Hemodynamic Flow Alteration Devices Market Size and Share
Hemodynamic Flow Alteration Devices Market Analysis by Mordor Intelligence
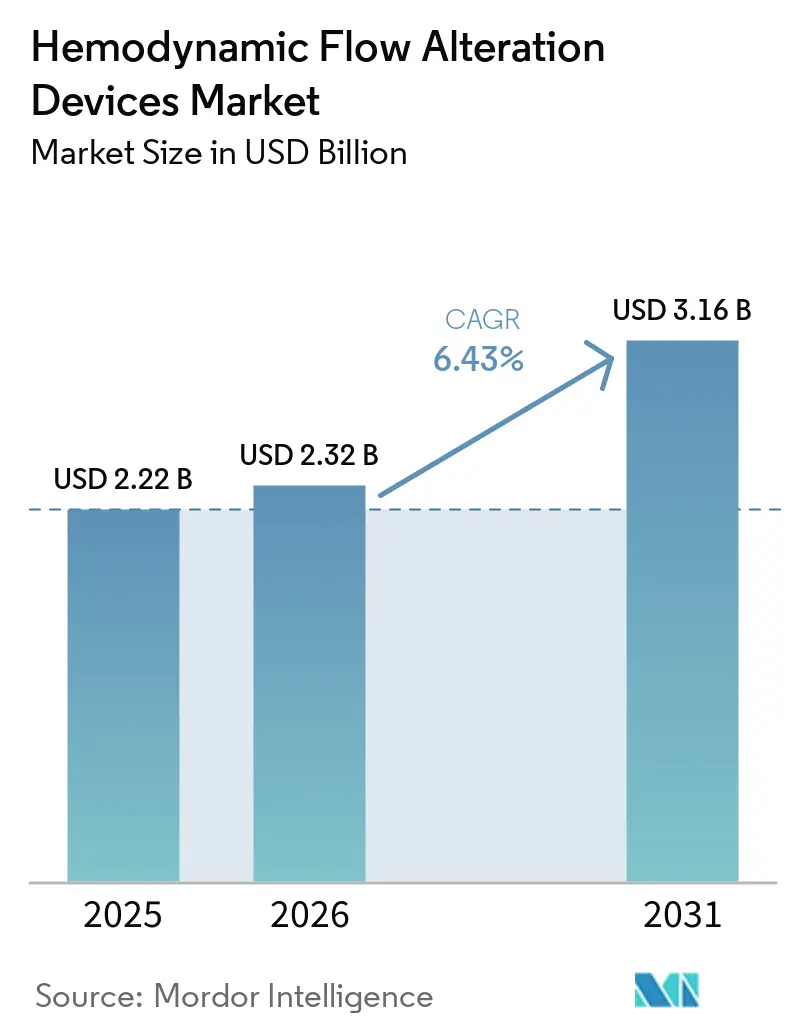
The Hemodynamic Flow Alteration Devices Market size is expected to increase from USD 2.22 billion in 2025 to USD 2.32 billion in 2026 and reach USD 3.16 billion by 2031, growing at a CAGR of 6.43% over 2026-2031.
Rising volumes of complex cardiovascular interventions and steady improvements in chronic total occlusion success rates are sustaining demand. Wider use of intravascular imaging, supported by clinical evidence, is improving procedural outcomes and reinforcing device adoption. The shift toward outpatient sites is reshaping purchasing decisions and accelerating throughput gains under structured quality frameworks. Headline evidence from large randomized and registry datasets is recalibrating the role of embolic protection in routine valve interventions, which is directing suppliers toward precision-indication strategies. Payment updates and coding clarifications are also shaping the pace and mix of adoption across care settings, influencing how health systems plan capital and disposable utilization in the hemodynamic flow alteration devices market.
Key Report Takeaways
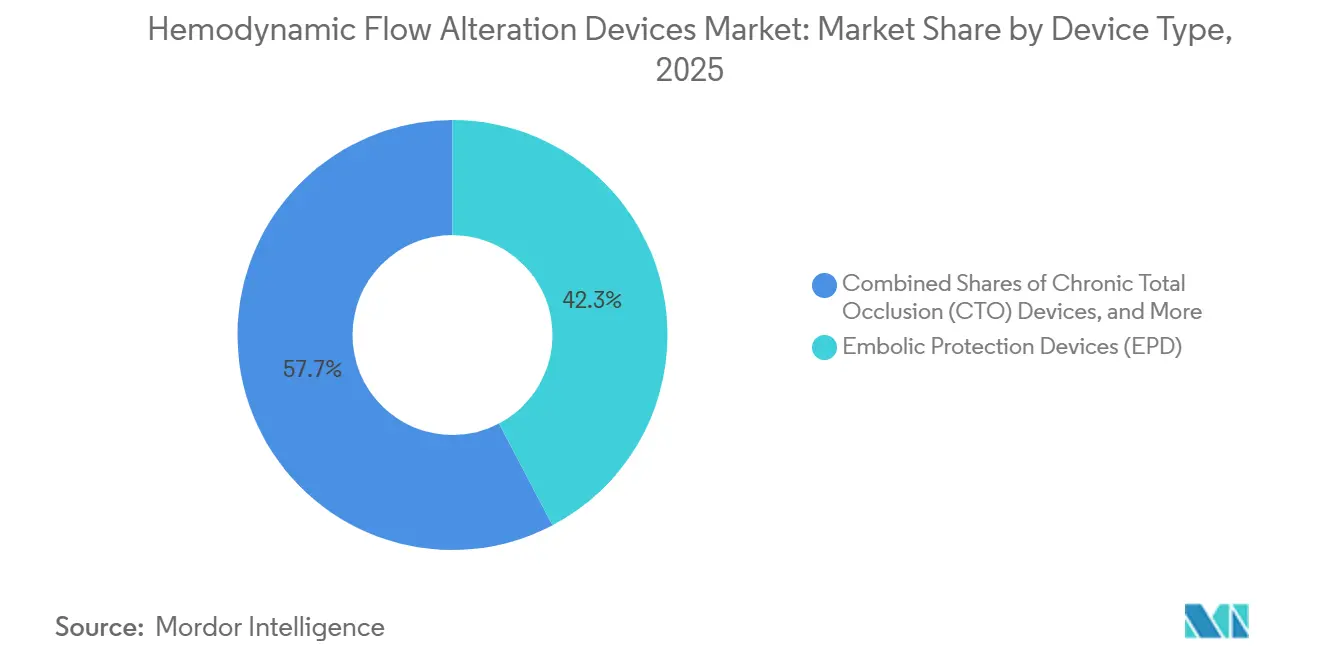
- By device type, Embolic Protection Devices led with 42.3% revenue share in 2025, while Chronic Total Occlusion devices are projected to expand at a 7.94% CAGR through 2031.
- By application, Coronary Interventions accounted for a 36.5% share in 2025, and Structural Heart applications are forecast to advance at a 10.37% CAGR to 2031.
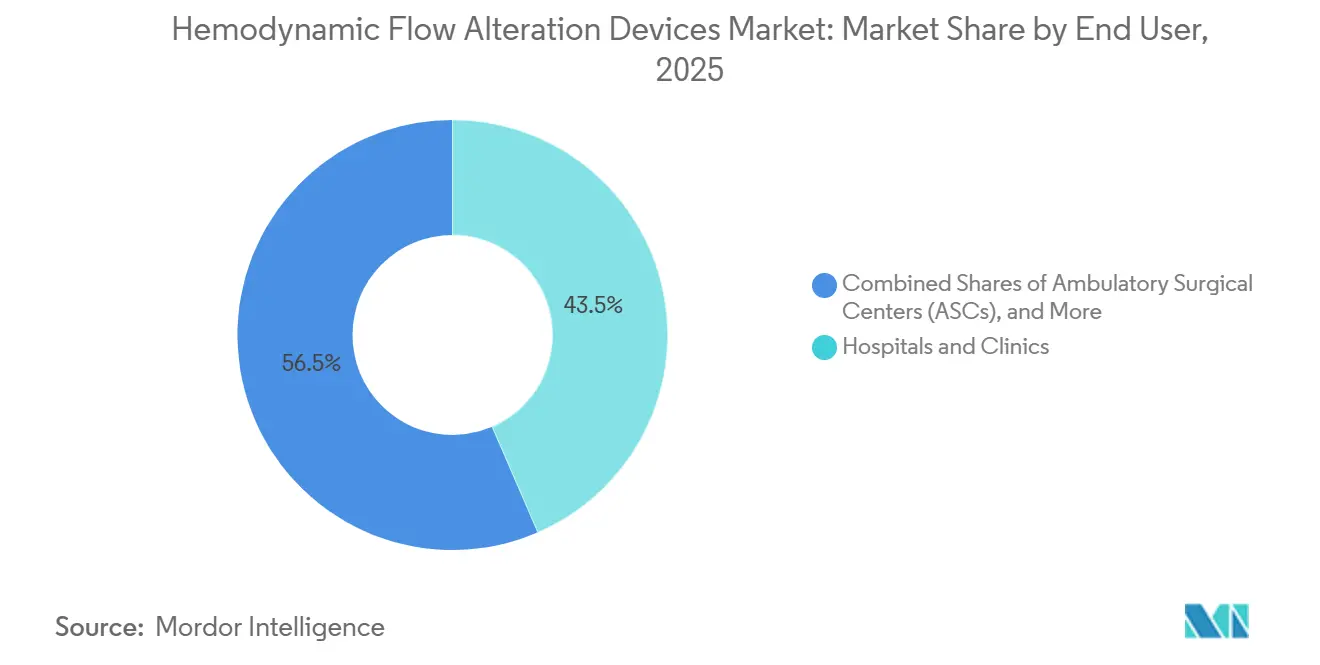
- By end user, Hospitals and Clinics held 43.5% share in 2025, and Ambulatory Surgical Centers are projected to post the fastest growth at a 7.58% CAGR through 2031.
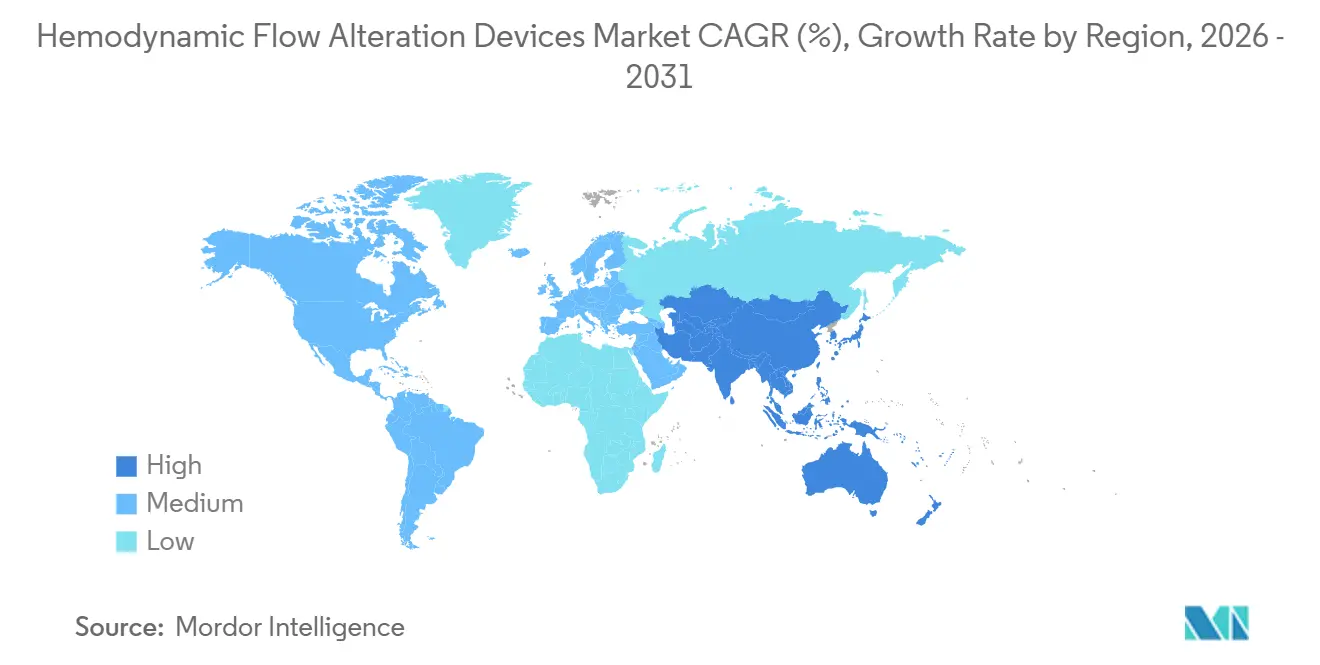
- By geography, North America led with 42.3% share in 2025, while Asia-Pacific is projected to record the fastest regional growth at a 9.57% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Hemodynamic Flow Alteration Devices Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising interventional procedure volumes in PCI, CAS, and TAVR | +1.8% | Global, led by North America and Asia-Pacific core | Medium term (2-4 years) |
| North American leadership and expanding reimbursement supporting adoption | +1.3% | North America, spill-over to EU regulatory harmonization | Short term (≤ 2 years) |
| Advancements in CTO crossing and re-entry systems raising success rates | +1.5% | Global, concentrated in high-volume tertiary centers | Medium term (2-4 years) |
| Aging populations and PAD/diabetes burden increasing occlusion cases | +1.2% | APAC (China, India), sub-Saharan Africa, North America | Long term (≥ 4 years) |
| Shift to outpatient cath labs and ASCs accelerating demand for protection and crossing tools | +0.9% | North America, early gains in Santiago, Valparaíso, Concepción | Medium term (2-4 years) |
| Portfolio bundling and recent acquisitions (e.g., TCAR) speeding integrated adoption | +0.7% | North America &and EU, emerging in Brazil, Mexico | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Rising Interventional Procedure Volumes in PCI, CAS, and TAVR
Technical success for chronic total occlusion percutaneous coronary intervention in contemporary European expert centers reached 89.1%, with low major adverse cardiac and cerebrovascular event rates, which shows how operator experience and standardized algorithms are improving outcomes in the hemodynamic flow alteration devices market[1]Giuseppe Vadalà et al., “Contemporary Outcomes of Chronic Total Occlusion Percutaneous Coronary Intervention in Europe, The ERCTO Registry,” EuroIntervention, eurointervention.pcronline.com. British Cardiovascular Intervention Society guidance sets minimum procedural volumes per center and per operator to maintain proficiency, aligning training and credentialing with safer and more efficient practice.
Intravascular imaging has been associated with materially lower failure and reintervention compared with angiography alone in CTO cohorts, which is helping to standardize imaging-guided approaches in high-complexity lesions. Carotid artery stenting demand is reinforced by transcarotid artery revascularization utilization in stroke prevention programs, where an FDA-cleared platform is now part of a larger vascular portfolio following a strategic acquisition, which is embedding carotid solutions more deeply in U.S. hospital contracting.
The convergence of growing CTO PCI, carotid interventions, and transcatheter valve procedures is broadening the procedural base that depends on protection and crossing tools across the hemodynamic flow alteration devices market. As high-volume centers disseminate best practices and proctor peers, adoption in mid-volume settings is expected to rise in measured steps, further expanding addressable volumes for suppliers in the hemodynamic flow alteration devices market.
North American Leadership and Expanding Reimbursement Supporting Adoption
Payment levels for endovascular valve replacement with or without major complications established a baseline for hospital reimbursement that private plans often reference, which supports continued program investment in devices and imaging that complement these procedures in the hemodynamic flow alteration devices market. National fee-for-service Medicare spending on ambulatory surgical center services increased with more ASCs operating and more procedures per beneficiary, which underscores a system-level shift to outpatient settings where protection and crossing tools are feasible within facility capabilities. CMS continued to update and clarify hospital outpatient and ASC payment rules and codes, including changes executed through quarterly program transmittals, which influence the timing and economic rationale for migrating eligible cases to outpatient environments. National coding and billing manuals also set expectations for special services and device-related reporting, which helps standardize administrative workflows and can reduce friction in the hemodynamic flow alteration devices market as hospitals and ASCs operationalize new procedure combinations.
Clinical societies and registries provide performance tracking and help define best practices that are adopted across U.S. networks, reinforcing procurement continuity for devices that align with evidence-backed protocols. As coverage criteria and site-of-service rules evolve under Medicare guidance, device makers that design for outpatient efficiency and compatible coding pathways are positioned to gain share in the hemodynamic flow alteration devices market.
Advancements in CTO Crossing and Re-Entry Systems Raising Success Rates
Imaging guidance reduced target lesion failure and reintervention compared with angiography alone in CTO cohorts, which supports broader use of intravascular ultrasound during complex PCI and bolsters demand for compatible crossing portfolios in the hemodynamic flow alteration devices market. European multicenter registry data documented high overall success and low complication rates under expert operators, with meaningful but variable imaging utilization influenced by reimbursement and access, showing room for standardized adoption. Wire escalation strategies increasingly rely on families with distinct tip and coating profiles, while soft polymeric wires begin most attempts and hydrophilic wires complete many crossings, reinforcing the importance of diverse, stepwise toolkits within labs. Dual-lumen microcatheters support parallel-wire techniques and retrograde navigation, and guiding catheter extensions improve reach and stability during subintimal dissection and re-entry, which together optimize efficiency in difficult anatomies.
Calcium modification options, including rotational atherectomy and intravascular lithotripsy, address subsets of highly calcified lesions where conventional escalation fails, with contemporary data indicating evolving modality preferences tied to safety and efficacy tradeoffs. This interplay of imaging, access tools, and plaque modification is lifting reproducibility in expert labs and setting a roadmap for diffusion to mid-volume centers across the hemodynamic flow alteration devices market.
Aging Populations and PAD/Diabetes Burden Increasing Occlusion Cases
Cardiovascular disease in the elderly remains a heavy burden even as age-standardized rates eased in some regions, which increases absolute case load as populations age and raises interventional demand in the hemodynamic flow alteration devices market. A systematic review across sub-Saharan Africa reported higher peripheral artery disease prevalence among adults with type 2 diabetes than high-income benchmarks, with older age and longer diabetes duration linked to greater risk, which signals a sustained need for revascularization capacity. A clinical cohort found that patients with diabetes and PAD faced higher tissue-loss rates and intense polypharmacy complexity, which complicates medical therapy and may tip decisions toward revascularization when anatomy permits.
Data from Asia showed the clustering of atherosclerotic risk factors among CTO patients, including high rates of diabetes and hypertension, which suggests continued growth in complex coronary and peripheral interventions. As life expectancy rises and metabolic disease remains prevalent, caseloads that require specialized crossing and protection tools are likely to expand in regions scaling catheterization infrastructure within the hemodynamic flow alteration devices market. Health systems will continue to balance screening and prevention with investment in interventional capacity, given the clinical and economic impact of advanced atherosclerotic disease.
Restraints Impact Analysis
| Restraint (as applicable in title case) | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Neutral primary endpoint in protected TAVR dampens routine CEP uptake | -0.8% | Global, pronounced in evidence-based EU and U.S. markets | Short term (≤ 2 years) |
| Device costs and reimbursement variability constrain usage | -0.6% | APAC (excluding Japan), Latin America, MEA | Medium term (2-4 years) |
| Product safety notices and recalls increase caution | -0.3% | Global, heightened scrutiny in FDA and EMA jurisdictions | Short term (≤ 2 years) |
| Steep learning curve and longer procedure times limit CTO device diffusion beyond referral centers | -0.4% | National, concentrated in Tier-2/ and Tier-3 hospitals globally | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Device Costs and Reimbursement Variability Constrain Usage
Intravascular ultrasound use in CTO interventions varied widely across European centers, reflecting national reimbursement heterogeneity and access constraints, and leaving meaningful room for increased adoption even in advanced programs within the hemodynamic flow alteration devices market. Authors of contemporary CTO algorithms have also noted that procedural time, resource intensity, and modest professional payment can discourage routine attempts at more complex lesions in mid-volume centers, which slows diffusion outside of expert hubs. National outpatient and ASC payment rules continue to evolve through CMS program updates and transmittals, which may change site-of-service incentives and affect how providers budget for advanced imaging and crossing tools.
The scale and direction of these payment updates interact with capital budgets, staffing models, and device contracting, creating variation in adoption within and across markets in the hemodynamic flow alteration devices market. Manufacturers adapt through clinical education, value analyses, and portfolio options, but variability in local coverage and purchasing power still slows the pace of standardization. Over time, convergence in policy and continued clinical evidence can narrow these gaps and support steadier adoption.
Product Safety Notices and Recalls Increase Caution
A Class I safety notice for a braided embolization device described increased risks of braid deformation and incomplete wall apposition in certain configurations, with specific subgroups at higher relative risk, which prompted product withdrawal actions and updated instructions across related devices. A separate Class I recall of intra-aortic balloon catheter kits was issued after reports of device malfunction, injuries, and deaths, with regulatory guidance advising device inspection, backup availability, imaging confirmation during use, and prompt replacement upon alarms, which elevated procedural vigilance in critical-care settings that overlap with interventional workflows[4]U.S. Food and Drug Administration, “Teleflex/Arrow International Recall Arrow FiberOptix and UltraFlex Intra-Aortic Balloon Catheter Kits,” FDA, fda.gov. An additional recall affecting a venous clot retrieval system included revisions to indications and warnings, highlighting entrapment and embolic risks in certain thrombus types, and raised caution around device selection and case planning in complex venous interventions.
Isolated reports in federal adverse-event databases have also documented device migration without patient sequelae in specific procedures, which keeps hospital value-analysis committees focused on failure modes and post-market surveillance plans before formulary expansion. Heightened scrutiny redirects suppliers toward user training, case-selection guidance, and targeted updates to instructions for use, all of which add operational steps that can temper early adoption in the hemodynamic flow alteration devices market. Over time, corrective actions and follow-up evidence can restore physician confidence, but the near-term effect is more careful institutional review.
Segment Analysis
By Device Type: EPDs Lead Share, CTO Devices Accelerate on Imaging Integration
Embolic Protection Devices accounted for 42.3% of device-type revenue in 2025, with carotid interventions driving steady use of distal filters, and with transcatheter workflows incorporating tailored approaches to debris capture across anatomies in the hemodynamic flow alteration devices market. EPDs accounted for a 42.3% share of the hemodynamic flow alteration devices market size in 2025, and physicians have reassessed routine use in valve procedures after randomized trial data showed no significant reduction in stroke with routine cerebral embolic protection during TAVR, which is encouraging more selective deployment for higher-risk patients.
As a result, suppliers are investing in clearer indication guidance and refined device designs rather than blanket-use positioning in the hemodynamic flow alteration devices market. At the same time, professional society reporting and registry infrastructure maintain attention on stroke endpoints, which anchors physician decision making around evidence and safety. Ongoing procedural growth in carotid and select structural settings sustains a base of EPD usage, while the precision-indication pattern is reshaping purchasing norms within integrated vascular programs across the hemodynamic flow alteration devices market.
Chronic Total Occlusion devices are projected to be the fastest-growing device category at a 7.94% CAGR, supported by imaging-guided strategies that have been linked with lower failure and reintervention compared with angiography alone, which underpins more routine imaging in difficult occlusions. Registry data covering thousands of procedures across expert operators reported high overall technical success near 90% with low event rates, helping codify the role of specialized guidewires, microcatheters, re-entry systems, and plaque-modification options in reproducible workflows in the hemodynamic flow alteration devices industry. Escalation patterns that start with polymer-coated wires and finish with hydrophilic wires were prominent. At the same time, dual-lumen microcatheters enabled parallel-wire techniques and retrograde navigation in selected anatomies, which supports the value of comprehensive crossing portfolios. Guiding catheter extensions also improved support during antegrade dissection and re-entry maneuvers, boosting efficiency in long or calcified segments. Contemporary analyses have described shifts toward intravascular lithotripsy for heavy calcium in some settings, reflecting perceived advantages in safety endpoints, while rotational systems remain staples in specific lesion subsets. Together, these developments signal durable momentum for CTO portfolios within the hemodynamic flow alteration devices market.

By Application: Structural Heart Surges, Coronary Procedures Stabilize
Coronary Interventions held 36.5% share in 2025, supported by robust primary PCI programs and dedicated CTO services that operate under defined volume and training standards across major health systems in the hemodynamic flow alteration devices market. Coronary Interventions held 36.5% of the hemodynamic flow alteration devices market share in 2025, and guideline-backed case thresholds continue to concentrate complex work in high-volume centers with skills and devices to manage long, calcified, and tortuous lesions. European registry findings substantiate high CTO success and low event rates in expert networks, which provide a template for diffusion to mid-volume centers and support steady use of crossing and imaging tools. As systems standardize CTO pathways and training, imaging and crossing toolkits become routine in advanced coronary programs across the hemodynamic flow alteration devices market. Improvements in upstream risk management and emergency networks also sustain stable primary PCI volumes, which preserve a strong base of demand in this application segment.
Structural Heart applications are projected to grow at a 10.37% CAGR on the strength of expanding transcatheter valve programs and measured improvements in outcomes as centers scale their experience in the hemodynamic flow alteration devices market. The hemodynamic flow alteration devices market size for Structural Heart is projected to expand at 10.37% CAGR between 2026 and 2031, while high-volume surgical programs have been associated with lower in-hospital mortality for transcatheter aortic valve replacement, reinforcing the link between surgical backup, learning curves, and transcatheter outcomes. Large randomized evidence and national registry updates reported no significant benefit for routine cerebral embolic protection in all-comer TAVR populations, which has tilted adoption toward targeted subgroups where risk-benefit analysis is favorable.[3]Rajesh K. Kharbanda et al., “Routine Cerebral Embolic Protection During Transcatheter Aortic-Valve Implantation,” New England Journal of Medicine, nejm.orgEven with routine-use caution, the continued maturation of transcatheter programs sustains demand for crossing and adjunct tools in complex valve cases and concomitant coronary. In parallel, peripheral arterial procedures remain an avenue of growth as diabetes and PAD burdens rise, and as centers strengthen below-the-knee and multivessel limb-salvage pathways supported by imaging and specialized devices.
By End User: ASCs Gain Ground, Hospitals Retain Bulk
Hospitals and Clinics held 43.5% end-user share in 2025, reflecting the concentration of structural heart, complex coronary, and carotid interventions that require surgical backup and 24-hour support in the hemodynamic flow alteration devices market. Institutions with higher surgical valve volumes have reported lower in-hospital mortality during TAVR, which supports routing complex cases to centers with full-service cardiothoracic infrastructure and adds durable demand for advanced protection and crossing tools. National society guidance on minimum case volumes and training expectations further concentrates complex work in referral hospitals, where imaging and device portfolios are standard in cath labs and hybrid ORs. Same-day discharge in selected CTO cohorts has increased under defined criteria, which fits hospital efforts to improve throughput and align with value-based care frameworks in the hemodynamic flow alteration devices market.
Ambulatory Surgical Centers are projected to post the fastest end-user growth at a 7.58% CAGR as Medicare-certified ASC capacity expands and more procedures per beneficiary shift to outpatient sites, which strengthens the commercial outlook for tools compatible with these settings in the hemodynamic flow alteration devices industry. Medicare data show year-on-year growth in ASC facilities and fee-for-service spending, which confirms the gradual migration of eligible interventions to lower-acuity locations under national policy and coding frameworks. CMS quarterly updates to hospital outpatient and ASC payment schedules continue to refine eligible services and payment details, which supports procedural planning and coding accuracy across outpatient locations.
Independent outpatient cath labs also benefit from clearer billing guidance in national manuals for special services, which eases administrative adoption as case complexity rises. Specialty cardiac centers and academic facilities contribute smaller volumes but exert strong influence on technique standardization and training that further supports adoption in hospitals and ASCs. Across end users, supply chains and education models are adjusting to meet growth outside inpatient hospitals, reinforcing demand diversity in the hemodynamic flow alteration devices market.
Geography Analysis
North America held 42.3% of the hemodynamic flow alteration devices market share in 2025, supported by reimbursement baselines for endovascular valve procedures and coding clarity for cerebral protection that help align hospital financial planning with device adoption. For example, payment values under MS-DRG 266 and 267 created reference points for program budgeting in structural heart care, while add-on coding clarified physician reporting for protection in TAVR when used. Acquisitions that add carotid platforms into major portfolios have widened access to transcarotid solutions and integrated them into hospital contracting structures across stroke-prevention pathways.[2]Boston Scientific Corporation, “Boston Scientific Closes Acquisition of Silk Road Medical, Inc.,” PR Newswire, prnewswire.com National guidance on procedural volumes and training continues to support concentration of complex coronary work in higher-volume settings, which sustains imaging and crossing utilization in referral centers. These dynamics underpin steady demand for protection and CTO toolkits across a mature provider base in the hemodynamic flow alteration devices market.
Asia-Pacific is projected to post the fastest regional growth at a 9.57% CAGR through 2031, with China’s expanding TAVR programs illustrating a clear learning-curve effect and safety gains as experience increases in the Hemodynamic Flow Alteration Devices market. National registry-level data reported more than twelve thousand TAVR procedures across hundreds of hospitals, with in-hospital mortality falling as centers accumulated cases, and with higher surgical valve volumes associated with lower TAVR mortality. Regional case mix also includes a high burden of metabolic disease, and CTO cohorts in South Asia show concentrated risk profiles with diabetes and hypertension that necessitate revascularization in complex anatomies. Japan’s emphasis on intravascular imaging and operator training supports higher imaging adoption during complex PCI than in many European settings, which reinforces procedural quality and device penetration in advanced labs. With tertiary networks scaling capacity and training, adoption of CTO and protection portfolios continues to expand across the hemodynamic flow alteration devices market in Asia-Pacific.
Europe, the Middle East and Africa, and South America show mixed but improving readiness, as leading centers align with volume and training standards and as hospitals weigh expanded outpatient delivery models within local coverage frameworks in the hemodynamic flow alteration devices market. National clinical audits and professional society guidance help unify practice and maintain focus on outcomes, which encourages consistent imaging and crossing protocols in complex coronary cases. Currency volatility and import tariffs in some markets affect pricing and procurement timetables, which adds friction to standardized adoption but does not alter long-run clinical need. Government safety notices and regulatory expectations across the EU and other regions also shape deployment decisions and training priorities around embolization and protection devices. As health systems consolidate experience and clarify local coding, a broader base of providers is positioned to adopt the hemodynamic flow alteration devices market toolkits in a measured and sustainable way.
Competitive Landscape
The hemodynamic flow alteration devices market features moderate consolidation at the top, alongside meaningful specialization in CTO and carotid platforms that has preserved opportunities for focused manufacturers. A prominent transaction announced in January 2026 will add a large thrombectomy and peripheral embolization portfolio to an established structural heart and coronary leader, which enables bundling strategies across service lines and broader reach into neurovascular and peripheral sites of care. This builds on a 2024 acquisition that integrated the only FDA-cleared transcarotid artery revascularization platform into the same corporate portfolio, creating a comprehensive vascular offering that simplifies contracting and cross-training. Portfolio breadth and contracting leverage are likely to shape account strategies as providers standardize technologies across coronary, structural, and carotid pathways in the hemodynamic flow alteration devices market.
Innovation agendas remain active around imaging-enhanced complex coronary work and calcium modification. A multinational trial program for a new intravascular lithotripsy system in coronary disease was launched in 2025, signaling a more competitive pipeline following a large acquisition in calcium modification that changed the market structure and accelerated rival development timelines. Registry and randomized data continue to refine the perceived tradeoffs among rotational and lithotripsy options in calcified CTO lesions, guiding operator choice and training investments that directly impact tool utilization in the hemodynamic flow alteration devices market. Suppliers with strong education programs and cross-modality portfolios are better positioned to capture share as labs align on chronic total occlusion algorithms that integrate imaging, access tools, and calcium modification.
Safety surveillance and regulatory scope influence competitive postures as well. Field safety notices and recall actions prompted manufacturers to execute product withdrawals or update instructions for use, with downstream effects on hospital committees and purchasing cycles in the hemodynamic flow alteration devices market. Government recall notices with detailed recommendations have become a core reference for provider policies and procedural checks, which increases the value of robust training, clear indications, and transparent post-market plans. Over time, companies that integrate safety feedback quickly and support providers with practical mitigation steps can protect brand equity while sustaining adoption in the hemodynamic flow alteration devices market.
Hemodynamic Flow Alteration Devices Industry Leaders
Abbott
ASAHI INTECC CO., LTD.
Boston Scientific Corporation
Terumo Corporation
Medtronic
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: Boston Scientific announced a definitive agreement to acquire Penumbra, Inc. for approximately USD 14.5 billion enterprise value to integrate mechanical thrombectomy systems and peripheral embolization devices.
- September 2024: Boston Scientific completed its acquisition of Silk Road Medical, Inc. for approximately USD 1.18 billion enterprise value, integrating the only FDA-cleared transcarotid artery revascularization platform into its vascular portfolio.
Global Hemodynamic Flow Alteration Devices Market Report Scope
As per the scope of the market, hemodynamic flow alteration devices are medical devices designed to modify, redirect, or control blood flow within blood vessels to improve the effectiveness and safety of endovascular and cardiovascular procedures. These devices work by altering vascular flow dynamics, stabilizing lesions, preventing embolic events, or enabling vessel recanalization during minimally invasive interventions. They are commonly used in coronary, peripheral, carotid, neurovascular, and structural heart procedures and are typically deployed via catheter‑based techniques under medical supervision.
The Hemodynamic Flow Alteration Devices Market Report segments the market by device type, including embolic protection devices that comprise distal filter, proximal occlusion, distal occlusion, cerebral EPD for TAVR; chronic total occlusion devices that comprise guidewires, microcatheters, crossing catheters, re-entry systems, laser/atherectomy; and others. It also categorized the market in terms of application, which includes coronary interventions, structural heart disease, carotid artery stenting, peripheral arterial interventions, and others. Along with this, the market is segmented into end users such as hospitals & clinics, ASCs, outpatient/independent cath labs, specialty cardiac centers, and others. Geographically, the market is segmented into North America, Europe, Asia-Pacific, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Embolic Protection Devices (EPD) | Distal Filter Systems |
| Proximal Occlusion Systems | |
| Distal Occlusion Systems | |
| Cerebral EPD for TAVR | |
| Chronic Total Occlusion (CTO) Devices | CTO Guidewires |
| Microcatheters | |
| Crossing Catheters | |
| Re-entry Systems | |
| Laser/Atherectomy & Powered Crossing | |
| Others (Flow Diverters, Venous Stents) |
| Coronary Interventions |
| Structural Heart Protection |
| Carotid Artery Stenting |
| Peripheral Arterial Interventions |
| Others (Neurovascular Interventions, Renal & Visceral Artery Interventions, etc.) |
| Hospitals & Clinics |
| Ambulatory Surgical Centers (ASCs) |
| Outpatient/Independent Cath Labs |
| Specialty Cardiac Centers |
| Others ( Research Institutes & Clinical Trial Centers, Academic Institutes) |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Device Type | Embolic Protection Devices (EPD) | Distal Filter Systems |
| Proximal Occlusion Systems | ||
| Distal Occlusion Systems | ||
| Cerebral EPD for TAVR | ||
| Chronic Total Occlusion (CTO) Devices | CTO Guidewires | |
| Microcatheters | ||
| Crossing Catheters | ||
| Re-entry Systems | ||
| Laser/Atherectomy & Powered Crossing | ||
| Others (Flow Diverters, Venous Stents) | ||
| By Application | Coronary Interventions | |
| Structural Heart Protection | ||
| Carotid Artery Stenting | ||
| Peripheral Arterial Interventions | ||
| Others (Neurovascular Interventions, Renal & Visceral Artery Interventions, etc.) | ||
| By End User | Hospitals & Clinics | |
| Ambulatory Surgical Centers (ASCs) | ||
| Outpatient/Independent Cath Labs | ||
| Specialty Cardiac Centers | ||
| Others ( Research Institutes & Clinical Trial Centers, Academic Institutes) | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the Hemodynamic Flow Alteration Devices market size outlook through 2031?
The Hemodynamic Flow Alteration Devices market size is expected to increase from USD 2.22 billion in 2025 to USD 2.32 billion in 2026 and reach USD 3.16 billion by 2031 at a 6.43% CAGR.
Which applications are growing fastest within the Hemodynamic Flow Alteration Devices market?
Structural Heart is projected to grow at a 10.37% CAGR through 2031 as transcatheter valve programs scale with improving outcomes and refined patient selection.
Which device category leads revenue and which grows fastest in the Hemodynamic Flow Alteration Devices market?
Embolic Protection Devices led with 42.3% revenue share in 2025, while Chronic Total Occlusion devices are projected to expand at a 7.94% CAGR.
Which region leads and which is the fastest growing for the Hemodynamic Flow Alteration Devices market?
North America led with 42.3% share in 2025, while Asia-Pacific is projected to post the fastest growth at a 9.57% CAGR through 2031.
How is reimbursement shaping adoption in the Hemodynamic Flow Alteration Devices market?
Medicare payment baselines for structural heart procedures and ongoing outpatient and ASC coding updates are supporting adoption and the shift of eligible cases to lower-acuity settings.
Page last updated on:









