Retractable Needles Market Size and Share
Retractable Needles Market Analysis by Mordor Intelligence
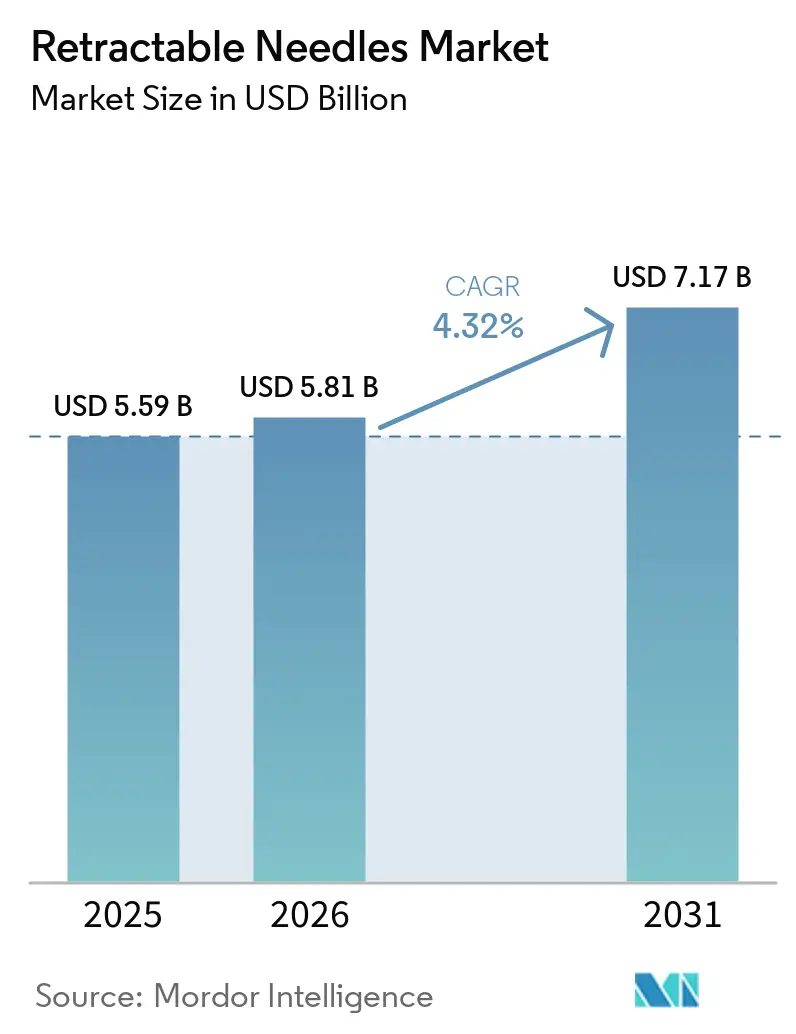
The Retractable Needles Market size is expected to increase from USD 5.59 billion in 2025 to USD 5.81 billion in 2026 and reach USD 7.17 billion by 2031, growing at a CAGR of 4.32% over 2026-2031.
Growth reflects a structural shift in purchasing behavior as hospital systems embed stringent injury-prevention clauses into group-purchasing contracts, while OSHA’s January 2025 penalty increase has made safety compliance a direct balance-sheet concern [1]U.S. Department of Labor, “OSHA Penalties,” OSHA, osha.gov. Mass immunization programs and surging GLP-1 volumes are intensifying syringe demand, and passive retraction technology is displacing manual mechanisms because it removes clinician-activation errors. At the same time, tariff pressures and reshoring costs are squeezing suppliers, accelerating investments in North American capacity but raising unit prices that challenge price-sensitive buyers. Needle-free and on-body delivery systems represent plausible substitutes, yet their cost and regulatory hurdles keep them secondary to mainstream retractable solutions for the forecast horizon.
Key Report Takeaways
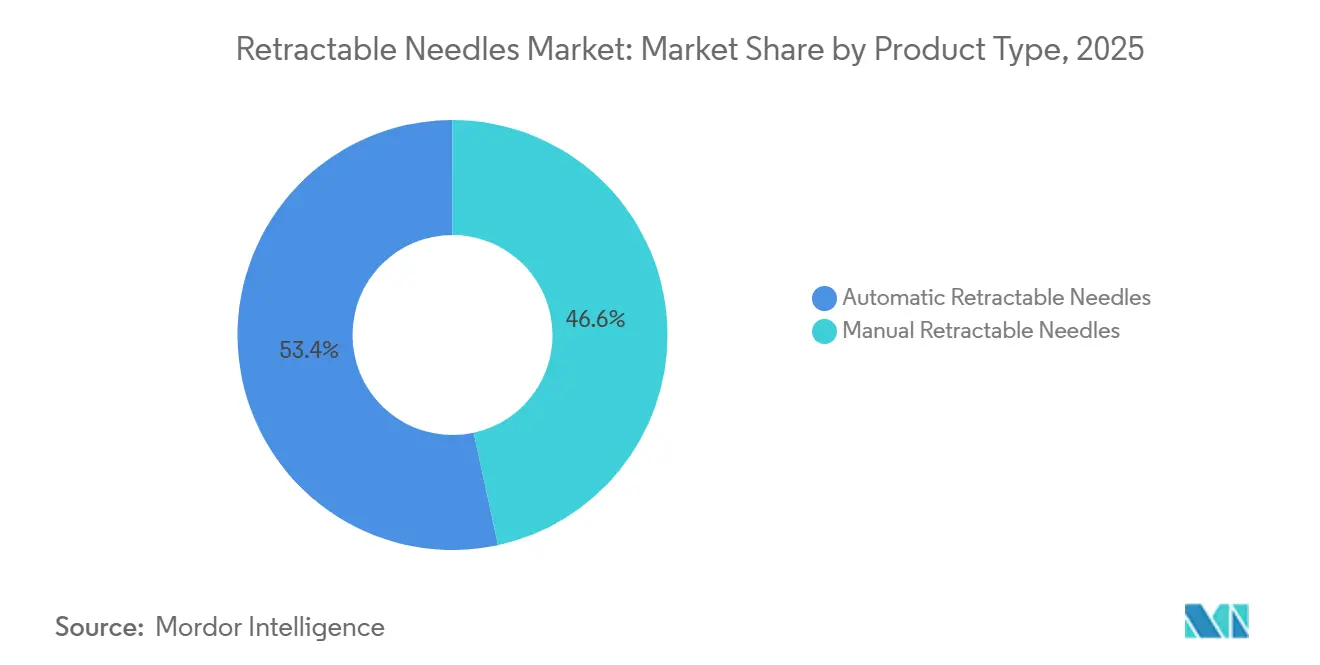
- By product type, automatic retractable needles commanded 53.39% of the retractable needles market share in 2025; manual formats are losing ground as hospitals migrate to passive devices that expand at a 5.23% CAGR through 2031.
- By application, subcutaneous injections accounted for 43.48% of the retractable needles market in 2025 and are projected to grow at a 4.78% CAGR, driven by GLP-1 therapies and expanded weight-management coverage.
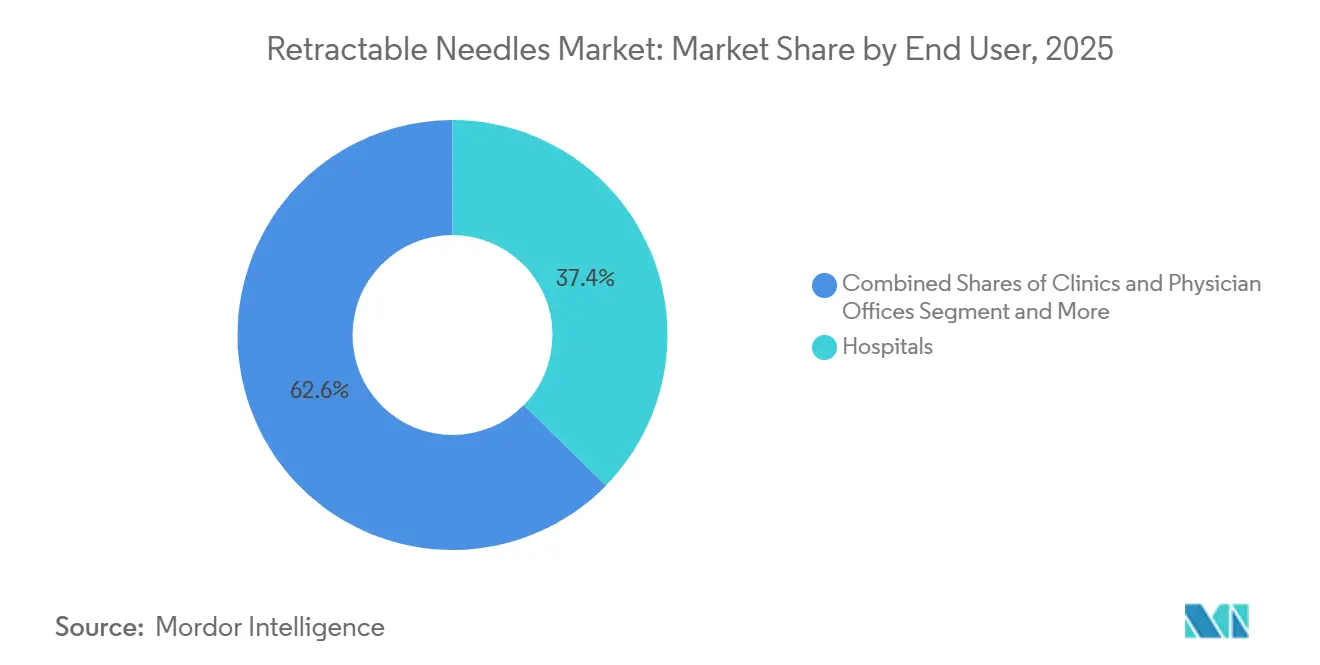
- By end user, clinics and physician offices are forecast to grow fastest at a 4.63% CAGR through 2031, even though hospitals retained 37.35% revenue share in 2025.
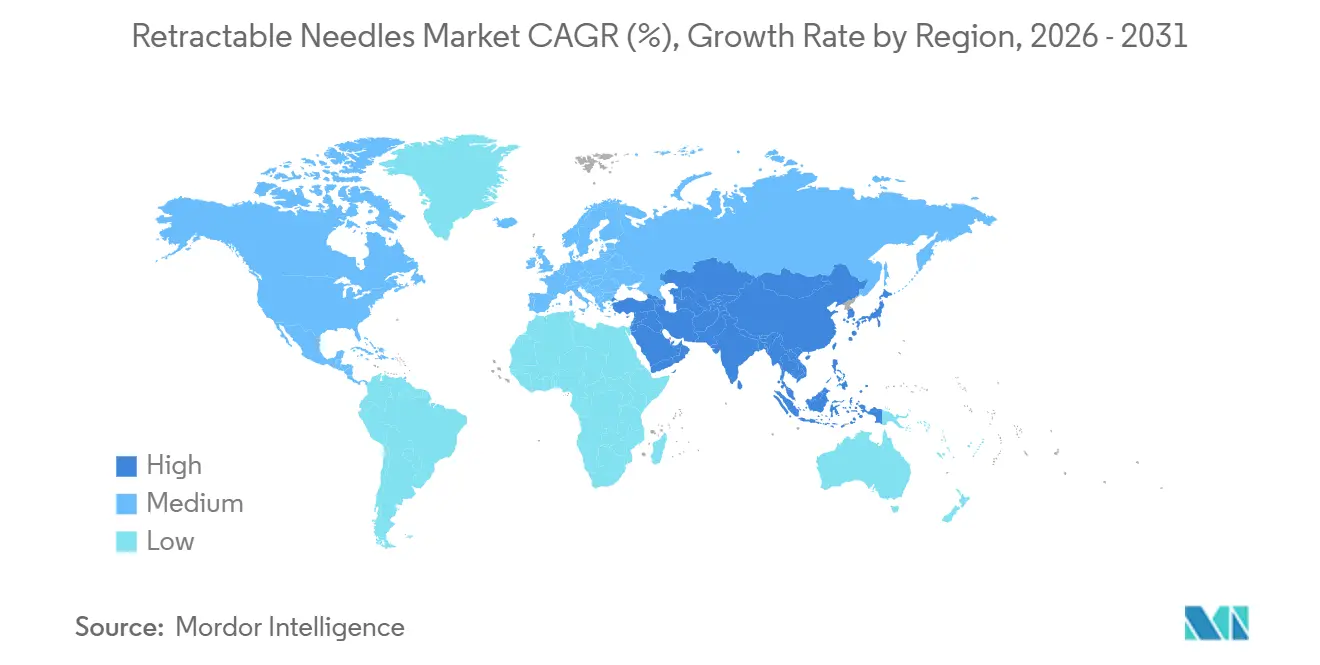
- By geography, North America accounted for 45.49% of global revenue in 2025, while Asia-Pacific is set to post the highest 5.12% CAGR as China and India expand vaccination and chronic disease programs.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Retractable Needles Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising injection volumes from chronic diseases and mass immunization programs | +1.5% | Global, Asia-Pacific and North America lead | Medium term (2-4 years) |
| Regulatory mandates and sharps-injury prevention policies | +1.2% | Global, strongest in North America and EU | Short term (≤ 2 years) |
| Shift to prefilled drug-device combinations with embedded safety | +1.1% | U.S. and Europe lead, Asia-Pacific rising | Medium term (2-4 years) |
| Innovation in passive retraction and low-dead-space designs | +0.9% | Global R&D hubs in U.S., EU, Japan | Long term (≥ 4 years) |
| Hospital procurement via GPOs favoring integrated safety and bundled contracts | +0.8% | North America expanding to EU & Asia-Pacific | Medium term (2-4 years) |
| Supply-chain localization and tariff-driven reshoring | +0.7% | U.S. and EU, selective Asia-Pacific | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Injection Volumes From Chronic Diseases And Mass Immunization Programs
Global diabetes prevalence is projected to reach 783 million adults by 2045, and U.S. GLP-1 utilization alone is expected to add roughly 1 billion subcutaneous doses per year by 2030 [2]International Diabetes Federation, “IDF Diabetes Atlas 10th Edition,” IDF, idf.org . Eli Lilly’s Zepbound became the top weight-management prescription in 2025, and its Employer Connect channel launched in March 2026 at USD 449 per pen, spurring uptake across 67 million Medicare Part D beneficiaries. Recent FDA approvals of biosimilars Yusimry and Wezlana extend safety-device demand to autoimmune therapies. WHO’s 2030 immunization agenda mandates auto-disable syringes for mass campaigns, further embedding retractable technology in emerging markets. Syringe Services Programs, now active in 44 states, distribute sterile sharps that increasingly incorporate passive safety tips in harm-reduction settings.
Regulatory Mandates And Sharps-Injury Prevention Policies Accelerating Safety Device Adoption
OSHA raised fines for serious violations to USD 16,550 and for willful violations to USD 165,514 in January 2025, elevating sharps-injury prevention from a compliance checkbox to a financial priority. Roughly 385,000 U.S. needlestick injuries still occur annually, and the majority of them remain unreported, exposing hospitals to litigation risk [3]Centers for Disease Control and Prevention, “Bloodborne Pathogens and Needlestick Prevention,” CDC, cdc.gov. The EU MDR requires safety mechanisms in labeling and packaging, forcing manufacturers to undergo 13- to 18-month CE mark reviews that now bottleneck legacy product renewals. WHO prequalification ties GAVI funding to auto-disable syringes that conform to ISO 23908:2024, effectively hard-wiring retractable needles into low-income immunization tenders. State-level statutes, such as California’s AB 2975, further tighten occupational-safety scrutiny and reinforce the commercial case for passive devices.
Shift To Prefilled Drug-Device Combinations with Embedded Safety
FDA cleared several prefilled combinations—Wezlana, Yusimry, Skyrizi—between 2024 and 2025, each embedding passive retractable mechanisms to cut dosing errors. Unither-Needle Concept’s Euroject blow-fill-seal line, announced in February 2025, will deliver one billion+ unit-dose syringes per year in Europe, tightening integration between drug and device manufacturing. Embecta secured 10-year cannula supplies from BD and will launch GLP-1 biosimilars in Canada, Brazil, China, and India in 2026, boosting retractable demand in price-sensitive markets. Large-volume wearables handling up to 10 mL push biologics into home settings and magnify the role of safety designs for non-professionals. Yet combination products carry an added 18- to 24-month development cycle due to ICH Q1A stability and human-factors demands, delaying smaller pharma entrants.
Innovation In Passive/Auto-Retractable Mechanisms And Low-Dead-Space Designs
Retractable Technologies owns patents US 11 331 448 and US 11 660 386, covering spring-loaded designs that retract automatically upon injection completion, eliminating user dependence. BD’s UltraSafe Plus guard, introduced in 2024, solved the 15% activation failure rate often observed with manual devices and quickly secured Vizient contracting wins. Low-dead-space designs recover 1–2 extra vaccine doses per vial, a decisive benefit during supply-constrained campaigns. Dual-chamber syringes now reconstitute biologics at the point of care, shrinking contamination risk. Environmental scrutiny is steering R&D toward reloadable autoinjectors because a projected 1 billion single-use GLP-1 pens would generate 35,000 tonnes of landfill waste by 2030.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Higher unit costs versus conventional devices and budget constraints | -1.3% | Global, acute in Asia-Pacific and Latin America | Short term (≤ 2 years) |
| Workflow and training gaps with manual safety mechanisms | -0.6% | Global, high-turnover facilities | Short term (≤ 2 years) |
| Competition from needle-free and alternative delivery modalities | -0.5% | North America and Europe | Long term (≥ 4 years) |
| Regulatory crackdowns or recalls causing intermittent supply disruptions | -0.4% | Global, acute under EU MDR | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Higher Unit Costs Versus Conventional Devices And Budget Constraints
Safety needles typically carry 20–30% unit premiums versus plain steel, straining procurement in Asia-Pacific and Latin America, where reimbursement growth lags. Reshoring adds immediate cost pressure: ICU Medical paid USD 33.6 million in tariffs during 2025, a sum likely passed through to hospitals. GPO contracts soften premium shock through bulk discounts, but long commitments risk obsolescence if next-gen devices emerge mid-cycle. Low-income immunization programs sometimes default to lowest-bid tenders that lack passive safeguards, undermining the intent of safety policies. Payer formularies echo this austerity: CVS dropped tirzepatide in July 2025, signaling broader resistance to high-priced injectable regimens.
Workflow And Training Gaps With Manual Safety Mechanisms
Manual retractors rely on staff activation, yet studies report failure rates of up to 15% in busy wards. High staff turnover amplifies protocol drift; new hires often default to conventional practice. California’s AB 2975 extends mandatory security training to 8 hours, demonstrating how competing mandates complicate workflow compliance, including sharps handling. Passive systems erase the activation step but cost more. Home-care patients show 38% needle aversion and elevated error rates, underscoring unmet education needs.
Segment Analysis
By Product Type: Automatic Designs Capture Procurement Momentum
Automatic retractable needles accounted for 53.39% of 2025 revenue, and is expected to grow with 5.23% CAGR through 2031. Hospital GPOs gravitate toward passive models that document lower failure rates and fewer OSHA reportables. Patents held by Retractable Technologies restrict fast-follower designs, compelling challengers to license IP or innovate around it. Manual needles linger in emerging markets where upfront costs dictate choice, but rising GAVI and WHO tenders embed auto-disable criteria that steadily shrink manual needles' share of the retractable needles market.
Supplier consolidation is tightening. MTD Group’s August 2024 purchase of Ypsomed’s pen-needle business vaulted it to the world’s number-two volume slot at 2.5 billion units annually, heightening bargaining power in GPO negotiations. Environmental concerns add a fresh angle: reloadable autoinjectors promise landfill cuts but lack finalized regulatory frameworks, delaying any immediate threat to single-use retractables. Overall, automatic devices will remain the volume and value engine inside the retractable needles market.
By Application: Subcutaneous Injections Benefit From GLP-1 Wave
Subcutaneous formats accounted for 43.48% of revenue in 2025 and will grow at a 4.78% CAGR, reflecting the momentum of semaglutide and tirzepatide. That growth will lift the subcutaneous portion of the retractable needles market size to roughly USD 3.12 billion in 2031. Medicare Part D coverage for anti-obesity injections, effective 2026, and employer adoption exceeding 65% by Q1 2026 anchor U.S. volume. On-body 10 mL platforms will divert some biologics away from syringes, yet cost and patient preference keep standard retractables in dominance this decade.
Intramuscular injections, notably vaccines, can achieve efficiency gains from low-dead-space tips that reclaim additional doses per vial, supporting immunization blitzes in LMICs. Intravenous use cases pivot toward safety IV catheters, but retractable needles persist in emergency and blood-draw settings where their quick deployment advantage stands. Needle-free entrants will nibble at both IM and SC routines but stay niche until economics improve.
By End User: Clinics And Physician Offices Outpace Hospitals
Hospitals accounted for 37.35% of 2025 demand due to bundled GPO contracts, yet ambulatory clinics will log the swiftest 4.63% CAGR through 2031 as chronic care shifts to outpatient settings. Prefilled GLP-1 pens and biosimilar autoimmune syringes align with physician-office workflows that prioritize turnaround time and patient convenience. Smart sharps containers with IoT tracking now appear in these settings, aligning with state waste-management rules.
Home-care channels add complexity: patient training gaps and needle aversion hamper perfect adoption. Nevertheless, telehealth coaching and smartphone-linked dispensers are improving technique compliance. Harm-reduction programs running 551 U.S. sites distribute safety-engineered syringes, reinforcing community-level demand that supplements institutional volumes.

Geography Analysis
North America accounted for 45.49% of 2025 global sales as OSHA penalties escalated and GLP-1 approvals drove injection counts. BD’s Nebraska expansion, due to go online mid-2026, illustrates the retreat from domestic capacity even as tariffs elevated manufacturing costs. Medicare coverage for anti-obesity drugs and employer uptake will add an estimated 1 billion annual doses by 2030, safeguarding regional growth prospects despite the reimbursement squeeze.
Asia-Pacific will pace the retractable needles market at a 5.12% CAGR to 2031. China and India invest heavily in vaccination drives and medical tourism infrastructure, boosting both institutional and home-care purchases. Chronic disease prevalence skews heavily toward the region, and Embecta’s 2026 GLP-1 biosimilar launches are set to capture pent-up demand. Domestic firms such as Nipro, Terumo, and Hindustan Syringes maintain price advantages, although uneven regulatory enforcement permits counterfeit penetration that erodes premium adoption.
Europe straddles opportunity and compliance risk. The EU MDR’s protracted certification process threatens temporary shortages when legacy SKUs retire before approvals. Yet, MTD Group’s pan-European capacity and Unither’s Euroject line anchor a robust supply base. Auto-disable mandates in GAVI-supported Eastern European programs deliver incremental tailwinds. Elsewhere, South America and the Middle East & Africa lag in absolute terms but show policy-driven openings as the WHO procurement specifications require ISO-23908 compliance.
Competitive Landscape
Competition is moderately concentrated; the top five suppliers account for the majority of the 2025 value. Becton, Dickinson and Company, Terumo Corporation, and B. Braun Melsungen AG own the majority of U.S. GPO volumes, leveraging vertically integrated drug-device offerings. BD’s USD 110 million Columbus plant adds Neopak syringes for biologics, demonstrating first-mover advantage in reshoring. Embecta’s 10-year cannula supply deal with BD secures raw-tube pipeline while it co-packages GLP-1 generics.
MTD Group’s Ypsomed buyout cemented its status as the second-largest pen-needle maker. Smaller innovators rely on patent-licensing playbooks; Retractable Technologies’ IP portfolio garners royalty streams while shielding U.S. sales. Needle-free developers such as Vaxxas remain strategic threats, but high cost and sterile-processing hurdles slow mass uptake. Tariff volatility and FDA QMSR tightening strain margins for import-dependent rivals, making localized smart factories a key differentiator.
Retractable Needles Industry Leaders
Becton, Dickinson and Company
Cardinal Health
Terumo Corporation
Nipro Corporation
B. Braun Melsungen AG
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2026: Orion Medical acquired Vernacare’s harm-reduction unit, gaining exclusive rights to distribute sharps containers to 551 U.S. syringe-service sites.
- March 2026: Eli Lilly rolled out Employer Connect, pricing Zepbound KwikPen at USD 449 across all strengths, with 15 administrator partnerships.
- January 2026: BD committed USD 110 million for Nebraska Neopak capacity, adding 120 jobs and mid-2026 go-live.
Global Retractable Needles Market Report Scope
As per the scope of the report, retractable needles are safety-engineered medical devices designed to prevent accidental needlestick injuries and reduce the risk of cross-contamination by drawing the needle into protective housing after use.
The retractable needles market is segmented by product type, application, end user, and geography. Based on product type, the market is segmented into automatic and manual retractable needles. By application, the market is segmented into subcutaneous, intramuscular, and intravenous injections. By end users, the market is segmented into hospitals, clinics, and physician offices, ambulatory surgical centers, and home care and community health.
Geographically, the market is segmented into North America, Europe, Asia-Pacific, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Automatic Retractable Needles |
| Manual Retractable Needles |
| Subcutaneous Injections |
| Intramuscular Injections |
| Intravenous Injections |
| Hospitals |
| Clinics and Physician Offices |
| Ambulatory Surgical Centers |
| Home Care and Community Health |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of MEA | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Automatic Retractable Needles | |
| Manual Retractable Needles | ||
| By Application | Subcutaneous Injections | |
| Intramuscular Injections | ||
| Intravenous Injections | ||
| By End User | Hospitals | |
| Clinics and Physician Offices | ||
| Ambulatory Surgical Centers | ||
| Home Care and Community Health | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of MEA | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large will global revenue be for retractable needles in 2031?
The retractable needles market size is forecast to reach USD 7.17 billion by 2031, rising at a 4.32% CAGR from 2027 to 2031.
Which product type is growing fastest?
Automatic retractable designs, representing 53.39% of 2025 value, are expanding at a 5.23% CAGR as hospitals prefer passive safety mechanisms.
Why is Asia-Pacific the most attractive regional opportunity?
Healthcare infrastructure growth in China and India plus rising chronic-disease injections push Asia-Pacific to a 5.12% CAGR, the quickest worldwide
What key regulation is shaping device design in Europe?
EU MDR mandates sharps-injury protection features and imposes 13–18 month certification cycles, compelling manufacturers to integrate passive safety systems.
Page last updated on:









