Generic Injectable Market Size and Share
Generic Injectable Market Analysis by Mordor Intelligence
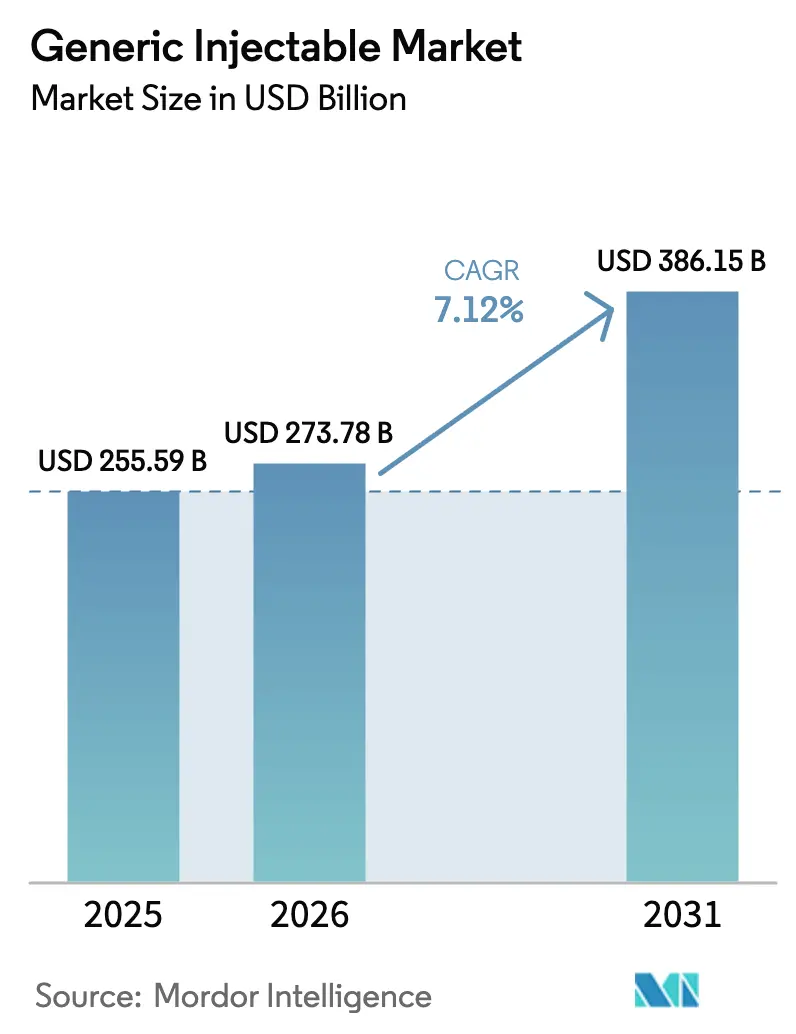
The Generic Injectable Market was valued at USD 255.59 billion in 2025 and expected to grow from USD 273.78 billion in 2026 to reach USD 386.15 billion by 2031, at a CAGR of 7.12% during the forecast period (2026-2031).
Patent expirations on high-revenue biologics, expanding chronic disease prevalence, and hospital drug-shortage mitigation programs jointly underpin this expansion. Supply-chain localization incentives in Asia and the fast-maturing biosimilar approval landscape further accelerate capacity additions, while artificial-intelligence process controls are beginning to improve sterile yield and shorten release cycles. Group purchasing organizations, however, continue to press prices downward, reshaping profitability bands for mid-tier suppliers. Against this backdrop, companies with scale, quality credentials, and flexible fill-finish assets are positioned to capture outsized shares of upcoming biologics conversions.
Key Report Takeaways
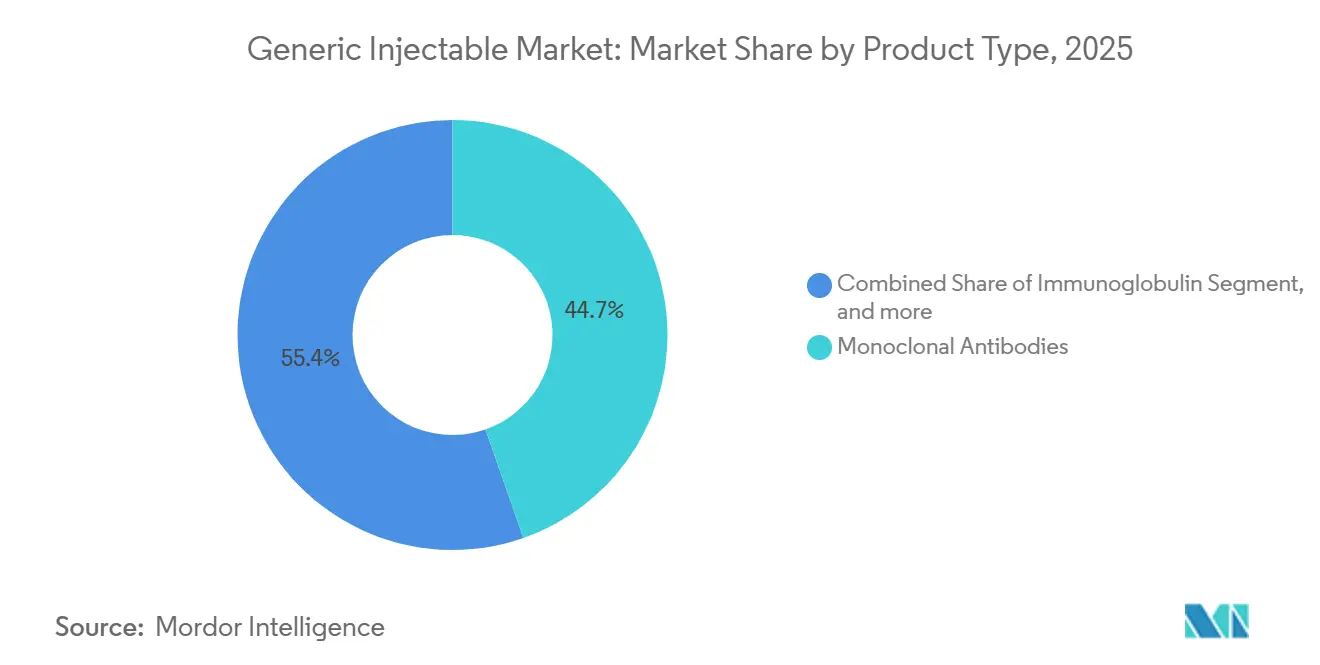
- By product type, monoclonal antibodies secured 39.4% of generic injectable market share in 2025, while immunoglobulin formulations are set to grow at a 9.54% CAGR through 2031.
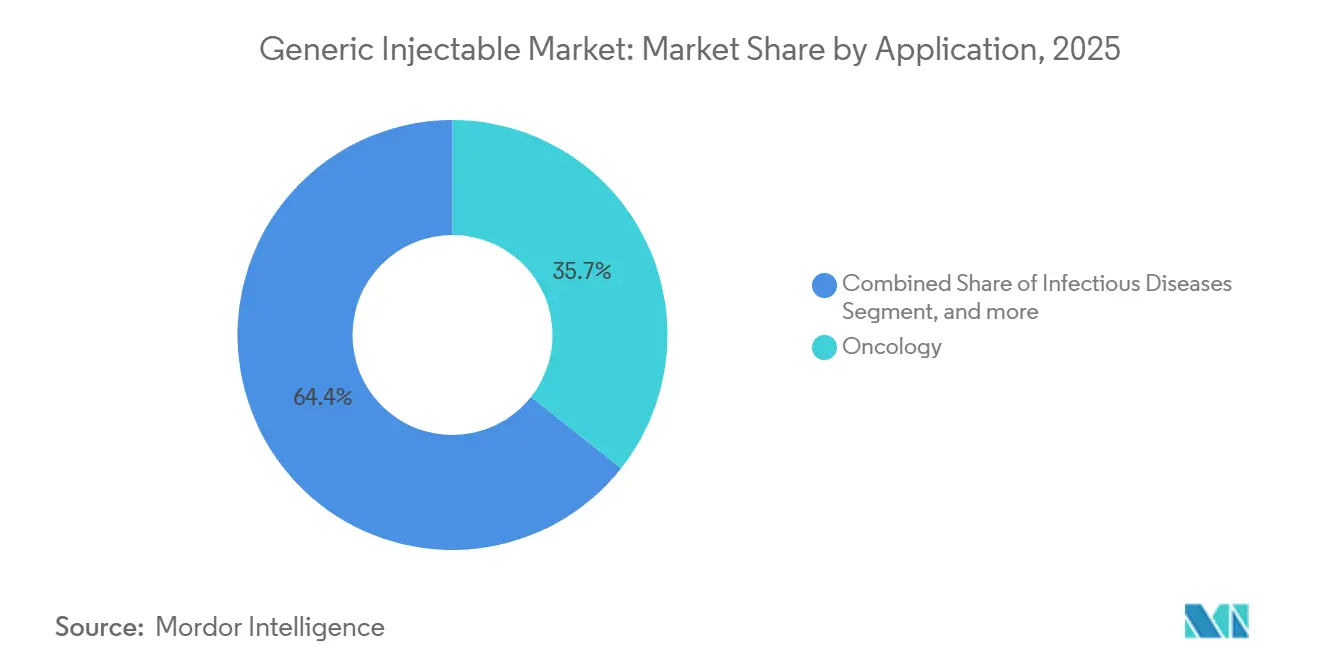
- By application, oncology dominated revenue with a 35.65% share in 2025, yet diabetes management injectables are forecast to expand at a 10.55% CAGR over the same period.
- By distribution channel, hospital pharmacies accounted for 53.23% of the generic injectable market size in 2025, whereas online pharmacies posted the fastest growth at 10.43% CAGR to 2031.
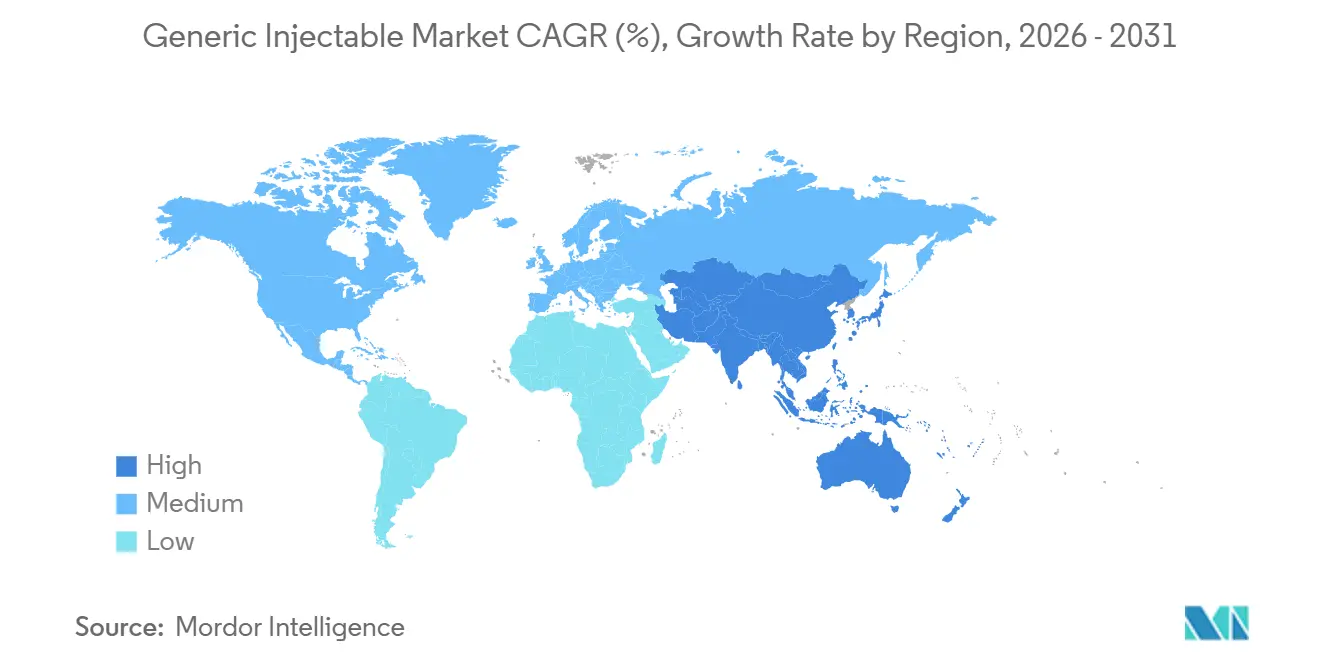
- By geography, North America led with a 44.76% share of global revenue in 2025, while Asia-Pacific is projected to grow at an 8.54% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Generic Injectable Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Escalating Global Burden of Chronic Diseases | +1.2% | North America, Europe, urban Asia-Pacific | Long term (≥ 4 years) |
| Large Patent Cliff for Biologics | +1.8% | North America and Europe | Medium term (2–4 years) |
| Cost-Containment Policies Favoring Generics | +1.1% | Europe, Latin America, Middle East & Africa | Medium term (2–4 years) |
| Supply-Chain Localization Incentives | +0.9% | Asia-Pacific core, spill-over to Middle East & South America | Long term (≥ 4 years) |
| Hospital Drug-Shortage Response | +0.7% | North America, emerging in Europe | Short term (≤ 2 years) |
| AI-Driven Sterile-Process Analytics | +0.5% | North America and Europe, pilots in Asia-Pacific | Medium term (2–4 years) |
| Source: Mordor Intelligence | |||
Escalating Global Burden of Chronic Diseases
Noncommunicable diseases caused 41 million deaths worldwide in 2024, with cardiovascular disorders representing 17.9 million fatalities. Cancer diagnoses climbed to 20 million cases, boosting demand for chemotherapy injectables and supportive biologics. Diabetes prevalence reached 537 million adults and is projected to hit 643 million by 2030, sustaining volume growth for insulin analogs and GLP-1 receptor agonist biosimilars[1]International Diabetes Federation, “IDF Diabetes Atlas 2025,” idf.org. Payers are favoring lower-cost generics for chronic therapy lines as evidence on biosimilar interchangeability deepens. These epidemiological realities magnify baseline consumption, thereby contributing 1.2 percentage points to forecast CAGR.
Large Patent Cliff for Biologics
Biologics with originator sales exceeding USD 100 billion will lose protection between 2024 and 2028. Eight adalimumab biosimilars seized 35% U.S. volume share within 18 months of Humira’s loss of exclusivity. Complex generics such as liposomal doxorubicin also gained approvals, broadening hospital formularies. Oncology and immunology show the sharpest cliff effects, catalyzing brisk uptake of cost-effective alternatives. The resulting competitive realignment is adding 1.8 percentage points to generic injectable market CAGR.
Cost-Containment Policies Favoring Generics
The European Medicines Agency shortened biosimilar review timelines by three months in 2024[2]European Medicines Agency, “Biosimilar Medicines: Regulatory Overview,” ema.europa.eu. Germany introduced automatic substitution for biosimilars in early 2025, while Brazil’s centralized tenders awarded BRL 2.1 billion (USD 420 million) for oncology and insulin injectables in 2025. U.S. Medicare price negotiations commence in 2026, amplifying the pricing gap between branded and generic injectables. These policy levers accelerate adoption but compress margins, collectively lifting CAGR by 1.1 percentage points.
Supply-Chain Localization Incentives
India’s Production Linked Incentive scheme disbursed INR 150 billion (USD 1.8 billion) through 2025 to spur fill-finish capacity. China approved 12 new sterile plants with annual capacity topping 500 million vials under its 14th Five-Year Plan. Saudi Arabia followed with SAR 3.2 billion (USD 850 million) commitments for injectable factories. Localization shortens supply lanes and builds regional resilience, adding 0.9 percentage points to forecast CAGR.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Intensifying Price Erosion From Tendering and GPO Consolidation | -1.3% | Europe, Latin America, Middle East & Africa; moderate in North America | Medium term (2–4 years) |
| Stringent Sterility and Regulatory Compliance Costs | -0.9% | Global, highest burden in North America and Europe | Long term (≥ 4 years) |
| Volatile Supply of Type-I Glass and COP Polymer Components | -0.6% | Global, acute in North America and Europe | Short term (≤ 2 years) |
| Skilled Workforce Shortage in Aseptic Manufacturing Operations | -0.4% | North America and Europe, rising in Asia-Pacific | Medium term (2–4 years) |
| Source: Mordor Intelligence | |||
Intensifying Price Erosion from Tendering and GPO Consolidation
Germany’s 2024 biosimilar adalimumab tender closed 72% below originator pricing, and Brazil’s oncology injectable averages fell 28% between 2023 and 2025. Consolidated U.S. GPOs negotiate 15–25% discounts on high-volume hospital injectables, prompting Teva to exit 14 low-margin products in 2024. Persistent deflation narrows profitability and deters pipeline investment, trimming CAGR by 1.3 percentage points.
Stringent Sterility and Regulatory Compliance Costs
The FDA performed 412 foreign sterile inspections in 2024 and issued 67 warning letters over aseptic deficiencies. EMA’s Annex 1 revision forced cleanroom retrofits costing EUR 5–20 million per plant. ISPE logged a 12% vacancy rate for sterile technicians in 2025, lengthening order-to-cash cycles[3]International Society for Pharmaceutical Engineering, “2025 North America Sterile Workforce Survey,” ispe.org. Smaller firms struggle to amortize these expenses, shaving 0.9 percentage points from market CAGR.
Segment Analysis
By Product Type: Immunoglobulin Leads Growth Trajectory
Immunoglobulin posted a 9.54% CAGR, eclipsing the 7.12% baseline for the generic injectable market as autoimmune diagnoses rise and biosimilar IVIG candidates secure approvals. Monoclonal antibodies remain the largest revenue block, buoyed by mature biosimilars of adalimumab, rituximab, and trastuzumab. Yet their expansion moderates as payer formularies saturate and focus pivots toward next-generation assets. Cytokines hold steady demand in oncology support despite encroachment from oral agents. Peptide hormones, such as somatropin, sustain niche growth despite high technical barriers.
Regulators approved the first biosimilar IVIG in 2024 and two more in 2025, signaling the maturity of the signaling pathway. Octapharma and Takeda raised plasma collection and added subcutaneous formats to defend their share. Supply chain bottlenecks in plasma, however, may temper immediate volume acceleration. Overall, immunoglobulin’s outsized trajectory positions it as the pivotal contributor to incremental growth in the generic injectable market over the forecast window.

By Application: Diabetes Management Accelerates
Diabetes injectables are projected at 10.55% CAGR, the fastest among applications, on the back of rising incidence and biosimilar insulin uptake. Oncology continued to command 35.65% revenue share in 2025, but growth normalizes as regimen cycling stabilizes. Cardiovascular anticoagulants keep hospital demand resilient, yet oral competitors chip away at chronic prescriptions. Infectious disease injectables stay relevant for severe bacterial and viral cases.
The U.S. launch of biosimilar insulin glargine pens by Viatris-Biocon in 2024 widened access and lowered costs. Generic GLP-1 receptor agonists scheduled post-2027 patents promise another step-change in affordability. Large incumbents Novo Nordisk and Eli Lilly reported 12% and 9% erosion in U.S. insulin revenue, respectively, in 2024, confirming competitive pressure. Consequently, diabetes therapies will deliver the greatest absolute gain in the generic injectable market size through 2031.

By Distribution Channel: Hospital Pharmacies Anchor Market
Hospital pharmacies provided 53.23% of 2025 throughput, a position unlikely to shift materially given their role in acute and infusion services. Group purchasing agreements and embedded clinical pathways cement their dominance, especially in oncology, critical care, and parenteral nutrition. Retail outlets dispense stabilized pre-filled syringes, but cold-chain and training hurdles limit share.
Online pharmacies, though still smaller, record a 10.43% CAGR as payers endorse home infusion for chronic conditions. Amazon Pharmacy and CVS Health expanded cold-chain depots and adherence platforms in 2025 to accommodate injectable biologics. The FDA’s 2025 draft on home administration lays regulatory groundwork for continued channel migration. Manufacturers must therefore design multifaceted distribution strategies to maximize the reach of the generic injectable market while safeguarding cold-chain integrity.
Geography Analysis
North America accounted for 44.76% of global revenue in 2025, driven by U.S. biosimilar uptake and high hospital spending. Price negotiations under the Inflation Reduction Act temper future growth, but the FDA’s streamlined pathways sustain a healthy launch cadence. Canada’s mandatory biosimilar switching policies in Ontario and British Columbia advanced adoption ahead of schedule. Mexico increased generic injectable procurement by 18% in 2025, though national cold-chain gaps persist.
Asia-Pacific is projected at 8.54% CAGR, the fastest regional rate. India, China, and South Korea collectively invested more than USD 3 billion in sterile capacity between 2021 and 2025, creating export hubs and bolstering regional self-reliance. China authorized 22 biosimilars in 2024–2025, the highest global tally, while South Korea’s Samsung Biologics and Celltrion fielded over 600,000 liters of bioreactor capacity. These supply additions underpin robust growth in the generic injectable market across Southeast Asia and Oceania.
Europe combines mature penetration with policy-induced upside. Germany achieved a 42% biosimilar share of the eligible biologics market in 2025, and the U.K. aims for 90% penetration by 2027. France centralized hospital procurement in 2025, resulting in an average 19% price concession. Eastern Europe and the Nordics capture incremental volume as local tenders prioritize affordable oncology injectables. Middle East and Africa diversify via Saudi localization and donor-supported African programs, whereas South America’s outlook hinges on Brazil and Argentina’s public formularies amid currency swings.
Competitive Landscape
The top 10 suppliers controlled about 45% of 2025 revenue, reflecting moderate concentration within the generic injectable market. Samsung Biologics, Biocon Biologics, and Sandoz leverage scale and regulatory fluency to introduce high-value biosimilars rapidly, while Catalent and Thermo Fisher’s Patheon units monetize flexible fill-finish slots for third parties. Mid-sized firms Fresenius Kabi, Hikma, and Gland Pharma retain competitive footing by ensuring uninterrupted hospital supply and attracting multi-year GPO contracts.
Technology adoption is accelerating. Pfizer’s AI-enabled monitoring reduced contamination events by 18%, an advantage translating to higher plant utilization and quicker lot release. Smaller players retrofit legacy suites with predictive maintenance and electronic batch records to narrow the compliance gap. Intellectual-property settlements, such as Amgen’s 2024 Enbrel litigation, alter regional launch windows and keep market positions fluid.
Regulatory scrutiny remains stringent. The FDA and EMA intensified inspections in 2024–2025, issuing import alerts that temporarily sidelined several facilities in India and China. Companies with robust quality management now enjoy clearer competitive lanes, while entrants must absorb higher compliance overhead. Ultimately, manufacturing scale, sterile-quality pedigree, and agility in capacity deployment define leadership in the evolving generic injectable market.
Generic Injectable Industry Leaders
Pfizer Inc
Novartis AG (Sandoz)
Fresenius Kabi
Teva Pharmaceutical Industries Ltd
Hikma Pharmaceuticals Plc
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: PharmaTher Holdings Ltd., a specialty pharmaceutical company, announced a new strategic initiative to pursue Health Canada approval for generic semaglutide in Canada and, subject to regulatory approval, its commercialization. Semaglutide is the active ingredient in Ozempic and Wegovy.
- August 2025: Teva Pharmaceuticals, Inc., a U.S. affiliate of Teva Pharmaceutical Industries Ltd., received FDA approval for Saxenda 1 (liraglutide injection).
Global Generic Injectable Market Report Scope
As per the scope of the report, generic injectables are medications that contain the same active ingredients, dosage, and form as brand-name injectables but are sold under their chemical names. They are typically more affordable and approved by regulatory authorities after the original drug's patent expires. These injectables are used in various therapeutic and cosmetic applications to treat medical conditions or enhance appearance.
The Generic Injectable Market is Segmented by Product Type (Monoclonal Antibodies, Immunoglobulin, Cytokines, Insulin, Peptide Hormones, and Other Product Types), Application (Oncology, Infectious Diseases, Diabetes Management, Cardiovascular Diseases, Critical Care & Emergency, and Parenteral Nutrition), Distribution Channel (Retail Pharmacies, Hospital Pharmacies, Online Pharmacies, and Other Distribution Channel), and Geography (North America, Europe, Asia-Pacific, Middle East & Africa, and South America). The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD million) for the above segments.
| Monoclonal Antibodies |
| Immunoglobulin |
| Cytokines |
| Insulin |
| Peptide Hormones |
| Other Product Types |
| Oncology |
| Infectious Diseases |
| Diabetes Management |
| Cardiovascular Diseases |
| Critical Care & Emergency |
| Parenteral Nutrition |
| Retail Pharmacies |
| Hospital Pharmacies |
| Online Pharmacies |
| Other Distribution Channel |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest Of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest Of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest Of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest Of South America |
| By Product Type | Monoclonal Antibodies | |
| Immunoglobulin | ||
| Cytokines | ||
| Insulin | ||
| Peptide Hormones | ||
| Other Product Types | ||
| By Application | Oncology | |
| Infectious Diseases | ||
| Diabetes Management | ||
| Cardiovascular Diseases | ||
| Critical Care & Emergency | ||
| Parenteral Nutrition | ||
| By Distribution Channel | Retail Pharmacies | |
| Hospital Pharmacies | ||
| Online Pharmacies | ||
| Other Distribution Channel | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest Of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest Of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest Of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest Of South America | ||


Key Questions Answered in the Report
What is the current valuation of the generic injectable market?
The generic injectable market size stands at USD 273.78 billion in 2026 and is projected to reach USD 386.15 billion by 2031.
Which application area is expanding the fastest?
Diabetes management injectables register the highest forecast growth at a 10.55% CAGR through 2031, driven by biosimilar insulin uptake and future generic GLP-1 entries.
Why is Asia-Pacific expected to outpace other regions?
Capacity incentives in India, China, and South Korea, alongside rising chronic disease prevalence, propel Asia-Pacific to an 8.54% CAGRÑthe fastest regional rate to 2031.
How are hospitals addressing injectable drug shortages?
U.S. hospital groups now engage multiple suppliers, often accepting modestly higher prices to secure reliable access, benefiting compliant mid-tier manufacturers.
What technological trends are improving sterile manufacturing reliability?
Artificial-intelligence process analytics, including real-time environmental monitoring and predictive maintenance, have cut contamination events by up to 18% at leading plants.
Page last updated on:









