Rheumatology Therapeutics Market Size and Share
Rheumatology Therapeutics Market Analysis by Mordor Intelligence
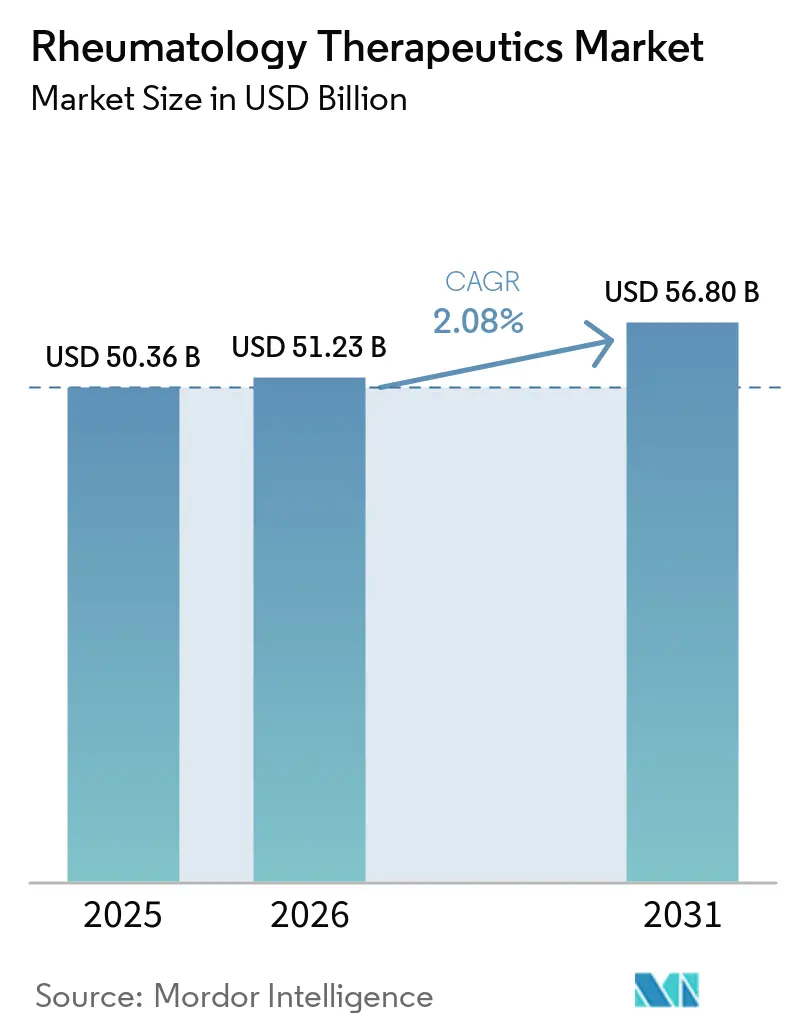
The Rheumatology Therapeutics Market size is expected to increase from USD 50.36 billion in 2025 to USD 51.23 billion in 2026 and reach USD 56.80 billion by 2031, growing at a CAGR of 2.08% over 2026-2031.
Aging populations, wider autoimmune screening, and payer-backed treat-to-target protocols enlarge the treatment pool, yet net growth remains muted because biosimilar-driven price compression and boxed warnings on JAK inhibitors offset rising volume. Interchangeability rulings for adalimumab biosimilars now enable pharmacy-level substitution, accelerating share gains for low-cost competitors and squeezing originators’ margins. Meanwhile, subcutaneous autoinjectors and on-body pumps are steering patients away from infusion centers, reinforcing home-based care incentives set by U.S. payers that trim site-of-care costs by up to 50%. Manufacturers are pivoting to indication expansion, device innovation, and patient support programs to defend value as net prices erode.
Key Report Takeaways
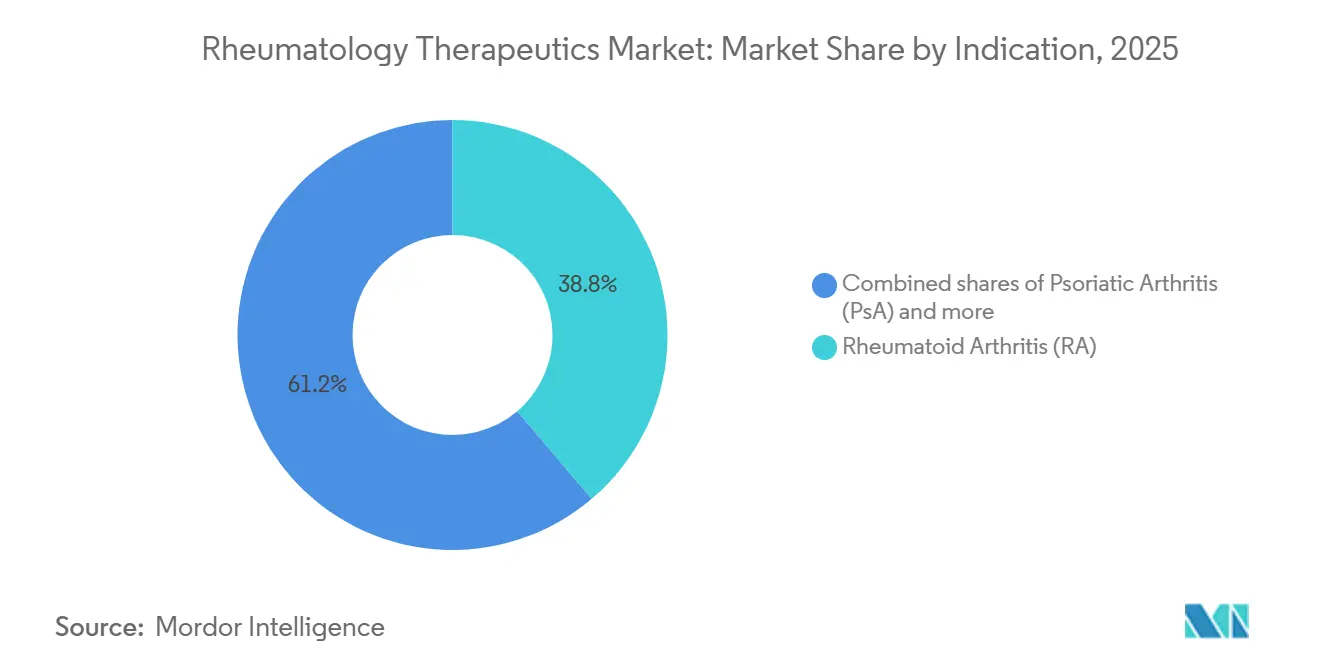
- By indication, Rheumatoid Arthritis (RA) accounted for 38.78% of the rheumatology therapeutics market share in 2025, and Psoriatic Arthritis (PsA) is set to grow at a 4.12% CAGR through 2031.
- By drug class, conventional synthetic DMARDs accounted for 20.34% of the rheumatology therapeutics market share in 2025, and Targeted Synthetic DMARDs (tsDMARDs) are set to grow at a 3.45% CAGR through 2031.
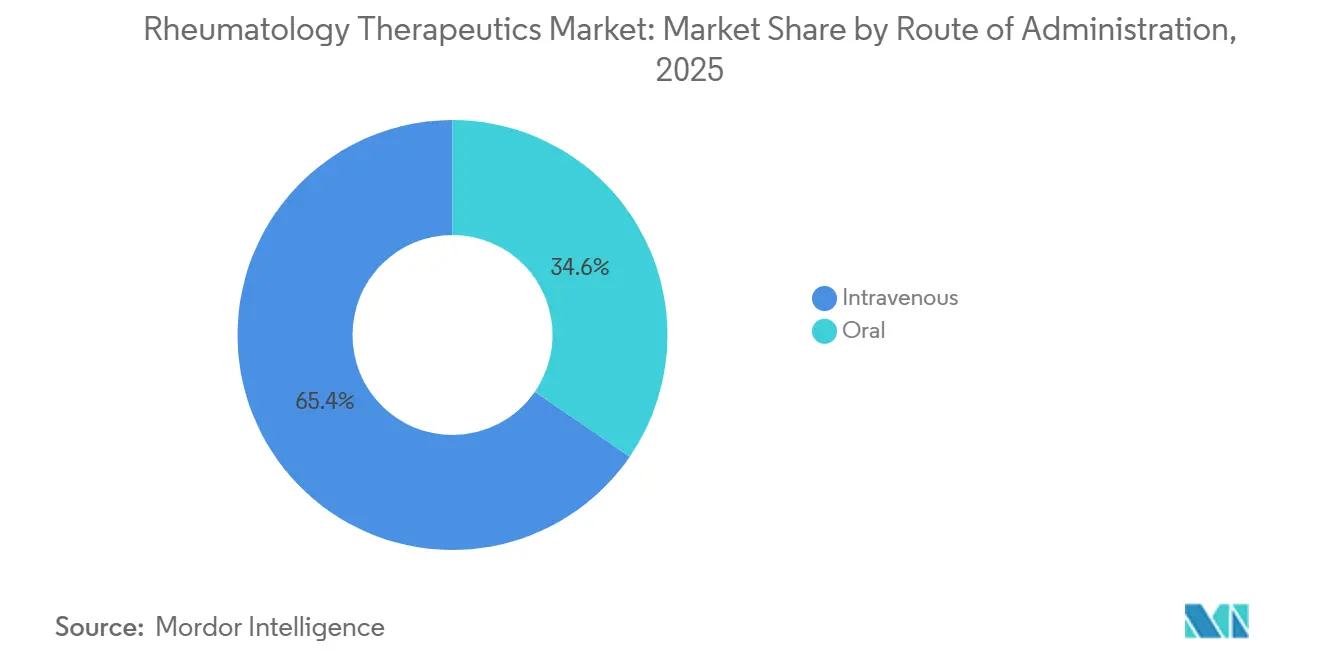
- By route of administration, intravenous captured 65.41% of the rheumatology therapeutics market in 2025, while the oral segment is the fastest-growing, with a 4.09% CAGR.
- By distribution channel, hospital pharmacies accounted for 48.91% of the rheumatology therapeutics market, and retail pharmacies are the fastest-growing, with a 4.65% CAGR.
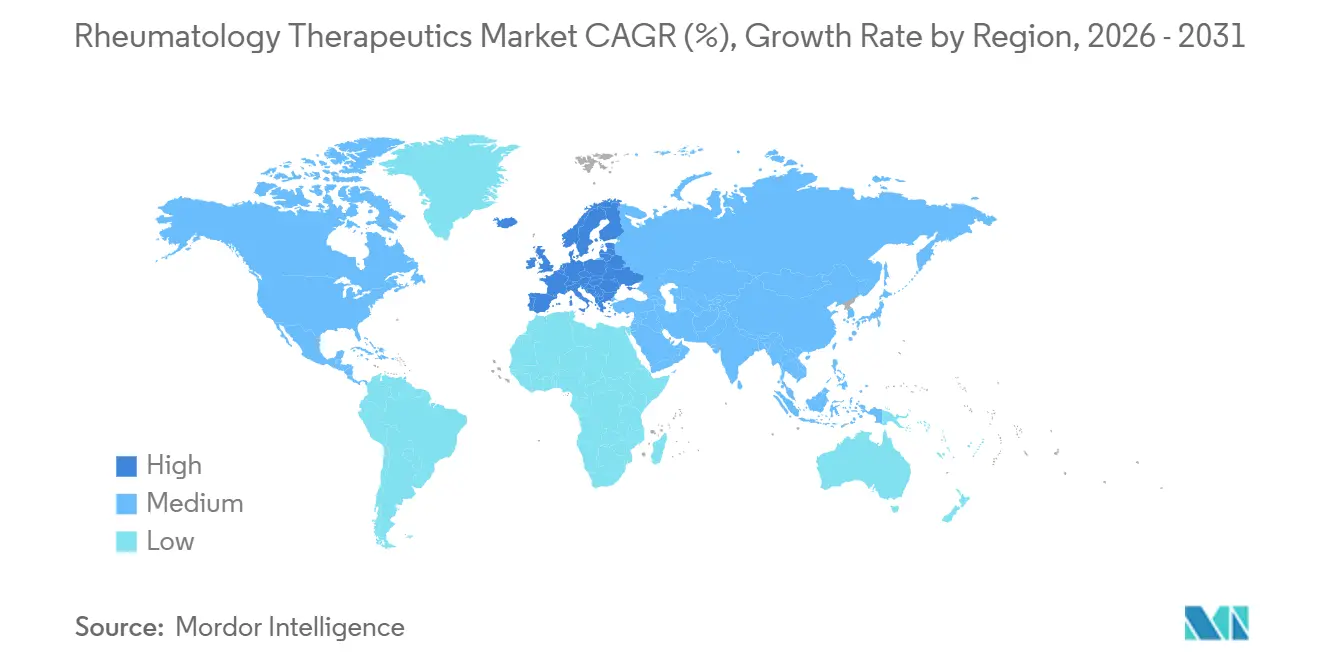
- By geography, North America led with a 39.34% value share in 2025; Europe is expected to deliver the highest regional CAGR of 4.34% through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Rheumatology Therapeutics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising autoimmune disease burden and aging patient populations | +0.5% | Global, highest absolute growth in North America and Europe | Long term (≥ 4 years) |
| Accelerating adoption of biologics and biosimilars in rheumatology | +0.4% | Global, led by North America and Europe; APAC catching up | Medium term (2-4 years) |
| Expansion of targeted small molecules (JAK/TYK2) | +0.3% | North America, Europe; selective uptake in APAC | Medium term (2-4 years) |
| Favorable reimbursement and guideline alignment for treat-to-target | +0.3% | North America, Europe, Japan | Short term (≤ 2 years) |
| Interchangeability designations boost U.S. biosimilar uptake | +0.2% | United States | Short term (≤ 2 years) |
| Shift to home/self-administration via SC devices | +0.2% | North America, Europe, urban APAC | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Autoimmune Disease Burden and Aging Patient Populations
Incidence of rheumatoid arthritis climbs steeply beyond age 50; concomitant surges in psoriatic arthritis and axial spondyloarthritis reflect better imaging and biomarker use. The World Health Organization projects a global cohort of 2.1 billion people aged 60 plus by 2050, doubling 2020 levels [1]World Health Organization, “Ageing and Health,” who.int. Although this enlarges the eligible pool, older patients accumulate cardiovascular and malignancy risks that limit JAK inhibitor prescribing under FDA boxed warnings, pushing clinicians toward csDMARDs and lower-intensity regimens. Payers reinforce this caution by mandating methotrexate failure before biologic approval, extending time to optimal disease control. These constraints temper uptake of premium therapy even as the population base expands.
Accelerating Adoption of Biologics and Biosimilars in Rheumatology
Biosimilar interchangeability has disrupted pricing. Simlandi was the first to receive an adalimumab interchangeability label in February 2024, followed by Hadlima and Yuflyma in 2025, enabling pharmacist substitution without prescriber consent [2]U.S. Food and Drug Administration, “Biosimilar and Interchangeable Biologics,” fda.gov. Evernorth data show that Humira biosimilars accounted for a significant share of adalimumab prescriptions by Q4 2024 and overtook the originator in Q1 2025. Net prices have fallen significantly, broadening access but compressing margins. FDA draft guidance (October 2025) proposes waiving comparative efficacy trials when analytical similarity suffices, potentially cutting biosimilar development timelines in half, a boon for integrated developers such as Samsung Bioepis and Celltrion.
Expansion of Targeted Small Molecules (JAK/TYK2) Across RA, PsA, axSpA
Oral JAK options offer convenience over injectables, yet class-wide safety warnings limit uptake. Pfizer discontinued new indications for Xeljanz after cardiovascular and malignancy signals, while AbbVie’s Rinvoq lifted Q3 2024 sales 46% to USD 1.8 billion but still carries the same boxed warning. Bristol-Myers Squibb’s TYK2 agent deucravacitinib bypasses some JAK-class risks and is in Phase 3 for psoriatic arthritis, positioning it as a differentiated oral entrant. Step-therapy rules that require biologic failure before JAK use slow penetration despite patient preference for pills.
Favorable Reimbursement and Clinical Guideline Alignment for Treat-to-Target
ACR and EULAR guidelines endorse rapid escalation until remission. U.S. insurers translate this into biosimilar-first policies; CVS Caremark, Cigna, and UnitedHealthcare instituted biosimilar step therapy across TNF and IL inhibitors during 2024-2025 [3]CVS Health, “Specialty Pharmacy Trends 2025,” cvshealth.com. The Inflation Reduction Act introduces Medicare price negotiations from 2026 and caps Part D out-of-pocket spending at USD 2,000 from 2025, shifting cost burdens onto plans and sharpening scrutiny of high-priced innovators. In Europe, France’s 2025 biosimilar action plan targets 80% penetration for adalimumab and etanercept by 2027, reinforcing similar dynamics.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Price erosion and payer pressure from rapid biosimilar diffusion | -0.3% | North America, Europe | Short term (≤ 2 years) |
| Safety headwinds for JAK class (boxed warnings, label restrictions) | -0.3% | Global, most acute in North America and Europe | Medium term (2-4 years) |
| High out-of-pocket costs and utilization management (step therapy, accumulators) | -0.2% | United States, with spillover to commercial plans in Canada | Short term (≤ 2 years) |
| Channel consolidation into PBM-owned specialty networks limits choice | -0.2% | United States, emerging in select European markets | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Price Erosion and Payer Pressure from Rapid Biosimilar Diffusion
Humira biosimilars achieved high prescription share by Q4 2024, overtaking the originator a quarter later as PBMs mandated biosimilar-first protocols. Comparable erosion looms for tocilizumab and etanercept as new biosimilars secure FDA and EMA green lights in 2025-2026. Originators counter with deeper rebates and copay assistance, but net prices keep sliding, weighing on topline growth in high-income markets.
Safety Headwinds for JAK Class (Boxed Warnings, Label Restrictions)
Safety warnings for the JAK family of drugs have reshaped both prescribing and coverage rules. After the ORAL Surveillance trial linked JAK inhibitors to higher rates of heart events, cancer, and blood clots in rheumatoid-arthritis patients aged 50 and older, the Food and Drug Administration added boxed warnings to all approved agents, and payers quickly tightened utilization controls. Pfizer then stopped pursuing new uses for tofacitinib (Xeljanz) and let the patent lapse in 2024, effectively conceding much of the oral disease-modifying market to rivals.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Indication: PsA Expansion Outpaces RA Maturity
Rheumatoid arthritis retained 38.78% of the rheumatology therapeutics market share in 2025, yet its margin has narrowed as adalimumab biosimilars won a significant share of U.S. prescriptions by Q4 2024 and the majority in China during 2024, compressing originator pricing. Psoriatic arthritis accounts for a smaller share today, but it is forecast to grow at a 4.12% CAGR to 2031—more than double the rate of mature RA, making it the fastest-advancing indication within the rheumatology therapeutics market. AbbVie’s Skyrizi secured U.S. approval for psoriatic arthritis in June 2024 and posted USD 3.2 billion in Q3 2024 revenue, up significantly year-over-year, confirming robust demand for IL-23 inhibition across joint and skin disease.

By Drug Class: Targeted Synthetics Gain Despite Safety Scrutiny
Conventional synthetic DMARDs accounted for 20.34% of the rheumatology therapeutics market share in 2025, as guidelines still require methotrexate failure before escalating therapy. Methotrexate costs less than USD 20 per month, sustaining high volumes, though 30-40% of patients fail to meet response goals—so it keeps a durable floor under the rheumatology therapeutics market. Targeted synthetics, led by JAK inhibitors, are projected to expand at a 3.45% CAGR to 2031, the fastest among drug classes, yet boxed warnings on cardiovascular and malignancy risks limit use to younger, lower-risk patients. AbbVie’s Rinvoq generated USD 1.8 billion in Q3 2024, a 46% surge that underlines demand for convenient oral options across RA, PsA, and axial spondyloarthritis despite payer step therapy. Pfizer withdrew new-indication work on tofacitinib after patent expiry in 2024, effectively conceding the oral DMARD lane to newer rivals.
By Route of Administration: Oral Convenience Challenges IV Incumbency
Intravenous formulations accounted for 65.41% of the rheumatology therapeutics market in 2025. Oral DMARDs—methotrexate, leflunomide, and JAK or TYK2 inhibitors—are forecast to advance at 4.09% CAGR through 2031, propelled by younger cohorts who prefer pills and by growing reimbursement for outpatient telehealth follow-up. Subcutaneous biologics and newer oral agents are gaining favor because they eliminate infusion-center fees that U.S. insurers peg at 30-50% of total episode cost. AbbVie rolled out a citrate-free Skyrizi and funds autoinjector upgrades to reduce injection-site pain, while on-body pumps for tocilizumab and rituximab entered trials to deliver high volumes without clinic visits.

By Distribution Channel : Retail Gains as Specialty Shifts Outpatient
Hospital pharmacies held 48.91% of the rheumatology therapeutics market share in 2025, leveraging embedded infusion services and integrated-delivery procurement, yet retail pharmacies are expected to advance at 4.65% CAGR through 2031. Payer consolidation under CVS Caremark, Cigna’s Accredo, and UnitedHealth OptumRx couples formulary power with owned dispensing, steering subcutaneous and oral fills toward community or mail channels via lower cost-sharing. The 2025 Medicare Part D redesign, capping patient liability at USD 2,000, intensifies plan focus on net costs and nudges volumes to the lowest-fee channels. Specialty pharmacy remains indispensable for cold-chain biologics and complex adherence support, yet manufacturers are building direct patient services that duplicate these functions and undercut dispensing markups. Online pharmacies gain ground where e-commerce logistics are mature—India’s Biocon and Sun Pharma now ship biosimilars direct to chronic users—while hospital outlets defend turf through bundled-payment contracts and clinical-integration arguments.
Geography Analysis
North America led the rheumatology therapeutics market in 2025 with a 39.34% share, but interchangeability rulings for adalimumab biosimilars and looming Medicare price negotiations are expected to cap future growth. The 2025 Part D redesign places a USD 2,000 ceiling on patient out-of-pocket costs, shifting more liability to plans and stimulating tougher formulary management. Canada’s bulk-buying alliance and Mexico’s public insurers echo this down-pricing trend, compelling manufacturers to seek volume via new indications rather than price lifts. Canada’s pan-Canadian Pharmaceutical Alliance and Mexico’s IMSS and ISSSTE supplement the region’s volume by jointly negotiating biologic prices to ease budget pressure.
Europe is projected to expand at a 4.34% CAGR through 2031, the fastest among all regions, as national health systems embed mandatory switching targets and streamline approvals. Germany’s AMNOG framework aligns prices with assessed clinical benefit; France’s 2025 plan rewards prescribers who meet an 80% biosimilar quota for adalimumab and etanercept; and the United Kingdom’s cost-per-QALY threshold consistently steers payers toward lower-priced options.
Asia-Pacific shows the next wave of expansion, propelled by China’s dual-catalog National Reimbursement Drug List that cut average prices 63% for 114 drugs while preserving premium pricing for 19 breakthrough therapies starting January 2026. Domestic manufacturers already control most Chinese volume, with adalimumab biosimilars capturing majority share in 2024, tocilizumab, and infliximab held notable volume as list prices fall but margins stay positive. Japan’s National Health Insurance offers a JPY 1,500 (USD 10) dispensing bonus for select biosimilars, a policy that produced a 13.48% one-time jump in etanercept uptake and now extends to ustekinumab after its May 2025 listing. India’s market was valued at INR 437 crore (USD 51 million) in 2025 and is set for a 14.2% CAGR, underpinned by the USD 1.1 billion Biopharma SHAKTI program that scales low-cost manufacturing for export to regulated market.
Middle East & Africa and South America expand more slowly because reimbursement gaps and currency volatility curb multinational investment. Gulf Cooperation Council states are building specialty pharmacy networks and using volume-based contracts to narrow price spreads, while South Africa’s tender system awards business almost exclusively to the lowest bidder, putting biosimilars in pole position. Brazil’s public system limits biologic access mainly to wealthier patients with private insurance, creating a two-tier market, and Argentina’s currency slide in 2024 forces repeated price renegotiations that deter long-term capacity commitments.
Competitive Landscape
The rheumatology therapeutics market shows moderate concentration. AbbVie, Johnson & Johnson, Amgen, and Pfizer maintain leading positions through broad portfolios of TNF, IL, and JAK assets, yet the first wave of biosimilars from Samsung Bioepis, Celltrion, and Biocon has shaved originator shares to less than half in certain molecules. AbbVie is defending value via new indications—Skyrizi won U.S. approval for psoriatic arthritis in June 2024 and patient-friendly devices, while Rinvoq continues to penetrate multiple inflammatory conditions. Biosimilar specialists leverage process analytical technology to drop the cost of goods by 40%, enabling list prices 20-30% below originators even after steep rebates.
Technology and channel dynamics are reshaping power. PBM consolidation into vertically integrated networks gives CVS Caremark, Cigna, and UnitedHealthcare outsized formulary sway, pressuring manufacturers to trade rebates for tier placement. Innovators respond with on-body delivery platforms and real-world evidence packages that highlight the advantages of adherence and outcomes over copycats. Emerging entrants such as Fresenius Kabi gain ground through rapid filings that exploit FDA’s streamlined biosimilar guidance, exemplified by the Avtozma tocilizumab launch in January 2025. The playing field, therefore, balances scale advantages with agile low-cost challengers, fostering price competition but also innovation around patient experience.
Rheumatology Therapeutics Industry Leaders
AbbVie Inc.
Johnson & Johnson
Amgen Inc.
Pfizer Inc
Samsung Bioepis
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: The EMA issued a positive opinion for Fubelv for the treatment of rheumatoid arthritis, an etanercept biosimilar by Fresenius Kabi, foreshadowing EU market entry.
- May 2025: Yuflyma received U.S. interchangeability status for indications including rheumatoid arthritis, Crohn’s disease, and psoriasis, accelerating adalimumab biosimilar substitution.
- May 2025: Japan’s NHI added ustekinumab biosimilars to its price list for treating conditions like plaque psoriasis and psoriatic arthritis, broadening access to subsidies.
Global Rheumatology Therapeutics Market Report Scope
As per the scope of the report, rheumatology therapeutics encompasses a broad spectrum of medical strategies designed to manage chronic inflammatory and autoimmune conditions, such as rheumatoid arthritis (RA), lupus, and gout.
The rheumatology therapeutics market is segmented by indication, drug class, route of administration, distribution channel, and geography. Based on the indication, the market is segmented into rheumatoid arthritis, psoriatic arthritis, axial spondyloarthritis, systemic lupus erythematosus, gout, Sjögren’s syndrome, systemic sclerosis, vasculitides, and juvenile idiopathic arthritis. Based on drug class, the market is segmented into conventional synthetic DMARDs (csDMARDs), biologic DMARDs (bDMARDs), targeted synthetic DMARDs (tsDMARDs), and symptomatic agents. By route of administration, the market is segmented into oral and intravenous. By distribution channel, the market is divided into hospital pharmacies, retail pharmacies, and others.
Geographically, the market is segmented into North America, Europe, Asia-Pacific, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Rheumatoid Arthritis (RA) |
| Psoriatic Arthritis (PsA) |
| Axial Spondyloarthritis (incl. Ankylosing Spondylitis) |
| Systemic Lupus Erythematosus (SLE) |
| Gout |
| Sjögren’s Syndrome |
| Systemic Sclerosis (Scleroderma) |
| Vasculitides (e.g., GPA/MPA) |
| Juvenile Idiopathic Arthritis (JIA) |
| Conventional Synthetic DMARDs (csDMARDs) |
| Biologic DMARDs (bDMARDs) |
| Targeted Synthetic DMARDs (tsDMARDs) |
| Symptomatic Agents |
| Oral |
| Intravenous |
| Hospital Pharmacies |
| Retail Pharmacies |
| Others |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Indication | Rheumatoid Arthritis (RA) | |
| Psoriatic Arthritis (PsA) | ||
| Axial Spondyloarthritis (incl. Ankylosing Spondylitis) | ||
| Systemic Lupus Erythematosus (SLE) | ||
| Gout | ||
| Sjögren’s Syndrome | ||
| Systemic Sclerosis (Scleroderma) | ||
| Vasculitides (e.g., GPA/MPA) | ||
| Juvenile Idiopathic Arthritis (JIA) | ||
| By Drug Class | Conventional Synthetic DMARDs (csDMARDs) | |
| Biologic DMARDs (bDMARDs) | ||
| Targeted Synthetic DMARDs (tsDMARDs) | ||
| Symptomatic Agents | ||
| By Route of Administration | Oral | |
| Intravenous | ||
| By Distribution Channel | Hospital Pharmacies | |
| Retail Pharmacies | ||
| Others | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large will the rheumatology therapeutics market be by 2031?
It is forecast to reach USD 56.80 billion in 2031, up from USD 51.23 billion in 2026.
Which region is growing fastest in rheumatology therapeutics?
Europe posts the highest CAGR at 4.34% through 2031.
What drug classes dominate current spending?
Biologic DMARDs account for the largest spend, although conventional synthetic DMARDs still hold 20.34% share because they remain first-line.
How are biosimilars affecting pricing?
Interchangeability labels have driven U.S. net prices down as much as 80% versus originators, with similar pressure emerging in Europe.
Page last updated on:







