurate timing.
Which sales channels matter most for category expansion?
Direct-to-consumer brand websites maintain a leading share for premium bundles, while e-commerce marketplaces are growing at an 11.65% CAGR through 2031, and are critical for scaling in emerging markets.
Page last updated on: