Ozone Therapy In Dermatology Market Size and Share
Ozone Therapy In Dermatology Market Analysis by Mordor Intelligence
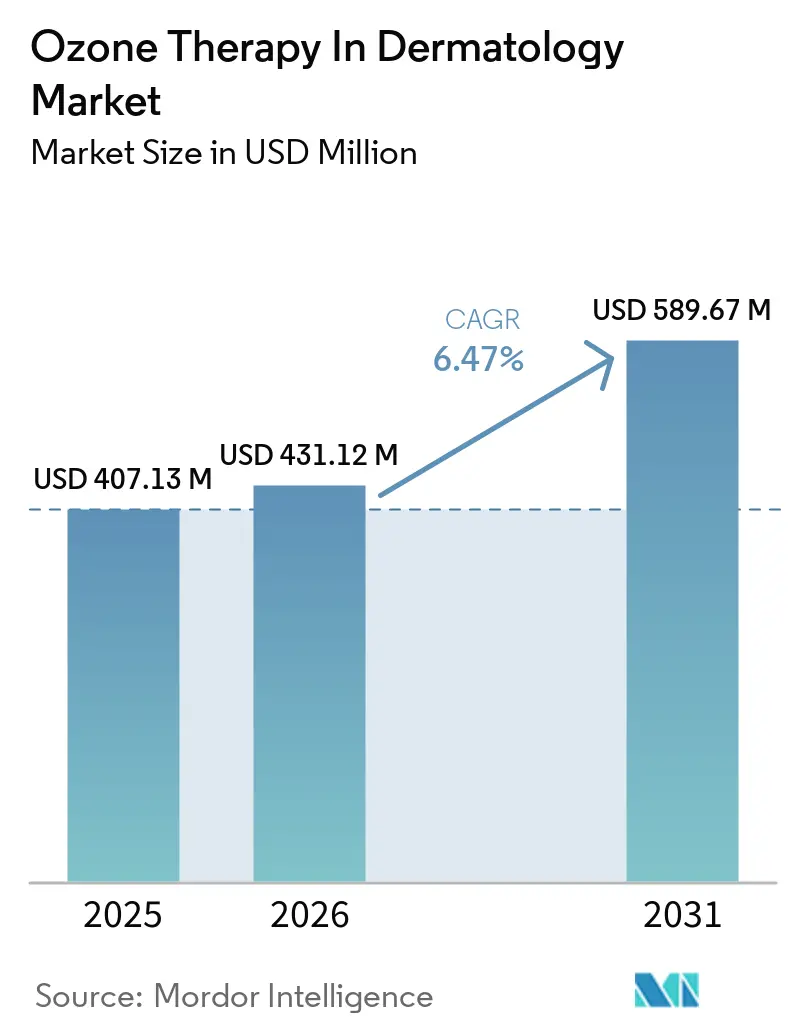
The Ozone Therapy In Dermatology Market size is projected to expand from USD 407.13 million in 2025 and USD 431.12 million in 2026 to USD 589.67 million by 2031, registering a CAGR of 6.47% between 2026 to 2031.
Demand is rising because clinicians increasingly validate ozone for chronic-wound protocols, regulators in Brazil and China have opened formal pathways, and patients seek regenerative options that avoid systemic drugs. Device makers now offer photometric concentration monitoring that improves dosing precision, while aesthetic clinics bundle ozone with platelet-rich plasma or exosomes to capture premium, cash-pay procedures. Competitive intensity is moderate because European manufacturers dominate certified equipment, but U.S. distributors operate under state health-freedom statutes instead of federal clearance.
Key Report Takeaways
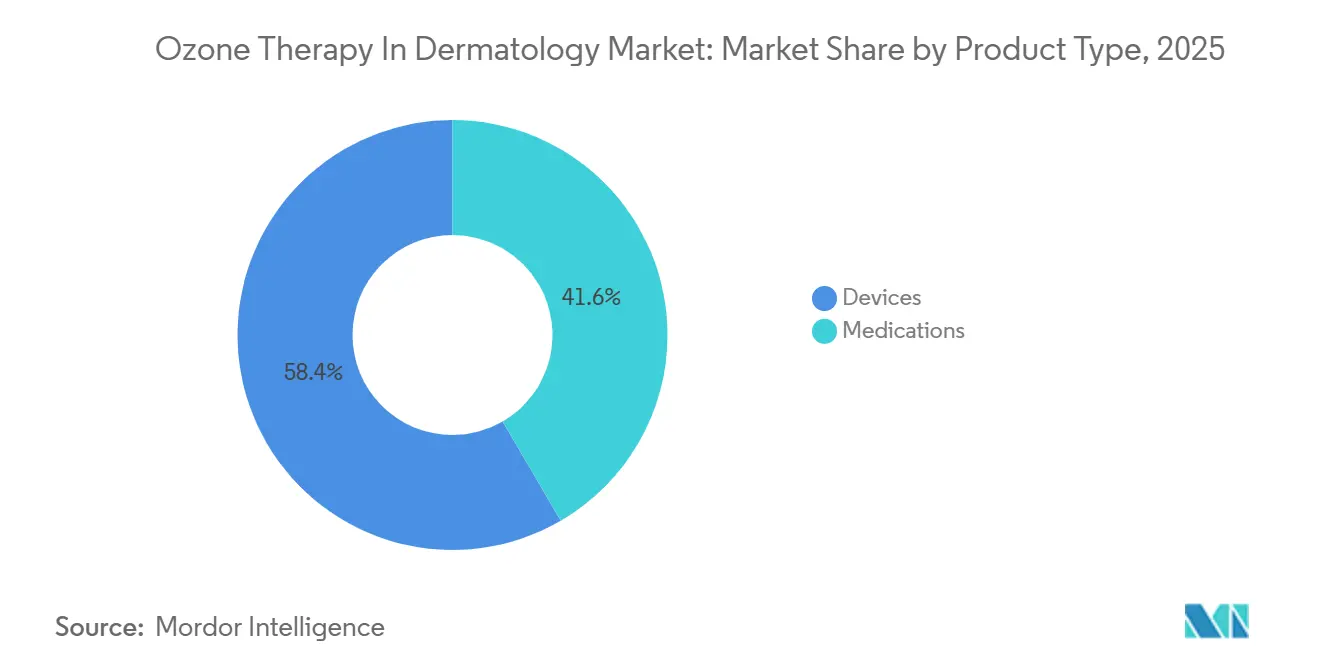
- By product type, devices led with 58.42% of the ozone therapy in dermatology market share in 2025, while medications are set to grow at a 7.52% CAGR through 2031.
- By treatment method, topical ozone commanded 24.78% of the ozone therapy in dermatology market in 2025; ozone water therapy records the highest projected CAGR at 7.25% to 2031.
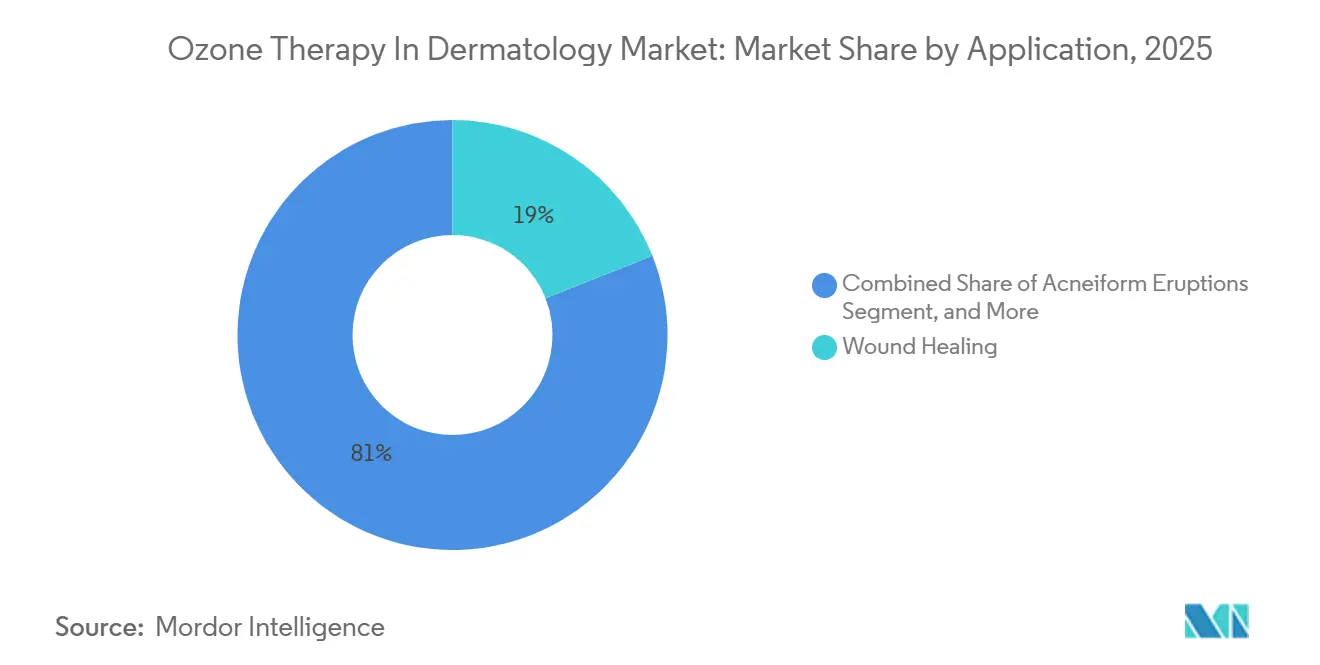
- By application, wound healing held 19.02% revenue in 2025, whereas anti-aging procedures are forecast to advance at an 8.09% CAGR to 2031.
- By end user, dermatology clinics captured 48.03% of 2025 revenue, yet aesthetic clinics show the fastest growth at 9.16% CAGR through 2031.
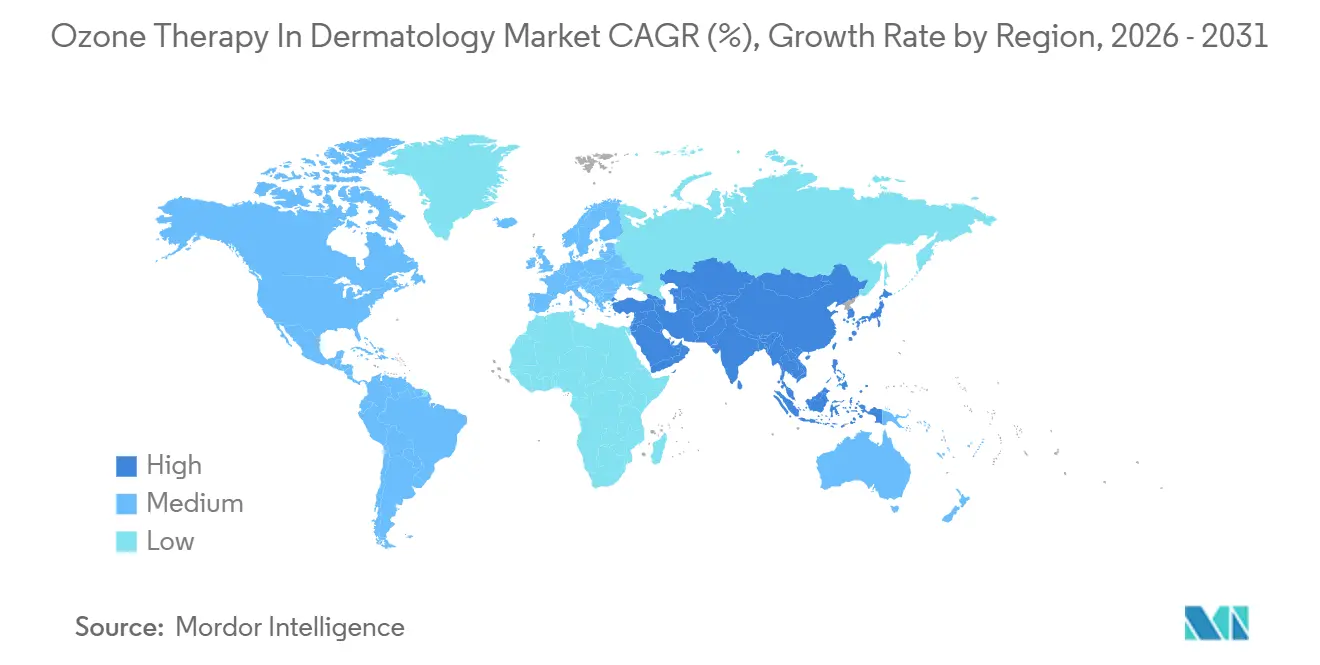
- By geography, North America led with 41.83% share of the ozone therapy in dermatology market size in 2025, while Asia-Pacific is projected to expand at a 8.06% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Ozone Therapy In Dermatology Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising prevalence of chronic skin disorders | +1.2% | Global, highest in low-SDI regions | Long term (≥4 years) |
| Growing demand for non-invasive “natural” care | +0.9% | North America, Europe, urban Asia-Pacific | Medium term (2–4 years) |
| Advances in ozone generators & topicals | +0.8% | Germany, Italy, Japan, China | Medium term (2–4 years) |
| Adoption surge in EU & NA clinics | +0.7% | Germany, Italy, Spain, selected U.S. states | Short term (≤2 years) |
| AI-driven smart-dosimetry systems | +0.5% | High-income markets | Long term (≥4 years) |
| Hybrid ozone + regenerative therapies | +1.0% | Brazil, Italy, Asia-Pacific aesthetic segment | Medium term (2–4 years) |
| Source: Mordor Intelligence | |||
Rising Prevalence of Chronic Skin Disorders
Acne, psoriasis, and atopic dermatitis continue to climb worldwide; adult acne cases reached 49.40 million in 2021 and remain highest in low-SDI countries.[1]Wenjie Yan et al., “Global Adult Acne Trends,” pmc.ncbi.nlm.nih.gov Ozone’s antimicrobial and wound-healing actions give clinicians a steroid-sparing adjunct, illustrated by a 2025 cohort in Poland where ozone plus negative-pressure therapy cut diabetic-foot-wound volume by 92.71% in three weeks.[2]Askar Abdykadyrov et al., “Intelligent Control of High-Frequency Ozone Generators,” mdpi.com
Growing Demand for Non-Invasive & “Natural” Treatments
Patients wary of corticosteroids or biologics gravitate to procedures perceived as natural. A 2024 Brazilian RCT showed platelet-rich plasma mixed with 30 µg/mL ozone improved wrinkle scores more than PRP alone, with only transient burning reported. Social-media promotion has accelerated cash-pay uptake in aesthetic clinics across Brazil and Italy.
Advances in Ozone Generators & Topical Formulations
Next-generation devices incorporate UV photometers and millisecond feedback loops that hold dose within ±1.6% of target, lowering energy use by almost 50%. German and Italian vendors market CE-marked systems offering 1–80 µg/mL output and touch-screen presets.
Adoption Surge in EU & NA Dermatology Clinics
Germany, Italy, and Spain embed ozone therapy in routine dermatology under MDR 2017/745. In the United States, Oregon, Texas, and Florida allow clinical use with informed-consent disclosure despite FDA’s “toxic gas” stance. Regional legality, not technology, now dictates clinic rollout speed.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Regulatory variability & limited FDA approvals | -1.5% | U.S., Canada, Australia | Long term (≥4 years) |
| Lack of large-scale RCTs & reimbursement gaps | -1.3% | High-income markets with HTA processes | Medium term (2–4 years) |
| Oxidative-reaction liability at high doses | -0.4% | Global where training is limited | Short term (≤2 years) |
| U.S. import tariffs on components | -0.2% | United States | Short term (≤2 years) |
| Source: Mordor Intelligence | |||
Regulatory Variability & Limited FDA Approvals
The U.S. Food and Drug Administration's classification of ozone as a "toxic gas" with "no known useful medical application" under 21 CFR 801.415 prohibits marketing ozone-generating devices for therapeutic use without 510(k) clearance or premarket approval, effectively barring dermatologic ozone-device commercialization in the world's largest healthcare market. China's May 2026 biomedical new-technology regulations establish a clinical-research filing pathway and translation-approval process for novel cellular/molecular therapies, potentially including ozone-based interventions, but require multi-center safety/efficacy evidence, ethics review, and risk-tiered institutional restrictions (high-risk technologies limited to research-participating institutions for 5 years).
Scarcity of Large-Scale RCT Evidence & Reimbursement Gaps
Aetna's 2026 clinical policy bulletin explicitly classifies ozone therapy as experimental, investigational, or unproven due to inadequate peer-reviewed evidence of safety and effectiveness, citing health-technology assessments from Canada (CCOHTA), Spain (AETSA), and Denmark (DACEHTA) that found insufficient evidence for topical ozone in diabetic leg ulcers and other indications.[3]Aetna, “Clinical Policy Bulletin 0388,” aetna.com Cigna's September 2025 hyperbaric and topical oxygen policy covers topical oxygen therapy (not ozone) for diabetic foot ulcers that failed standard care, supported by the TWO2 randomized double-blind trial and American Diabetes Association Standards of Care 2025 (Evidence grade A), but does not extend coverage to ozone-based modalities.
Segment Analysis
By Product Type: Devices Lead, Medications Gain Momentum
Devices generated the largest revenue, contributing 58.42% to the ozone therapy in dermatology market in 2025. Precision-controlled generators from German suppliers underpin capital sales, while syringes, bags, and tubing drive recurring consumables. The medications segment is poised to grow at 7.52% CAGR through 2031 as ozonated oils and gels migrate into home-care kits. One third of clinics now retail peroxide-indexed oils for postoperative wounds, signaling new OTC channels.
Institutional buyers increasingly demand CE-marked systems with integrated UV photometry. Portable generators priced below USD 8,000 open access for solo aestheticians, but regulatory compliance remains a gating factor. Meanwhile, formulated topicals avoid device-training burdens and pass directly to consumers, explaining their higher growth trajectory within the wider ozone therapy in dermatology market.

By Treatment Method: Topical Dominates, Ozone Water Therapy Accelerates
Topical exposure retained 24.78% share in 2025 and remains the entry point for most dermatology practices. Standard limb-bagging at 35 µg/mL shortens the healing of diabetic-foot ulcers by two weeks in controlled cohorts. Ozone water therapy, already common in Japanese wound units, is projected to post the segment’s highest 7.25% CAGR to 2031. Device vendors now attach dissolving cartridges that create 4 ppm ozonated rinse within 30 seconds, easing use in busy clinics.
Systemic autohemotherapy and injection techniques together account for under 15% of revenue because most jurisdictions classify them as experimental. Nonetheless, pockets of adoption in Italy and select U.S. states keep training demand alive and sustain equipment upgrades inside the broader ozone therapy in dermatology market.
By Application: Wound Healing Anchors, Anti-Aging Surges
Wound healing delivered 19.02% of 2025 sales, underpinned by mounting meta-analytic evidence that ozone lowers amputation risk by 54% in diabetic-foot cohorts. Aesthetic rejuvenation is the fastest mover; hybrid ozone-PRP protocols elevated FACE-Q satisfaction scores by 30% in a 2024 Brazilian trial.
Smaller niches, such as acne, psoriasis, and herpes, together represent roughly one-fifth of 2025 revenue, but limited RCT data and dosing variability restrain payer adoption. Targeted trials using HOME-aligned outcomes could unlock their latent potential in the ozone therapy in dermatology market.

By End User: Dermatology Clinics Anchor, Aesthetic Clinics Accelerate
Dermatology clinics controlled 48.03% of 2025 turnover, reflecting entrenched wound-care workflows and physician oversight. Aesthetic clinics, however, will expand fastest at 9.16% CAGR on the back of elective revenue streams. Bundled offerings, ozone plus microneedling plus exosomes, let providers charge USD 800–1,200 per session without insurer friction, fueling share gains. Hospitals contribute modestly because FDA rules deter capital expenditure, though Brazilian public facilities have begun buying CE-marked European units after the 2025 authorization.
Home-care users and wellness centers form an emerging micro-segment. If portable, FDA-listed devices reach the market, they could materially enlarge the ozone therapy in the dermatology industry by 2031.
Geography Analysis
North America generated 41.83% of 2025 revenue. State health-freedom laws in Oregon, Texas, Nevada, Florida, and Arizona permit ozone under informed consent, permitting clinics to advertise despite the FDA’s national prohibition. Canada follows a similar integrative-medicine path but on a smaller scale. Mexico leverages medical tourism, offering packages 40% cheaper than U.S. equivalents.
Europe ranks second in value. Germany and Italy house most certified manufacturers and benefit from MDR-cleared equipment. Italian insurers such as Axa Caring reimburse topical ozone for diabetic foot ulcers when documented by wound-size photography. Eastern Europe, especially Russia and Poland, maintains historic acceptance and supplies cost-competitive systems to Latin America.
Asia-Pacific posts the fastest 8.06% CAGR. China’s May 2026 rules give universities a five-year window to run multi-center trials, paving the way for National-Health-Commission approval. Japan’s physician-led society promotes ozone-water irrigation and has issued bilingual protocols since 2025. India’s urban dermatology hubs adopt EU-made devices, but training gaps slow rural uptake.
South America hinges on Brazil’s October 2025 decision that authorised bagging and ozonated oil for diabetic-foot care. Public hospitals in São Paulo have started procurement pilots, suggesting rapid scale once local device factories receive ANVISA certificates. Middle East & Africa remain small yet strategic; Dubai wellness chains and South Africa’s private clinics court medical tourists and diaspora patients but lack domestic reimbursement.
Competitive Landscape
The ozone therapy in dermatology market remains moderately concentrated. Herrmann Apparatebau and Dr. J. Hänsler GmbH together supply most CE-marked generators, leveraging 60-plus years of R&D and ISO 13485 conformity. MEDOZONS dominates in Russia with ISO-certified portable units. In 2025, Italy’s Prometheus acquired Alnitec to bundle PRP automation with ozone generators, an early sign of convergence between regenerative medicine and ozone platforms.
U.S. distributors such as Promolife and Longevity Resources sell “research only” systems to bypass FDA marketing claims, while offering 40-hour training courses and liability waivers. Chinese manufacturers eye domestic clinical-trial pathways to gain share, but export potential depends on CE marking and IEC 60601 compliance. Future competition will center on AI-driven dose control, IoT-enabled compliance logging, and disposable-kit ecosystems that lock clinics into proprietary consumables.
Portable, home-use generators priced below USD 2,000 could open a consumer channel, yet safety certification and real-time ozone-monitor sensors will be essential to avoid oxidative injuries. Companies that secure both medical-grade AI dosing and consumer usability may capture outsized value inside the growing ozone therapy in dermatology market.
Ozone Therapy In Dermatology Industry Leaders
BionMed
Ozolabs, LLC
Herrmann Apparatebau GmbH
Promolife, Inc.
Phoenix Medical Systems
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- December 2025: Italy's Prometheus acquired Alnitec, a leading ozone therapy device company specializing in dermatological, orthopedic, and vascular applications, integrating ozone platforms with Prometheus's PRP automation systems (Ematik) to create bundled regenerative-medicine solutions for aesthetic and medical clinics.
- October 2025: Brazil's Federal Council of Medicine (CFM) issued Resolution CFM No. 2,445/2025, authorizing ozone therapy as an adjuvant medical treatment for diabetic foot ulcers, ischemic arterial ulcers, acute infectious wounds, chronic venous ulcers, knee osteoarthritis, and low back pain due to herniated disc, restricting approved application methods to sealed plastic bag technique (ozone bagging), ozonated oil, and ointment, while prohibiting systemic or injectable ozone applications.
Global Ozone Therapy In Dermatology Market Report Scope
The ozone therapy in dermatology market involves using medical-grade ozone produced from pure oxygen to treat skin disorders, wounds, and aging via antimicrobial, anti-inflammatory, and regenerative properties. It covers devices (generators, injectors) and consumables, addressing conditions like acne, eczema, psoriasis, and infections, often as a non-pharmacological, complementary treatment.
The ozone therapy in dermatology market report is segmented by product type, treatment method, application, end user, and geography. By product type, the market is segmented into devices [ozone generators, ozone syringes, ozone chambers/bags, ancillary accessories], medications [ozonated oils & creams, ozonated gels & lotions, injectable ozone solutions]. By treatment method, the market is segmented into topical ozone therapy, systemic [autohemotherapy], ozone injection therapy, ozone steam sauna/bagging, ozone water therapy, and insufflation therapy. By application, the market is segmented into wound healing, acneiform eruptions, psoriasis, herpes & viral dermatoses, fungal/mycotic infections, anti-aging/skin rejuvenation, eczema & atopic dermatitis, and other dermatological conditions. By end user, the market is segmented into dermatology clinics, hospitals, aesthetic & cosmetic clinics, and other end users. By geography, the market is segmented into North America, Europe, Asia-Pacific, Middle East & Africa, and South America. The market forecasts are provided in terms of value (USD).
| Devices | Ozone Generators |
| Ozone Syringes | |
| Ozone Chambers/Bags | |
| Ancillary Accessories | |
| Medications | Ozonated Oils & Creams |
| Ozonated Gels & Lotions | |
| Injectable Ozone Solutions |
| Topical Ozone Therapy |
| Systemic (Autohemotherapy) |
| Ozone Injection Therapy |
| Ozone Steam Sauna / Bagging |
| Ozone Water Therapy |
| Insufflation Therapy |
| Wound Healing |
| Acneiform Eruptions |
| Psoriasis |
| Herpes & Viral Dermatoses |
| Fungal / Mycotic Infections |
| Anti-Aging / Skin Rejuvenation |
| Eczema & Atopic Dermatitis |
| Other Dermatological Conditions |
| Dermatology Clinics |
| Hospitals |
| Aesthetic & Cosmetic Clinics |
| Other End Users (Home Healthcare Settings, Spas & Wellness Centers, etc.) |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Devices | Ozone Generators |
| Ozone Syringes | ||
| Ozone Chambers/Bags | ||
| Ancillary Accessories | ||
| Medications | Ozonated Oils & Creams | |
| Ozonated Gels & Lotions | ||
| Injectable Ozone Solutions | ||
| By Treatment Method | Topical Ozone Therapy | |
| Systemic (Autohemotherapy) | ||
| Ozone Injection Therapy | ||
| Ozone Steam Sauna / Bagging | ||
| Ozone Water Therapy | ||
| Insufflation Therapy | ||
| By Application | Wound Healing | |
| Acneiform Eruptions | ||
| Psoriasis | ||
| Herpes & Viral Dermatoses | ||
| Fungal / Mycotic Infections | ||
| Anti-Aging / Skin Rejuvenation | ||
| Eczema & Atopic Dermatitis | ||
| Other Dermatological Conditions | ||
| By End User | Dermatology Clinics | |
| Hospitals | ||
| Aesthetic & Cosmetic Clinics | ||
| Other End Users (Home Healthcare Settings, Spas & Wellness Centers, etc.) | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the current value of the ozone therapy in dermatology market?
The market generated USD 431.12 million in 2026 and is projected to reach USD 589.67 million by 2031.
Which segment holds the largest market share of ozone therapy in the dermatology market?
Device sales, mainly ozone generators, contributed 58.42% of global revenue in 2025.
Which region is growing fastest in this field?
Asia-Pacific is forecast to post an 8.06% CAGR through 2031, led by regulatory openings in China and Japan.
Are topical ozone products reimbursed by insurers?
Coverage is limited; most U.S. payers deem ozone experimental, though some Italian and Brazilian insurers reimburse specific wound-care uses.
What are the main barriers to wider adoption?
FDA classification of ozone as a toxic gas, lack of large multicenter RCTs, and variability in dosing standards remain primary hurdles.
Page last updated on:









