Diagnostic Reagents Market Size and Share
Diagnostic Reagents Market Analysis by Mordor Intelligence
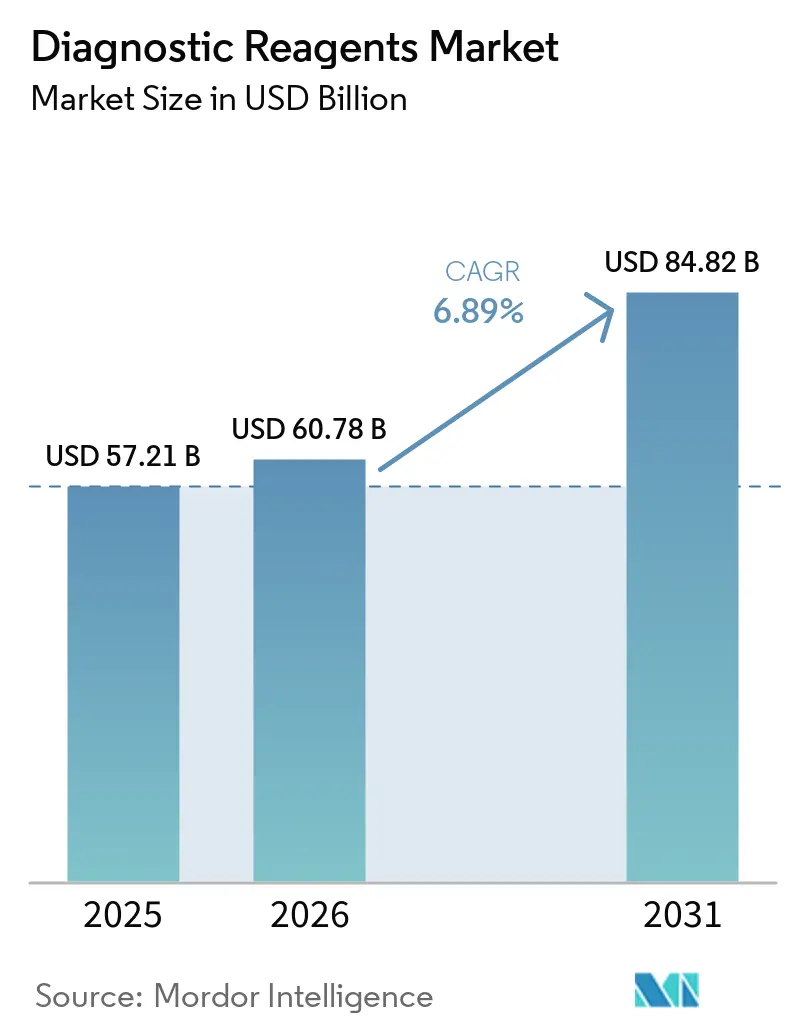
The Diagnostic Reagents Market size is expected to increase from USD 57.21 billion in 2025 to USD 60.78 billion in 2026 and reach USD 84.82 billion by 2031, growing at a CAGR of 6.89% over 2026-2031.
The near-term growth profile aligns with sustained pull-through from high-throughput analyzers and decentralized testing formats that lift average tests per patient while rebalancing cost-to-serve across centralized and distributed sites. The adoption of lyophilized and ambient-stable formats is compressing cold-chain spend and enabling reliable distribution into retail pharmacies and home settings, strengthening volume resilience across flu and RSV seasons. Core-lab consolidation concentrates purchasing power, but next-generation systems with automated calibration and higher test density deepen reagent annuities and improve reorder predictability for integrated platforms. In parallel, the EU IVDR transition continues to reshape launch sequencing and documentation depth, which favors suppliers with scalable quality systems and internal regulatory resources. Across regions, North America remains the largest corridor by share while Asia-Pacific leads growth, with tender dynamics and local manufacturing capacity determining price realization and access pathways for the diagnostic reagents market.
Key Report Takeaways
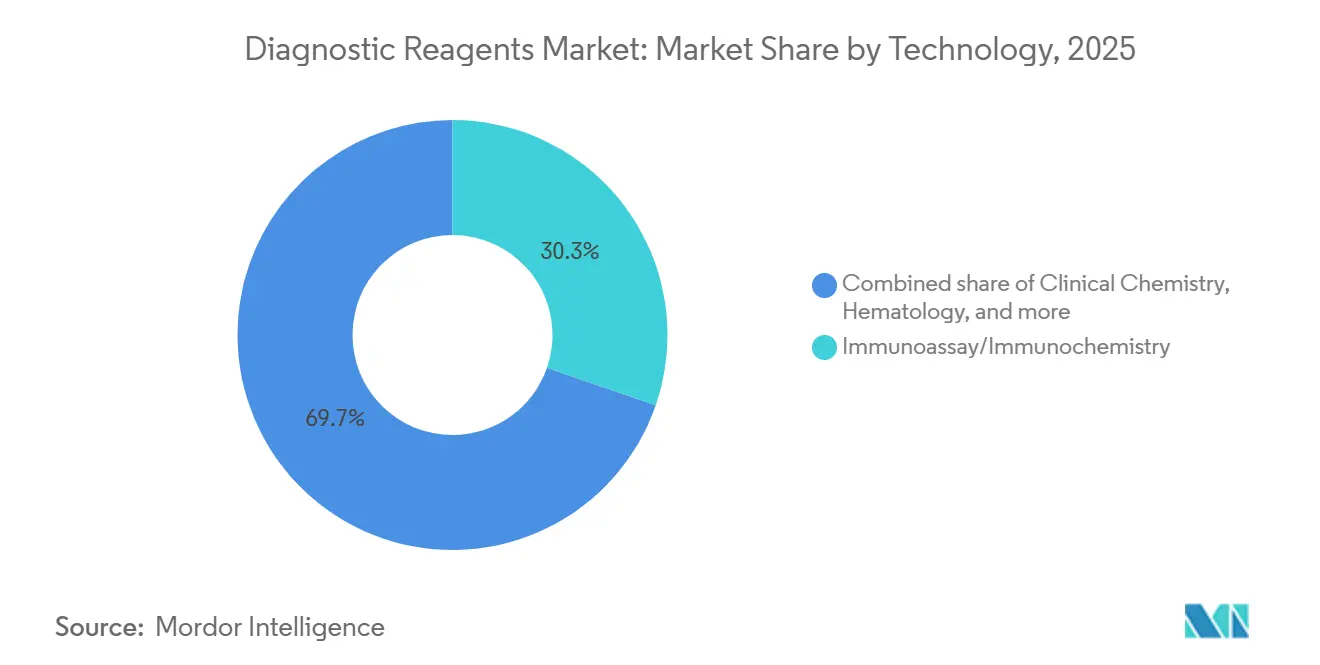
- By technology, immunoassay and immunochemistry led with 30.29% of diagnostic reagents market share in 2025; molecular diagnostics reagents are projected to expand at an 8.13% CAGR to 2031.
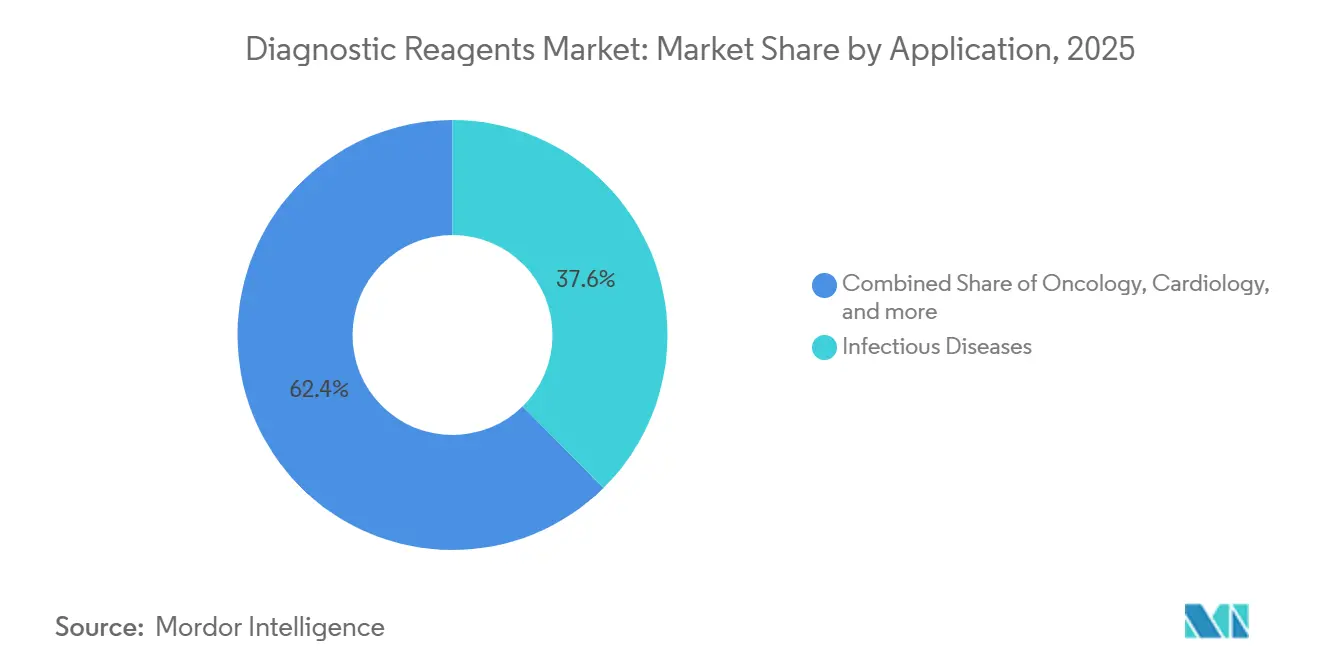
- By application, infectious disease accounted for a 37.6% share of the diagnostic reagents market size in 2025; oncology reagents are forecast to grow at a 7.15% CAGR to 2031.
- By end user, hospital laboratories absorbed 45.13% of shipments in 2025; homecare and self-testing reagents are set to rise at an 8.09% CAGR through 2031.
- By geography, North America held 41.37% of diagnostic reagents market share in 2025; Asia-Pacific is projected to post a 9.13% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Diagnostic Reagents Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Aging population and chronic disease burden elevate routine diagnostics demand | +1.2% | Global | Medium term (2-4 years) |
| Shift to point-of-care and decentralized testing accelerates reagent throughput | +1.4% | Global, early gains in North America & EU, spill-over to APAC | Short term (≤ 2 years) |
| Molecular (PCR/dPCR/NGS) adoption expands high-value assay pull-through | +2.1% | Global | Long term (≥ 4 years) |
| Core-lab consolidation and total lab automation increase consumables velocity | +0.9% | North America & EU core, APAC metro hubs | Medium term (2-4 years) |
| Lyophilized/ambient-stable formats reduce cold-chain costs and enable distributed sites | +0.8% | APAC core, spill-over to MEA, Latin America | Medium term (2-4 years) |
| OEM/contract manufacturing of reagents speeds launches and scales capacity | +0.6% | Global | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Aging Population And Chronic Disease Burden Elevate Routine Diagnostics Demand
Demographic aging increases the baseline rate of routine panels for cardiometabolic, oncology, and infectious conditions, which stabilizes recurring test volumes for laboratories and hospital networks. Chronic patients revisit testing on defined clinical intervals, which converts installed analyzers into predictable reagent annuities that cushion revenue variability across seasons. In the diagnostic reagents market, this steady cadence pairs with aligned Medicare fee schedule updates that shape the timing and pricing of run-rate testing across providers. Reagent throughput in high-volume labs benefits from new automated units that lift test density and reduce manual interventions while upholding quality controls.
That same dynamic supports integrated delivery networks that look to consolidate platforms and standardize assay menus for clinical chemistry, immunoassay, and specialized testing. As a result, the diagnostic reagents market sees a consistent mix of hospital, reference lab, and physician office demand anchored by chronic care pathways.
Shift To Point Of Care And Decentralized Testing Accelerates Reagent Throughput
Over-the-counter and CLIA-waived rapid tests continue to migrate testing closer to patients, which raises utilization and changes packaging economics for reagents. The Flowflex Plus 4 in 1 home test for RSV, influenza A and B, and COVID illustrates this shift by delivering a four-target result in minutes without instrumentation, which expands consumer access and supports faster clinical decisions in primary care follow-up[2]ACON Labs Product Team, “Flowflex Plus 4 in 1 Home Test,” Flowflex, flowflexcovid.com .
To supply these decentralized channels at scale, manufacturers are adopting lyophilized or air-dried chemistries that maintain stability at ambient conditions, cutting cold-chain dependence and reducing transport waste. These formats are now table stakes in the diagnostic reagents market, especially as retailers and telehealth models integrate self-collection and home testing into episodic care pathways. Packaging shifts from bottles to single-use cartridges or cassettes increase unit costs, yet they open new distribution lanes such as pharmacy shelves and mail-order fulfillment that lift total test counts.
As decentralized programs scale, vendors that prove reliability in ambient-stable formats capture recurring demand that is less exposed to hospital procurement cycles.
Molecular (PCR/dPCR/NGS) Adoption Expands High Value Assay Pull Through
Menu breadth and run configuration advances in PCR and NGS platforms raise the lifetime value of each instrument footprint in core labs and reference labs. Roche’s next-generation cobas 6800/8800 systems and software update in December 2025 improved lab efficiency and testing capabilities, which boosts system attractiveness for labs that need to consolidate multiple assays per run.
In parallel, bioMérieux strengthened its NGS capabilities by acquiring Day Zero Diagnostics’ solutions and technologies in June 2025, adding upstream sample preparation and bioinformatics assets to enable faster identification workflows[1]bioMérieux Corporate Communications, “bioMérieux Strengthens Its Next-Generation Sequencing Capabilities with the Acquisition of Day Zero Diagnostics,” bioMérieux, biomerieux.com . The diagnostic reagents market benefits when these platforms anchor monthly reagent cycles for oncology, infectious disease, and transplant panels rather than episodic surges. Long-lived compliance frameworks under IVDR and U.S. quality system regulations drive documentation depth and lot traceability for molecular kits, which reward manufacturers with mature quality systems. As system menus expand, labs increase reliance on proprietary reagent packs that integrate with analyzer software, which deepens the installed base lock-in effect for top suppliers.
Core Lab Consolidation And Total Lab Automation Increase Consumables Velocity
Consolidation of laboratories into larger core facilities raises average daily throughput and concentrates orders onto fewer but more capable analyzer families. Beckman Coulter’s integrated chemistry platform supports heavy-duty test volumes with automated workflows that reduce manual touches and maintain calibration stability at pace. Siemens Healthineers’ Atellica Solution also emphasizes throughput, assay breadth, and sample management, which aligns with IDN strategies that standardize operations across multi-site networks.
Automation layers such as Inpeco’s FlexLab X improve pre-analytical handling and tube routing at very high speeds, which allows labs to run 24 hour operations with fewer bottlenecks. In this setup, reagent packaging and formulations must tolerate extended onboard conditions and align with barcode and LIS standards to avoid downtime or QC flags. The diagnostic reagents market sees faster consumption velocity per instrument, and vendors that meet automation and integration specs can capture multi-year reagent annuities in core accounts.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| EU IVDR compliance burden and Notified Body bottlenecks delay launches, raise costs | -0.9% | EU core, ripple to global portfolios | Short term (≤ 2 years) |
| Reimbursement cuts and China's VBP/centralized tenders compress prices and margins | -1.1% | China national; US Medicare partial offset | Medium term (2-4 years) |
| Fragmented reimbursement and coding slow clinical adoption and scale-up | -0.5% | Global, acute in US payer landscape | Medium term (2-4 years) |
| Raw-material scarcity and lot-to-lot variability drive QC bridging and validation overhead | -0.7% | Global, acute in APAC import corridors | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
EU IVDR Compliance Burden And Notified Body Bottlenecks Delay Launches, Raise Costs
The IVDR transition requires expanded clinical evidence, unique device identification, and deeper post-market surveillance, which lift documentation and audit workloads for manufacturers. Industry surveys from European trade bodies and professional associations show companies have slowed or deferred EU launches while prioritizing faster review markets, underscoring ongoing Notified Body capacity constraints.
Labs that relied on laboratory-developed tests face stricter conditions under Article 5(5), pushing many to adopt manufacturer-supplied kits that meet IVDR requirements. For reagent makers, IVDR adds time and cost to product lifecycles, which can alter the European launch queue and drive global sequencing trade-offs. This dynamic favors vertically integrated platforms with internal regulatory teams and established QMS maturity that can manage multi-year projects with predictable cadence. The diagnostic reagents market therefore experiences a temporary slowdown in EU rollouts while U.S. and other markets absorb earlier releases.
Reimbursement Cuts And Centralized Tenders Compress Prices And Margins
The U.S. Medicare fee schedule and the clinical laboratory fee schedule updates continue to influence category pricing and test utilization, which can dampen near-term price realization for some panels. In several markets with centralized procurement, tender structures compress average selling prices and restrict volume shares per supplier, which reinforces the advantage of vendors with broader menus and integrated platforms. To mitigate pricing pressure, firms are prioritizing multiplex assays and integrated test panels that deliver more clinical value per run and align better with payer coverage decisions, including donor screening tests that combine multiple viral markers.
Suppliers with local manufacturing or channel partnerships can soften cost-to-serve and improve tender competitiveness by shortening lead times and aligning packaging to local logistics constraints. Over time, the mix shift toward higher-value molecular assays can offset part of the margin compression in commodity chemistries, stabilizing portfolio-level economics in the diagnostic reagents market.
Segment Analysis
By Technology: Fleets Propel Automation, Molecular Sprints Ahead
Immunoassay and immunochemistry reagents captured 30.29% of 2025 revenue within the diagnostic reagents market, supported by high-frequency testing across hospital networks that run large, integrated analyzers. In parallel, molecular diagnostics reagents spanning PCR, dPCR, and NGS are projected to post an 8.13% CAGR through 2031, lifted by oncology, infectious disease, and pharmacogenomic applications that create durable test menus. Systems-level upgrades on integrated chemistry platforms increase test density, raise uptime, and improve reagent pack efficiency, which accelerates pull-through in consolidated labs.
Automation layers that speed pre-analytical handling further support uninterrupted workflows in metro hubs that process high tube volumes per hour. Vendors that match analyzer throughput with stable, onboard-compatible formulations capture sole-source contracts that secure multi-year annuities, which is a defining feature of the diagnostic reagents market. Menu consolidation also continues in blood safety, where new donor screening assays combine multiple viral targets to reduce per-sample handling and lift per-run value.
Across the diagnostic reagents industry, decentralized rapid tests expand access and shift packaging from bulk bottles to single-use cassettes that preserve shelf life and support mass distribution through retail and home channels. This shift relies on lyophilized or air-dried chemistries that reduce cold-chain burdens and keep unit performance stable under variable transport conditions. Open-platform chemistries in clinical chemistry and hematology remain vital for routine labs, but competitive intensity and procurement consolidation put pressure on prices as labs standardize menus and leverage higher volumes.
Specialty segments such as tissue and cell-based assays keep pace as companion diagnostics expand into new indications, reinforcing the role of regulated kits that meet quality system and evidence requirements. As fleets modernize, buyers value analyzers with automated calibration cycles and predictive maintenance, which extends run time and reduces waste linked to repeat QC. This setup creates a path for vendors that balance equipment innovation with reliable reagent packs to protect share in the diagnostic reagents market.
Note: Segment shares of all individual segments available upon report purchase
By Application: Infectious Disease Volume, Oncology Velocity
Infectious disease testing led application revenue at 37.6% in 2025 within the diagnostic reagents market size, supported by respiratory seasons, hospital-acquired infection management, and donor screening programs that rely on consistent panel availability. The product roadmaps across infectious disease emphasize multiplex capability that reduces per-pathogen costs and streamlines sample handling, with blood safety panels that roll multiple viral targets into a single workflow.
Suppliers that reset plant scheduling and inventory policies for respiratory surges maintain service levels better across winter waves, which strengthens account retention. In clinical settings, automation and LIS integration stabilize turnaround times that are central to infection control protocols. For reference labs and donor screening, menu reliability and uptime assurance remain critical purchase criteria that tilt awards toward integrated platforms. Together these features preserve the role of infectious disease testing as the basal volume driver in the diagnostic reagents market.
Oncology reagents are projected to grow at 7.15% CAGR through 2031, paced by liquid-biopsy and tissue-based companion diagnostics that move more testing into routine care pathways and therapeutic selection. NGS-based panels and multi-biomarker immunohistochemistry assays shift test economics by concentrating more value into each kit, which supports sustainable reimbursement and lab interest in platform adoption. Vendors that partner with pharma for companion diagnostic development strengthen coverage positions and secure predictable volumes through label-linked testing programs. Clinical uptake also benefits from analyzers with streamlined sample prep and automated workflows that shorten time to result while preserving accuracy.
As oncology menus expand, labs rebalance capacity between molecular and immunohistochemistry, which raises the share of high-value tests in overall reagent consumption. These shifts collectively raise the average selling price mix and support value retention in the diagnostic reagents market.
Note: Segment shares of all individual segments available upon report purchase
By End User: Hospital Labs Dominate, Homecare Surges
Hospital laboratories absorbed 45.13% of global shipments in 2025, reflecting scale benefits and continuous operations that favor integrated analyzers with high throughput and automated maintenance cycles.
Analyzer innovations that couple chemistry and immunoassay capabilities in compact footprints allow hospitals to consolidate benches and elevate run-rate efficiency, which deepens reagent pull-through per site. Physicians’ office labs and urgent care sites expand use of CLIA-waived tests that trade unit pricing for speed and convenience, anchoring point-of-care growth in the diagnostic reagents market. Reference labs continue to concentrate specialized testing and absorb outreach volumes from community providers that prefer rapid collection over in-house testing capacity. Across these settings, quality system maturity and supply assurance are recurring selection criteria that determine account wins and renewals. The fit between analyzer fleets and lab informatics also remains central to uninterrupted throughput and reliable QC.
Homecare and self-testing reagents are projected to grow at an 8.09% CAGR through 2031, powered by the expansion of OTC and at-home molecular kits that allow people to test and act without a clinic visit. Multi-target antigen tests like Flowflex Plus 4 in 1 help standardize home workflows and support consistent user experience, which in turn drives repeat purchases in the retail channel.
The diagnostic reagents market benefits as these products bring in new users and expand test frequency beyond traditional care pathways. For suppliers, the challenge is to secure robust stability claims and packaging that guard performance under home storage conditions, while maintaining cost discipline. Companies with vertical integration across reagents, devices, and digital support tools are positioned to achieve better adherence and follow-up care, which translates into durable volumes. Over time, home testing will coexist with central lab services, with data integration improving clinical follow-up and population health analytics.
Geography Analysis
North America held a 41.37% share of diagnostic reagents market size in 2025, supported by strong payer coverage for high-acuity testing, companion diagnostics integration, and IDN consolidation that standardizes analyzer fleets. Medicare fee schedule and laboratory fee updates influence price realization and test utilization, which labs manage through automation and throughput gains that lower per-test costs. The throughput uplift seen with next-generation analytical units supports consolidated sites that run continuous operations and schedule maintenance around predictable workloads[3]Centers for Medicare & Medicaid Services, “Clinical Laboratory Fee Schedule 2026 Annual Update,” CMS, cms.gov .
Europe remains a large installed base with steady demand, but IVDR-related workload and Notified Body capacity constraints have extended certification timelines and altered launch sequencing for many companies. Surveys from professional associations and industry groups indicate that firms have reframed European pipelines to prioritize products with available review capacity, which has reduced the pace of some EU introductions. Article 5(5) implications have also pushed some labs to reduce reliance on LDTs and adopt manufacturer kits that meet IVDR requirements.
Asia-Pacific is projected to grow at a 9.13% CAGR to 2031 in the diagnostic reagents market, with urban centers expanding core-lab capacity and decentralized channels improving access in community settings. In several countries, centralized procurement programs and local manufacturing push drive pricing discipline and faster access to high-volume tests. Tender structures and local content preferences influence supplier selection, which helps domestic firms gain share in closed-system analyzers. International suppliers strengthen positions by pairing menu breadth with localized service, training, and supply chain flexibility.
Competitive Landscape
Competition segments into vertically integrated platforms that bundle instruments with closed menus, open-platform reagent specialists that sell across fleets, and contract manufacturers that enable scale for emerging diagnostics brands. Integrated platforms strengthen account loyalty with high-throughput analyzers and automated calibration, which reduce downtime and deepen reagent annuities in core labs. Suppliers that add high-value panels such as multi-target donor screening create more value per run, which supports share retention in competitive tenders. Open-platform specialists differentiate on assay performance and turnaround time, with acquisitions that bring in upstream sample prep and NGS analytics to speed time to result. Point-of-care expansions draw on compact immunoassay platforms that fit near-patient workflows and complement central lab menus.
Vendors with robust QMS and regulatory track records will be better positioned to navigate IVDR and U.S. quality system demands without supply disruptions. Companies that combine hardware, software, and reagent design into cohesive solutions gain leverage in tenders and IDN negotiations, particularly where uptime and throughput commitments are part of the contract. In decentralized channels, user-friendly packaging and long shelf life influence reorders and brand stickiness. Contract manufacturers will continue to enable new entrants and accelerators, but quality, traceability, and capacity assurance will decide which partnerships scale. The net effect is a competitive field where scale, menu breadth, and compliance maturity determine steady share defense and targeted share gains for leading portfolios.
Diagnostic Reagents Industry Leaders
F. Hoffmann-La Roche (Roche Diagnostics)
Abbott Diagnostics
Thermo Fisher Scientific
Danaher (Beckman Coulter; Cepheid)
Siemens Healthineers
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- March 2026: Roche Diagnostics received US FDA 510(k) clearance for cobas c 703 and cobas ISE neo analytical units that double throughput on cobas pro integrated solutions while improving automated calibration, maintenance, and reagent pack efficiency.
- March 2026: Abbott successfully completed its USD 21 billion acquisition of Exact Sciences, strategically enhancing its diagnostics portfolio. This acquisition incorporates Cologuard (a colorectal cancer screening solution), Oncotype DX (a breast cancer genomic assay), and Cancerguard (a multi-cancer early detection platform). The transaction is projected to contribute over USD 3 billion in annual reagent revenue, increasing Abbott's total diagnostics revenue to more than USD 12 billion. This positions Abbott as a market leader within the US cancer screening market, which is valued at approximately USD 60 billion.
Global Diagnostic Reagents Market Report Scope
As per the scope of the report, diagnostic reagents are chemical, biological, or immunological substances used to detect, measure, or analyze components in samples (such as blood or tissue) to diagnose diseases, monitor treatments, or evaluate health status. The diagnostic reagents market is segmented by technology, application, end user, and geography. By technology, the market is segmented as immunoassay/immunochemistry, clinical chemistry, molecular diagnostics (PCR, DPCR, NGS), hematology, coagulation, microbiology (ID/AST), urinalysis, rapid tests (lateral flow/POC), and tissue & cell-based (IHC/ISH). By application, the market is segmented as infectious diseases, oncology, cardiology, endocrinology/diabetes, autoimmune & inflammation, nephrology & liver function, and women’s health & prenatal/STD. By end user, the market is segmented as hospital laboratories, independent/reference laboratories, point-of-care/physician offices/urgent care, and homecare/self-testing. By geography, the market is segmented as North America, Europe, Asia-Pacific, the Middle-East and Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| Immunoassay/Immunochemistry |
| Clinical Chemistry |
| Molecular Diagnostics (PCR, dPCR, NGS) |
| Hematology |
| Coagulation |
| Microbiology (ID/AST) |
| Urinalysis |
| Rapid Tests (Lateral Flow/POC) |
| Tissue & Cell-based (IHC/ISH) |
| Infectious Diseases |
| Oncology |
| Cardiology |
| Endocrinology/Diabetes |
| Autoimmune & Inflammation |
| Nephrology & Liver Function |
| Women’s Health & Prenatal/STD |
| Hospital Laboratories |
| Independent/Reference Laboratories |
| Point-of-Care/Physician Offices/Urgent Care |
| Homecare/Self-testing |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Technology | Immunoassay/Immunochemistry | |
| Clinical Chemistry | ||
| Molecular Diagnostics (PCR, dPCR, NGS) | ||
| Hematology | ||
| Coagulation | ||
| Microbiology (ID/AST) | ||
| Urinalysis | ||
| Rapid Tests (Lateral Flow/POC) | ||
| Tissue & Cell-based (IHC/ISH) | ||
| By Application | Infectious Diseases | |
| Oncology | ||
| Cardiology | ||
| Endocrinology/Diabetes | ||
| Autoimmune & Inflammation | ||
| Nephrology & Liver Function | ||
| Women’s Health & Prenatal/STD | ||
| By End User | Hospital Laboratories | |
| Independent/Reference Laboratories | ||
| Point-of-Care/Physician Offices/Urgent Care | ||
| Homecare/Self-testing | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
Key Questions Answered in the Report
What is the diagnostic reagents market size outlook through 2031?
The diagnostic reagents market size is projected at USD 57.21 billion in 2025, USD 60.78 billion in 2026, and USD 84.82 billion by 2031, at a 6.89% CAGR from 2026 to 2031.
Which technology leads and which grows fastest within diagnostic reagents?
Immunoassay and immunochemistry lead by revenue with 30.29% in 2025, while molecular diagnostics reagents are the fastest with an 8.13% CAGR to 2031.
Which applications are most important for demand momentum?
Infectious disease holds the largest share at 37.6% in 2025, and oncology shows the strongest growth at a 7.15% CAGR through 2031.
Who are the primary end users driving reagent pull-through?
Hospital laboratories account for 45.13% of 2025 shipments, and homecare or self-testing is the fastest-growing with an 8.09% CAGR through 2031.




