Pharmaceutical Environmental Monitoring System Market Size and Share
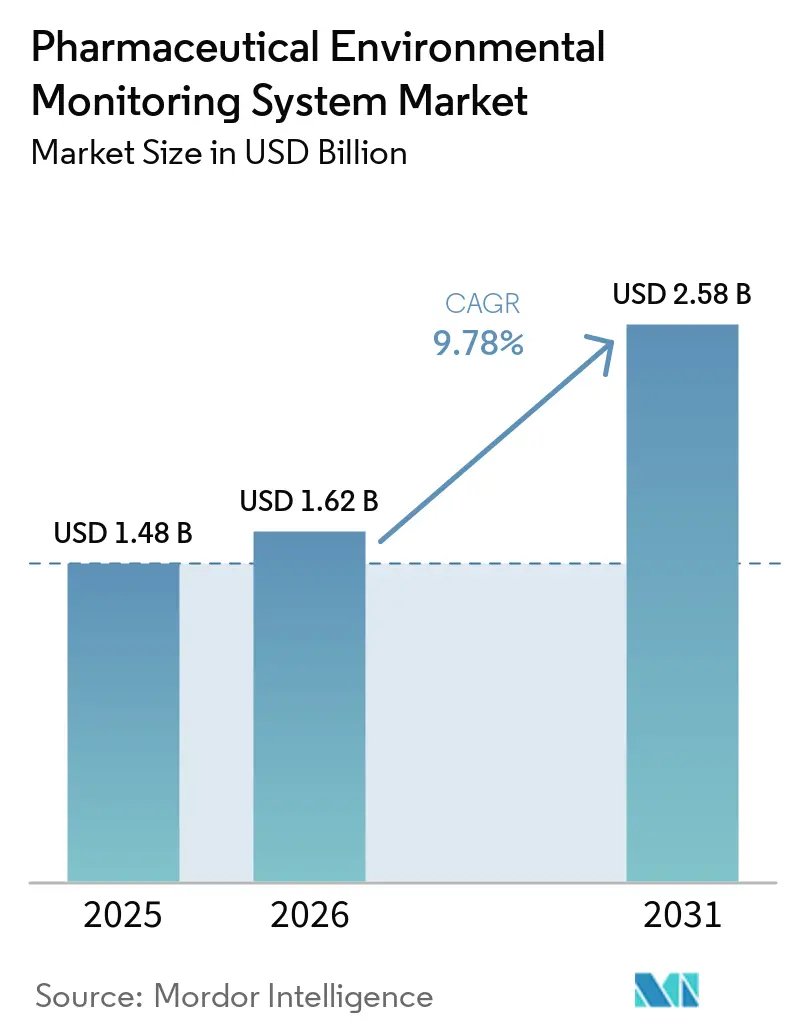
Pharmaceutical Environmental Monitoring System Market Analysis by Mordor Intelligence
The Pharmaceutical Environmental Monitoring System Market size was valued at USD 1.48 billion in 2025 and is estimated to grow from USD 1.62 billion in 2026 to reach USD 2.58 billion by 2031, at a CAGR of 9.78% during the forecast period (2026-2031).
The pharmaceutical environmental monitoring system market continues to advance as Annex 1 enforcement elevates continuous, real-time monitoring to a global expectation for sterile production, which is raising retrofit activity and standardizing practices across multinational sites. Strengthening of data integrity expectations under 21 CFR Part 11 keeps validated, audit-ready systems at the center of quality operations, and it is pushing investment in secure audit trails and role-based access across distributed teams. Capital commitments in biologics and cell and gene therapy fuel demand for higher monitoring density, especially around aseptic fill and finish and isolator-based production. The pharmaceutical environmental monitoring system market is also shaped by a measured shift to cloud-native and hybrid deployments that streamline validation and centralized oversight without sacrificing Part 11 controls. Competition remains active across instrumentation and software platforms, with vendors differentiating on real-time viable detection, secure architectures, and integration with MES, SCADA, and BMS layers.
Key Report Takeaways
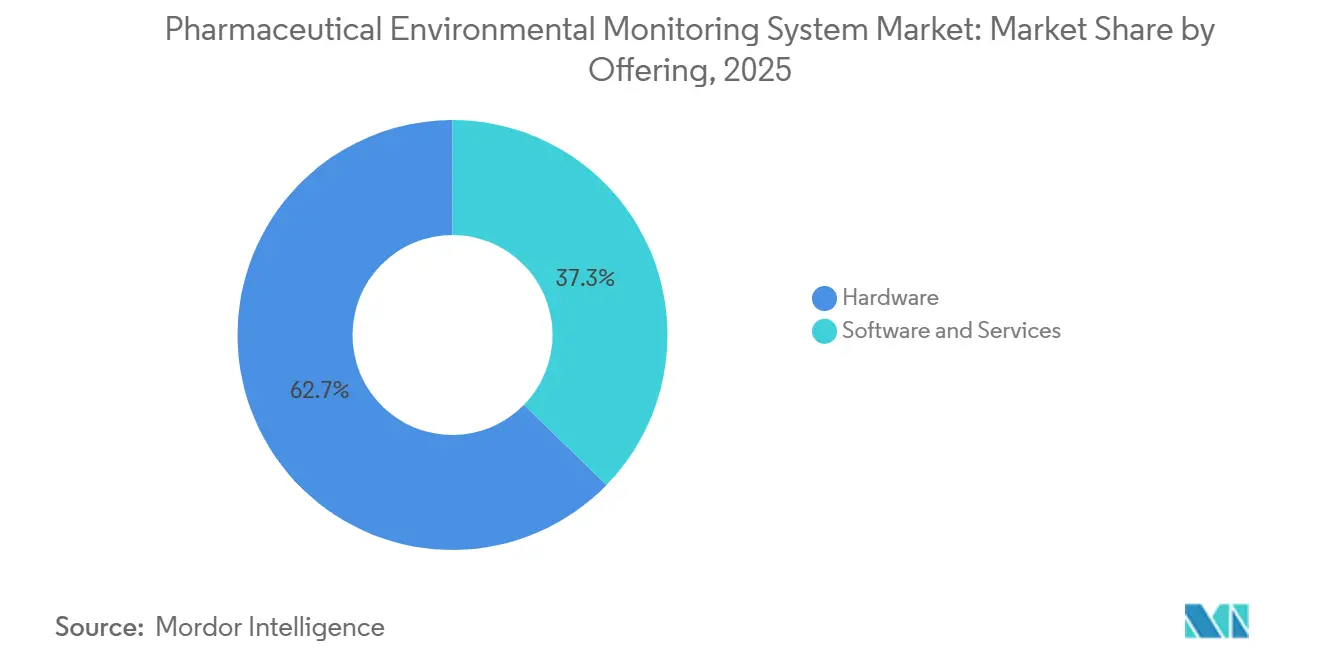
- By offering, Hardware led with 62.70% revenue share in 2025, while Software & Services is projected to grow at 7.18% CAGR through 2031.
- By monitoring type, Non-viable Air Particle Monitoring held 41.13% share in 2025, while Viable Environmental Monitoring is forecast to expand at 11.62% CAGR through 2031.
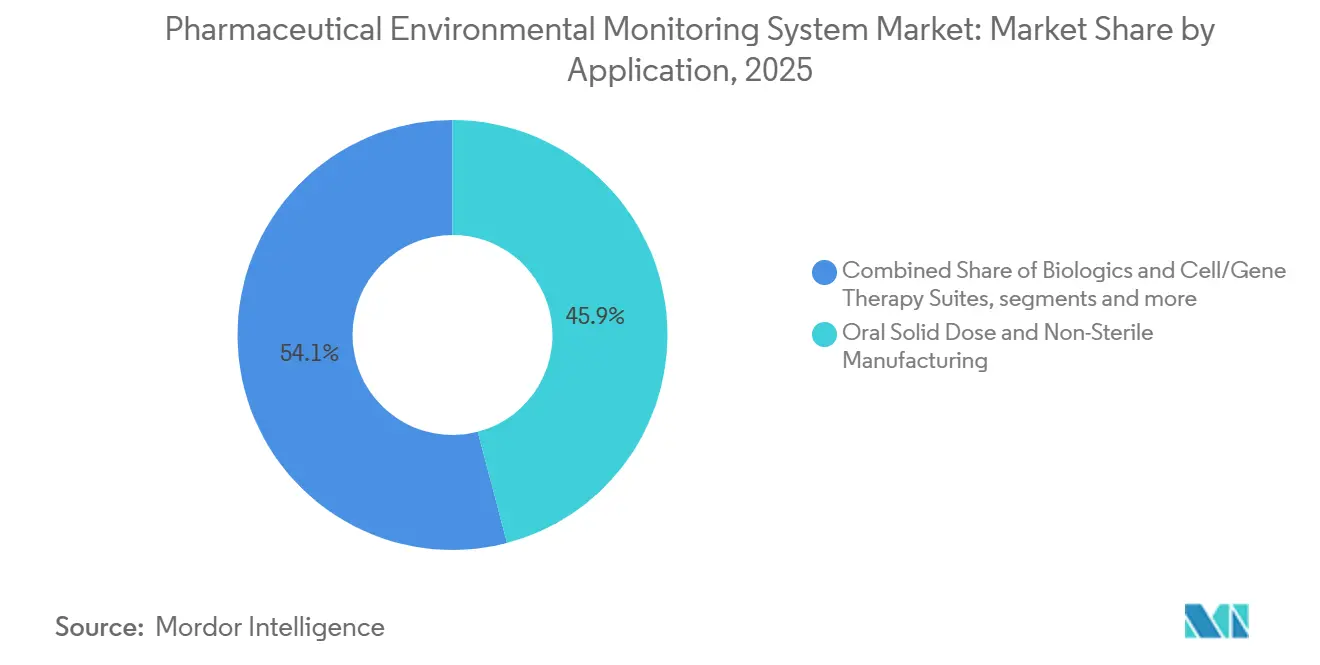
- By application, Oral Solid Dose and Non-Sterile Manufacturing accounted for 45.92% share in 2025, while Biologics and Cell or Gene Therapy Suites is projected to advance at 11.79% CAGR through 2031.
- By end user, Pharmaceutical and Biotechnology Manufacturers held 56.12% share in 2025, while CDMOs or CMOs are projected to grow at 12.15% CAGR through 2031.
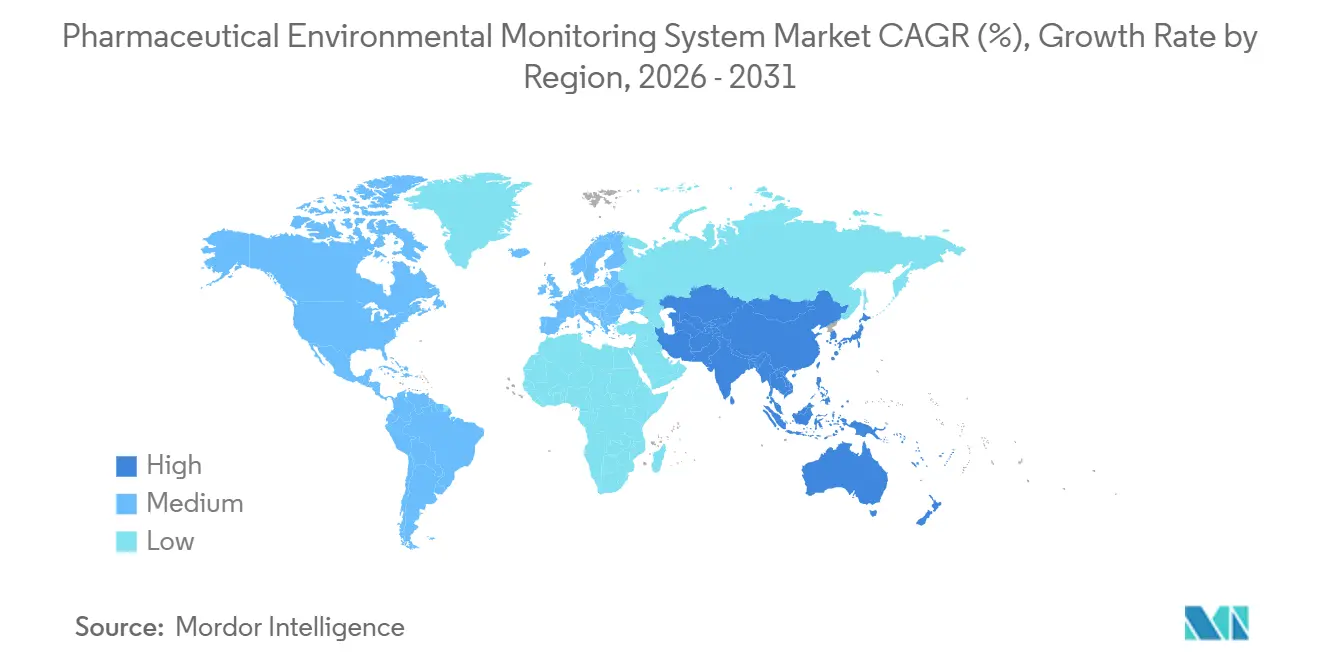
- By geography, North America retained 42.80% share in 2025, while Asia-Pacific is projected to post the fastest growth at 13.13% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Pharmaceutical Environmental Monitoring System Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Annex 1-Driven Shift to Continuous Monitoring in Grade A/B | +2.8% | Europe core, PIC/S jurisdictions with global spillover including North America and Asia-Pacific | Medium term (2-4 years) |
| Data-Integrity and 21 CFR Part 11 Validation Requirements | +2.1% | Global, with acute enforcement in United States, EU, and Japan | Short term (≤ 2 years) |
| Expanding Aseptic Capacity in Biologics and CGT | +2.4% | North America, Europe concentrated, China and India high growth | Long term (≥ 4 years) |
| Digitalization of EMS (Cloud/SCADA, Remote Alarming) | +1.5% | Global, spearheaded by multinational pharma and large CDMOs | Medium term (2-4 years) |
| USP <797> 2023 Enforcement in U.S. Compounding | +0.9% | United States primary, Canada aligned regulations | Short term (≤ 2 years) |
| Energy Optimization Needs Driving Sensor-Led Control | +0.7% | Global, with emphasis on EU sustainability mandates and North American operational cost reduction | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Annex 1-Driven Shift to Continuous Monitoring in Grade A/B
Harmonization through PIC/S has extended these expectations across a large set of inspection authorities, which reduces ambiguity for global manufacturers that operate common platforms across regions[1]Particle Measuring Systems, “EU GMP Guidelines for Environmental Monitoring, Questions and Answers,” Particle Measuring Systems, pmeasuring.com. Inspectors now look for evidence that cleanrooms meet classification at rest and maintain real-time process control in operation supported by airflow visualization and robust trending. Legacy plants are prioritizing risk assessments, particle transport loss evaluations, and alarm integration into SCADA or BMS so that excursions tie directly to documented corrective and preventive actions.
Data-Integrity and 21 CFR Part 11 Validation Requirements
Persistent enforcement of 21 CFR Part 11 has raised the compliance bar for environmental monitoring systems with secure, immutable audit trails and role-based access across users and devices. Environmental data, alarm acknowledgments, threshold changes, and user actions are expected to be captured with full metadata and maintained in validated systems that support periodic review. The regulatory posture also covers compounding environments in the United States where environmental monitoring programs and data integrity procedures are integral to inspection readiness. Vendors are responding with cloud-capable platforms that meet audit trail, encryption, and authentication expectations while easing patching and backup overhead for quality and IT teams. The underlying aim in the pharmaceutical environmental monitoring system market is to maintain trustworthy, reviewable records that stand during audits and support rapid deviation investigation without manual data stitching.
Expanding Aseptic Capacity in Biologics and CGT
Investment in advanced therapy and biologics manufacturing is expanding aseptic footprints that require higher-density particle and viable monitoring across isolators, Grade B buffer rooms, and critical utilities. Aseptic fill and finish requires continuous non-viable monitoring during critical operations along with viable methods that can inform investigations in closer to real time. Real-time viable detection technology is gaining traction to reduce time-to-result and enable faster root cause analysis that avoids long production holds. The scale and consequence of batch values in biologics and cell or gene therapy support premium architectures that tie real-time environmental data to electronic batch records and validated alarm workflows. The pharmaceutical environmental monitoring system market is therefore aligned with long-cycle capacity projects where monitoring capabilities are defined during design and commissioning rather than added late in validation.
Digitalization of EMS (Cloud/SCADA, Remote Alarming)
Cloud-native and hybrid EMS deployments are moving into mainstream adoption as validated vendors deliver platforms that centralize alarms and data across multiple sites while reducing on-premises server maintenance. The shift improves scalability for large portfolios and simplifies audit readiness through uniform configurations and controlled updates across distributed environments. With integrated SCADA and BMS connectivity, teams can correlate environmental excursions with equipment and process signals and then automate work orders and CAPA initiation within defined workflows. Advanced analytics and AI-assisted control are also being piloted to identify deviation patterns and trim HVAC energy consumption while preserving ISO 14644 compliance. The pharmaceutical environmental monitoring system market is responding with Part 11-capable audit trails, encryption, and role-based access that address regulatory expectations as teams adopt remote alarming and enterprise dashboards.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Upfront Cost and Validation Burden | -1.4% | Global, acute in small to mid-size biotech and CMOs with capital constraints | Short term (≤ 2 years) |
| Brownfield Integration with Legacy BMS/IT | -0.9% | Mature markets in North America and Europe with aging infrastructure | Medium term (2-4 years) |
| Slow Acceptance of Rapid Viable Monitoring | -0.5% | Global, faster adoption in North America and Europe versus conservative parts of Asia-Pacific | Medium term (2-4 years) |
| Cybersecurity and Cloud Policies Limiting Adoption | -0.6% | Global, with stringent data sovereignty in EU, China, and regulated healthcare settings | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
High Upfront Cost and Validation Burden
Capital and validation workloads weigh most on smaller organizations and contract manufacturers that must qualify hardware, software, and alarm logic without disrupting production. Qualification steps across IQ, OQ, and PQ, along with calibration and documentation, extend timelines and add service costs that are difficult to absorb mid-campaign. Higher performance viable monitoring technologies promise faster decisions, yet they require careful method validation and change control before replacing culture-based routines. Project teams also face room closures to install fixed sampling points and integrate sensor networks with SCADA or BMS, which can compress qualification windows and create scheduling pressure. The pharmaceutical environmental monitoring system market contends with these hurdles by offering pre-validated packages and cloud-managed platforms that reduce server costs and facilitate updates within controlled procedures.
Brownfield Integration with Legacy BMS/IT
Legacy facilities often run siloed HVAC controls, utilities monitoring, and environmental sensors that complicate unified alarming and data trending when teams attempt to integrate modern EMS platforms. Any change to validated control systems triggers change control and re-qualification, and this extends project timelines while technical teams align documentation with as-built configurations. Security hardening is also required when connecting older equipment to modern IT networks, since network segmentation and credential management can lag on aging devices. To mitigate risk, experienced integrators run staged testing, isolate system changes, and schedule commissioning to avoid unplanned downtime and cascading alarms. The pharmaceutical environmental monitoring system market addresses these constraints with integration-ready interfaces and validation documentation that streamline multi-vendor projects across entrenched automation layers.
Segment Analysis
By Offering: Capital Equipment Anchors While Software Gains
Hardware captured 62.70% of the pharmaceutical environmental monitoring system market share in 2025 as airborne particle counters, viable samplers, and environmental sensors remain the core sensing layer in compliant cleanrooms. Fixed and remote particle counters continue to replace portable-only routines in aseptic operations, as sites pursue continuous monitoring with validated alert and action thresholds. Microbial air samplers and real-time viable counters are drawing interest where faster detection supports on-shift root cause analysis and better excursion response. Environmental sensors for temperature, humidity, airflow, and differential pressure remain essential in every suite, and they are increasingly tied into centralized platforms. At the same time, software platforms that manage alarms, audit trails, and reporting across plants are gaining adoption as teams standardize across global portfolios.
Software & Services is the fastest-growing component, and the pharmaceutical environmental monitoring system market size for this segment is projected to expand at 7.18% CAGR through 2031 as cloud-native architectures reduce server maintenance while meeting Part 11 expectations. Vendors are delivering validated audit trails, encryption, and role-based access in subscription models that ease updates without full requalification. Integration with SCADA and BMS lets teams connect environmental trends to process and equipment signals, which improves deviation investigations and energy control. In the pharmaceutical environmental monitoring system market, this shift is strongest among multi-site enterprises that want unified configurations across aseptic sites while preserving local workflows.
By Monitoring Type: Real-Time Viable Detection Accelerates Fastest
Non-viable Air Particle Monitoring held 41.13% in 2025 as the baseline classification and control tool for ISO and GMP compliance across Grades A through D. Annex 1 requires continuous monitoring during critical Grade A operations, and this standard is increasing deployments of fixed and remote counters linked to validated historians. Remote particle counters help sites avoid the labor and timing limits of portable-only routines and enable real-time responses to excursions during filling. The pharmaceutical environmental monitoring system market continues to rely on non-viable trends to indicate process instability that can precede bioburden movement, which makes continuous sampling essential for critical steps.
Viable Environmental Monitoring is the fastest-growing type with an 11.62% CAGR to 2031, driven by rapid microbiological methods that deliver actionable signals closer to real time. Laser-induced fluorescence viable counters, ATP bioluminescence, and other rapid approaches reduce time-to-result and support earlier intervention and better release decisions when paired with confirmatory culture. Case studies in sterile facilities show that integrated ID strategies and rapid detection shorten investigations without displacing compendial requirements[2].Vaisala, “Cleanroom Environmental Monitoring,” Vaisala, vaisala.com Strategic investment by leading QC players underscores confidence in continuous viable monitoring as a complement to established microbiology. The pharmaceutical environmental monitoring system market therefore balances classical plate-based tools with rapid instruments that close information gaps during aseptic production.
By Application: Biologics and CGT Drive Premium Segment Growth
Oral Solid Dose and Non-Sterile Manufacturing held the largest share at 45.92% in 2025 due to the extensive installed base operating at ISO Class 7 or 8 and the long history of adoption in these facilities. These environments maintain strict temperature, humidity, airflow, and particle control, but they typically require fewer continuous viable points than aseptic suites. Even with incremental growth, replacement cycles and upgrades support steady demand for instrumentation and software platforms that standardize reporting and integrity controls. The pharmaceutical environmental monitoring system market in these applications tends to emphasize long-term reliability, calibration support, and integration with site quality systems.
Biologics and Cell or Gene Therapy Suites is the fastest-growing application with an 11.79% CAGR to 2031 and it features the densest monitoring across isolators and critical Grade B settings. Higher batch value and sterility assurance needs favor real-time viable detection that complements continuous non-viable trends, which reduces investigative lead times and supports electronic batch records. The pharmaceutical environmental monitoring system market aligns with investments in advanced therapy production where monitoring is engineered into facility design and commissioning. Aseptic fill and finish of high-value therapies underscores why validated alarm logic and continuous trending are non-negotiable in modern GMP settings. The pharmaceutical environmental monitoring system market size for this application is supported by capital programs that embed monitoring architectures early to avoid late-cycle remediation.

By End User: CDMOs Drive Double-Digit Growth
Pharmaceutical and Biotechnology Manufacturers held 56.12% in 2025 as large innovators and specialty manufacturers operate the majority of aseptic capacity, pilot plants, and commercial biologics lines. These sites standardize configurations and reporting across global networks to streamline validation and make cross-site trending easier for corporate quality. For these users, the pharmaceutical environmental monitoring system market is defined by platform stability, validated integration with MES, and field service that scales worldwide. Stronger enforcement in sterile compounding has also broadened adoption among hospital pharmacies and health systems that must maintain ISO-classified spaces and meet state inspections.
CDMOs and CMOs are the fastest-growing group with a 12.15% CAGR to 2031 as sponsors continue to outsource sterile manufacturing and clinical supply, which requires multi-tenant alarm thresholds and flexible reporting per client. The end-user pattern favors software-defined monitoring that reconfigures quickly between campaigns while maintaining Part 11 audit controls. The pharmaceutical environmental monitoring system market in CDMOs is often characterized by rapid changeovers, complex deviation analysis, and early integration with sponsor quality systems. Growth in advanced therapy outsourcing adds further momentum as contract suites implement dense non-viable and viable monitoring around patient or donor material handling.
Geography Analysis
North America retained 42.80% of the pharmaceutical environmental monitoring system market share in 2025 supported by concentrated biologics innovation, significant aseptic capacity, and strong Part 11 enforcement that anchors validated monitoring adoption. New U.S. projects in advanced therapies sustain demand for continuous non-viable and viable monitoring along with integrated alarm management that ties excursions to batch records. Canada and Mexico add incremental capacity with modernization in selected hubs, while U.S. compounding environments continue to formalize environmental monitoring under updated standards.
Europe remains a core pillar as Annex 1 enforcement catalyzes upgrades to continuous monitoring and harmonized contamination control strategies. Multinational manufacturers are aligning architectures across Germany, the United Kingdom, France, Italy, and Spain, which supports vendors able to provide consistent validation packages and field service. The pharmaceutical environmental monitoring system market in Europe also benefits from ongoing biologics investments and adoption of rapid viable technologies that reduce investigation delays without displacing compendial methods. Brownfield integration projects remain significant as plants update legacy controls and alarm logic to meet modern data integrity and continuous monitoring expectations.
Asia-Pacific is the fastest-growing region with a 13.13% CAGR through 2031 as greenfield aseptic capacity in China, India, and South Korea embeds continuous monitoring into project designs from day one. India’s push into biosimilars and regulated-market submissions and China’s drive for self-sufficiency in biologics are expanding the installed base that depends on validated, Annex 1-aligned monitoring. The pharmaceutical environmental monitoring system market size in the region reflects new facilities that prefer standardized, multi-site compatible platforms which ease training and validation as networks scale. Australia and Japan contribute with high-compliance deployments and cloud-forward monitoring across distributed networks, supported by vendors with proven Part 11 capabilities.
Competitive Landscape
The pharmaceutical environmental monitoring system market features established instrumentation leaders alongside software-first platforms that emphasize cloud and analytics. Particle Measuring Systems, TSI, Beckman Coulter Life Sciences, Lighthouse Worldwide Solutions, and Vaisala compete on sensing performance, regulatory pedigree, and validated software ecosystems. Installed-base inertia and calibration service relationships support retention, while rapid viable monitoring creates room for differentiated offerings.
On the software side, vendors expand SaaS portfolios with secure audit trails, access controls, and multi-site management that align with 21 CFR Part 11 expectations. Integration with SCADA and BMS is a key differentiator since customers want unified alarming, automated work orders, and faster deviation investigations that connect environment and equipment states. The pharmaceutical environmental monitoring system market also reflects the rise of analytics and AI-assisted control that complement standard monitoring by predicting excursions and optimizing HVAC energy without compromising ISO classifications.
Strategic moves underscore the direction of travel. bioMérieux invested in Plair in March 2026 to accelerate real-time viable particle counting for cleanrooms and to leverage its global QC footprint for adoption. Trescal acquired Facility Monitoring Systems in February 2025 to strengthen contamination control services including design, installation, and maintenance in regulated environments. Thermo Fisher’s acquisition of Solventum’s Purification and Filtration business in 2025 expanded bioproduction capabilities in adjacent filtration domains that interact with cleanroom and water-quality controls. These actions point to an ecosystem where sensing, software, and services converge to support validated, scalable monitoring for high-consequence production.
Pharmaceutical Environmental Monitoring System Industry Leaders
-
Beckman Coulter Life Sciences (MET ONE)
-
Lighthouse Worldwide Solutions
-
Particle Measuring Systems (Spectris)
-
TSI Incorporated
-
Vaisala
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2026: bioMérieux invested over CHF 1 million in Plair, a Geneva-based scale-up commercializing the RAPID-C+ real-time viable particle counter that combines viable and total particle counting with continuous certified sampling onto standard agar media. bioMérieux Senior Vice President of Pharma Quality Control Michael Reynier joined Plair's Board of Directors as part of the strategic partnership, signaling bioMérieux's commitment to advancing rapid environmental monitoring for pharmaceutical cleanrooms and leveraging its global commercial presence (160+ countries) to accelerate RAPID-C+ adoption.
- February 2026: Johnson & Johnson announced a USD 1+ billion investment to construct a next-generation cell therapy manufacturing facility in Lower Gwynedd, Montgomery County, Pennsylvania, to expand U.S. biomanufacturing capacity for cell therapies targeting cancer, immune-mediated, and neurological diseases. The facility, expected to create approximately 500 skilled biomanufacturing jobs when fully operational, is part of J&J's USD 55 billion U.S. investment in manufacturing, R&D, and technology through early 2029 and will require state-of-the-art environmental monitoring systems to support GMP compliance and contamination control in advanced therapy production.
- January 2026: Genentech increased its investment in a new manufacturing facility in Holly Springs, North Carolina, from an initial USD 700+ million to approximately USD 2 billion to boost production capacity for metabolic medicines targeting obesity and Type 2 diabetes. The 700,000 sq ft facility, expected to commission in 2029, will incorporate modern biomanufacturing technologies, advanced automation, and digital systems requiring validated environmental monitoring architectures aligned with FDA and GMP expectations.
Global Pharmaceutical Environmental Monitoring System Market Report Scope
As per the report’s scope, the pharmaceutical manufacturers implement an environmental monitoring system (EMS) as a structured program to routinely measure and manage particulate, microbial, and physical parameters. This approach ensures cleanrooms comply with established safety standards, maintaining product quality and sterility by mitigating contamination risks proactively.
The pharmaceutical environmental monitoring system market is segmented into offering, monitoring type, application, end user, and geography. By offering, the market is segmented into hardware and software & services. By hardware, the market is segmented into airborne particle counters (portable, remote, handheld), microbial air samplers (active, passive, rapid methods), environmental sensors (temperature, rh, dp, airflow, co2), data loggers & transmitters, controllers/io/plc & scada nodes, and others. By monitoring type, the market is segmented into non-viable air particle monitoring, viable environmental monitoring, environmental parameters monitoring (T/RH/DP/airflow/CO2), and utilities & water systems monitoring (WFI/PW, compressed gases). By application, the market is segmented into aseptic fill-finish and sterile injectables, biologics & cell/gene therapy suites, oral solid dose & non-sterile manufacturing, QC microbiology labs & cleanroom support, warehousing, cold rooms, and distribution, and others. By end user, the market is segmented into pharmaceutical and biotechnology manufacturers, CDMOs/CMOs, hospital compounding pharmacies, and others. By geography, the market is segmented as North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| Hardware | Airborne Particle Counters (portable, remote, handheld) |
| Microbial Air Samplers (active, passive, rapid methods) | |
| Environmental Sensors (temperature, RH, DP, airflow, CO2) | |
| Data Loggers & Transmitters | |
| Controllers/IO/PLC & SCADA nodes | |
| Others | |
| Software & Services |
| Non-viable Air Particle Monitoring |
| Viable Environmental Monitoring |
| Environmental Parameters Monitoring (T/RH/DP/airflow/CO2) |
| Utilities & Water Systems Monitoring (WFI/PW, compressed gases) |
| Aseptic Fill-Finish and Sterile Injectables |
| Biologics & Cell/Gene Therapy Suites |
| Oral Solid Dose & Non-Sterile Manufacturing |
| QC Microbiology Labs & Cleanroom Support |
| Warehousing, Cold Rooms, and Distribution |
| Others |
| Pharmaceutical and Biotechnology Manufacturers |
| CDMOs/CMOs |
| Hospital Compounding Pharmacies |
| Others |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Offering | Hardware | Airborne Particle Counters (portable, remote, handheld) |
| Microbial Air Samplers (active, passive, rapid methods) | ||
| Environmental Sensors (temperature, RH, DP, airflow, CO2) | ||
| Data Loggers & Transmitters | ||
| Controllers/IO/PLC & SCADA nodes | ||
| Others | ||
| Software & Services | ||
| By Monitoring Type | Non-viable Air Particle Monitoring | |
| Viable Environmental Monitoring | ||
| Environmental Parameters Monitoring (T/RH/DP/airflow/CO2) | ||
| Utilities & Water Systems Monitoring (WFI/PW, compressed gases) | ||
| By Application | Aseptic Fill-Finish and Sterile Injectables | |
| Biologics & Cell/Gene Therapy Suites | ||
| Oral Solid Dose & Non-Sterile Manufacturing | ||
| QC Microbiology Labs & Cleanroom Support | ||
| Warehousing, Cold Rooms, and Distribution | ||
| Others | ||
| By End User | Pharmaceutical and Biotechnology Manufacturers | |
| CDMOs/CMOs | ||
| Hospital Compounding Pharmacies | ||
| Others | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the pharmaceutical environmental monitoring system market growth outlook through 2031?
It is projected to reach USD 2.58 billion by 2031, registering a 9.78% CAGR from 2026 to 2031.
Which offering leads demand in the pharmaceutical environmental monitoring system market?
Hardware led with 62.70% revenue share in 2025, supported by continuous non-viable monitoring and dense sensor networks in aseptic operations.
Which monitoring type is expanding fastest in this space?
Viable Environmental Monitoring is the fastest-growing type with an 11.62% CAGR to 2031 as rapid methods move closer to real-time detection.
Which applications are driving premium deployments?
Biologics and Cell or Gene Therapy Suites show the highest growth at 11.79% CAGR, favoring continuous particle and viable monitoring integrated with electronic batch records.
Where is regional growth most pronounced for the pharmaceutical environmental monitoring system market?
Asia-Pacific is the fastest-growing region with a 13.13% CAGR through 2031 on the back of greenfield aseptic capacity in China, India, and South Korea.
What are the main regulatory factors shaping purchase decisions?
Annex 1 enforcement and 21 CFR Part 11 integrity expectations drive continuous monitoring, validated audit trails, and secure access in system selection.
Page last updated on:









