Cat Allergy In Humans Market Size and Share
Cat Allergy In Humans Market Analysis by Mordor Intelligence
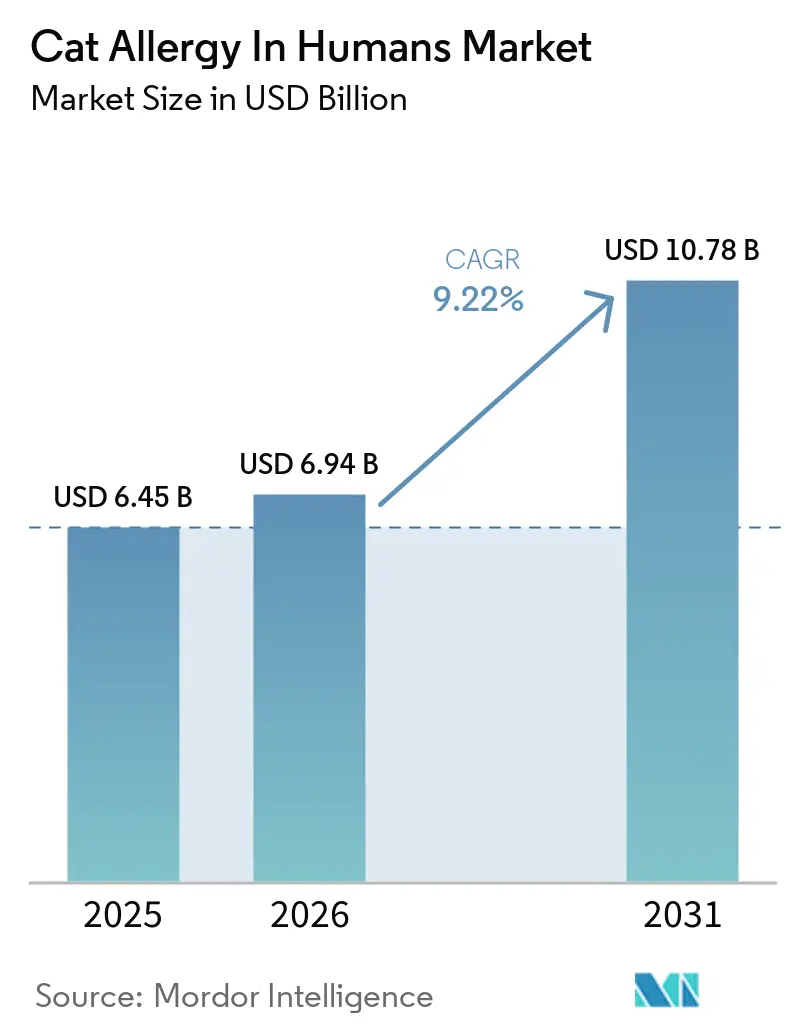
The Cat Allergy In Humans Market size is projected to expand from USD 6.45 billion in 2025 and USD 6.94 billion in 2026 to USD 10.78 billion by 2031, registering a CAGR of 9.22% between 2026 to 2031.
Growth reflects a broader therapeutic mix that spans widely used over-the-counter antihistamines, prescription intranasal corticosteroids for difficult cases, and a maturing biologics pipeline led by Fel d 1–blocking monoclonal antibodies moving toward confirmatory Phase 3 studies in 2026[1]Regeneron Pharmaceuticals, “Regeneron Advances Allergy Pipeline with Two Positive Phase 3 Trials,” GlobeNewswire, globenewswire.com. Demand expands as cat ownership rises and prolonged indoor cohabitation raises exposure among sensitized individuals in high-density markets. Clinical adoption of subcutaneous immunotherapy continues because it alters disease course and reduces long-term medication dependence in adherent patients, while sublingual approaches remain constrained in the United States by the lack of approved cat-specific tablets. Component-informed diagnostics and standardized extract labeling have improved prescribing decisions and dosing safety, while dietary allergen-reduction strategies and late-stage biologics open choices for non-adherent or needle-averse patients, keeping the cat allergy in humans market on a diversified growth path
Key Report Takeaways
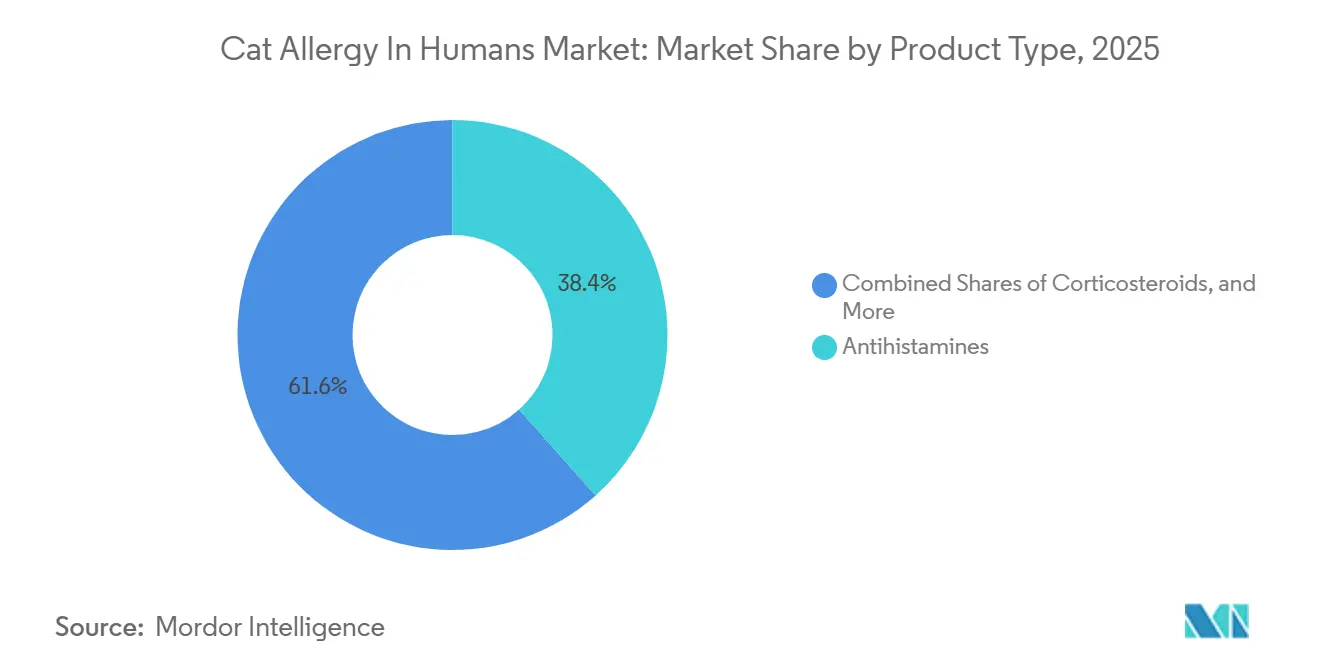
- By product type, antihistamines led with 38.43% share in 2025. Allergen-blocking monoclonal antibodies are projected to expand at an 11.87% CAGR through 2031.
- By route of administration, oral formulations held 45.34% in 2025. Subcutaneous administration is forecast to record a 9.87% CAGR through 2031.
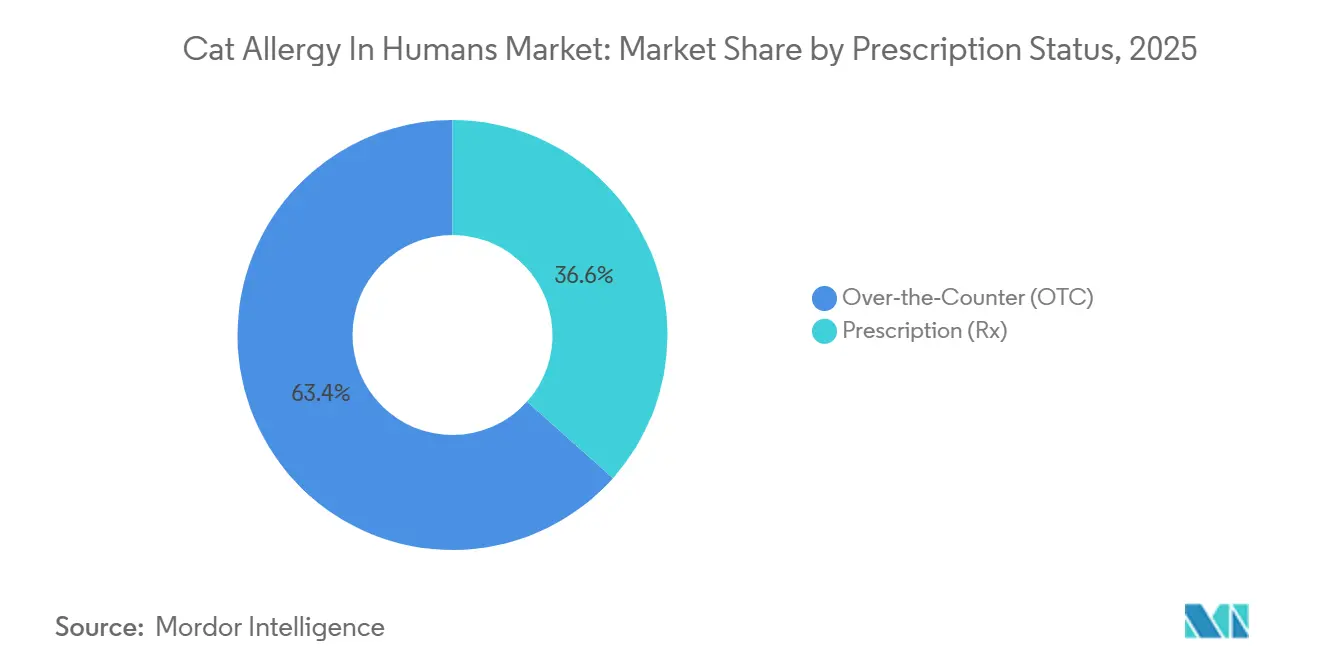
- By prescription status, over-the-counter products accounted for 63.42% in 2025. The OTC segment is projected to expand at an 9.65% CAGR through 2031.
- By distribution channel, retail pharmacies held 52.34% in 2025. Online pharmacies are expected to grow at an 11.51% CAGR through 2031.
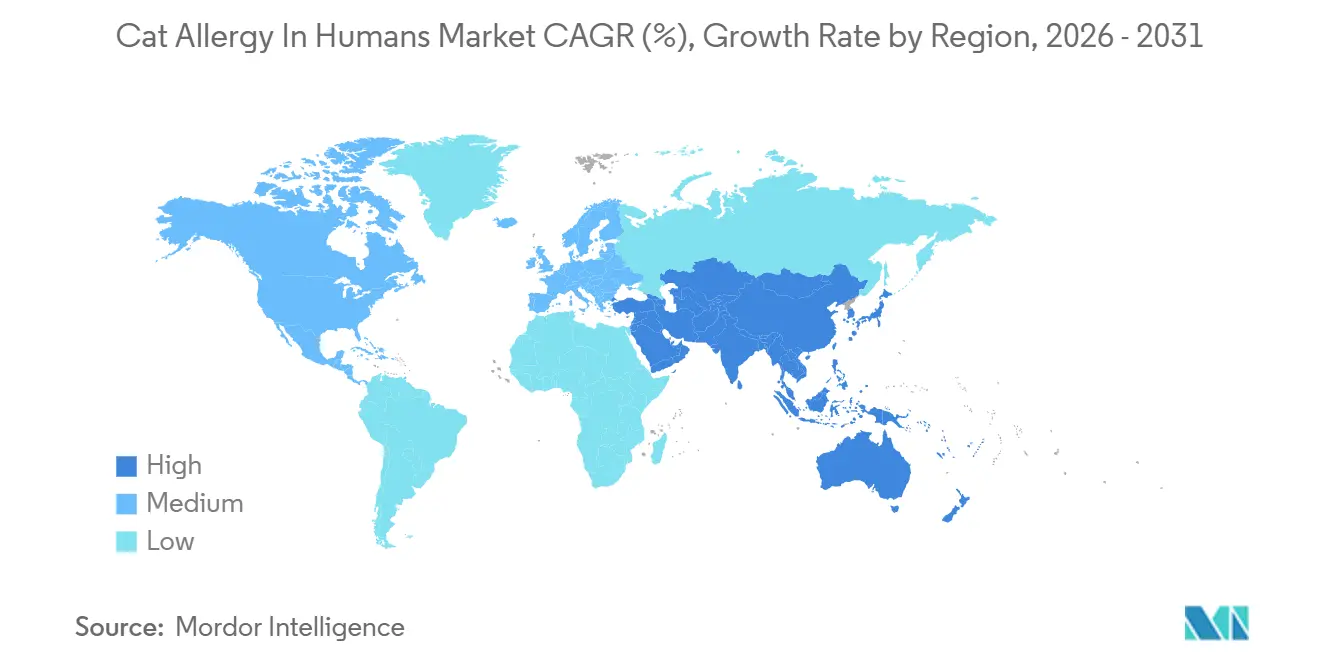
- By geography, North America held 42.44% of the global total in 2025; Asia-Pacific is forecast to grow at an 11.95% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Cat Allergy In Humans Market Trends and Insights
Drivers Impact Analysis
| Driver | % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising prevalence and sensitization to cat allergens among allergic rhinitis/asthma patients | +2.8% | Global, concentration in urbanized Asia-Pacific (China, India) and North America | Medium term (2-4 years) |
| Growing pet cat ownership and indoor cohabitation increasing exposure intensity | +2.3% | North America, Europe, urban China | Short term (≤ 2 years) |
| Demonstrated disease-modifying benefit and physician confidence in SCIT for animal dander | +1.5% | North America, Western Europe | Long term (≥ 4 years) |
| Expansion of treatment options across OTC, Rx, AIT, and emerging biologics | +1.9% | Global, early gains in North America and Europe | Medium term (2-4 years) |
| Allergen-exposure reduction solutions broadening adjunct management | +1.2% | North America, spillover to Europe | Medium term (2-4 years) |
| Emerging Biologics (E.g., Allergen-Blocking Mabs; TSLP Blockade Adjunct To AIT) Enlarging Addressable Pool | +1.1% | North America, Europe, Asia-Pacific (Japan) | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Rising Prevalence And Sensitization To Cat Allergens Among Allergic Rhinitis/Asthma Patients
Clinical studies in large urban cohorts document a steady rise in cat-dander sensitization that coincides with extended indoor exposure and lifestyle patterns that keep people and pets together in smaller living spaces[2]Li Le et al., “Sensitization Profiles of Aeroallergens Among Allergic Rhinitis Patients in Central China: A Six-Year Real-World Study,” Asian Pacific Journal of Allergy and Immunology, apjai-journal.org. In central China, the share of allergic rhinitis patients with cat sensitization climbed across a six-year period, highlighting a clear need for therapy pathways that can offer durable control beyond short-term symptomatic relief. Pediatric and adolescent cohorts show a delayed peak for cat dander compared with dust mite, which suggests that cumulative environmental exposure and behavioral shifts, rather than early innate susceptibility, shape timing and severity.
These patterns broaden the base of candidates for immunotherapy and for supportive measures such as component-informed counseling that help align treatment with the specific allergen profile. They also sustain demand across multiple access points because patients often begin with OTC antihistamines and later escalate to prescription therapies or immunotherapy when symptoms persist despite self-management. As a result, the cat allergy in humans market continues to add patients across both symptomatic and disease-modifying care streams, reinforcing a durable growth outlook
Growing Pet Cat Ownership And Indoor Cohabitation Increasing Exposure Intensity
Pet-cat ownership in the United States rose quickly in 2024, lifting cat-owning households to 49 million and increasing the number of people who experience direct and frequent exposure to Fel d 1 indoors[3]American Pet Products Association, “The American Pet Products Association (APPA) Releases 2025 Dog & Cat Report, Revealing a New Era of Pet Ownership,” American Pet Products Association, americanpetproducts.org. The United Kingdom maintained a large cat population as well, and a quarter of households reported owning cats, confirming cross-market patterns that keep the allergen present in living spaces for long periods. These exposure trends feed demand for front-line OTC options that provide quick relief, followed by physician-directed regimens for persistent symptoms that fail to resolve on oral antihistamines alone. Allergen persistence within homes also supports interest in environmental and source-reduction strategies that can be paired with drug therapy to lower overall symptom burden during high-exposure periods. Providers apply this context to recommend integrated plans combining medication, immunotherapy when indicated, and everyday mitigation steps that match patient preference and risk tolerance. This ecosystem supports a balanced and growing cat allergy in humans market that serves a broad population across age groups and severity profiles.
Demonstrated Disease-Modifying Benefit And Physician Confidence In SCIT For Animal Dander
Subcutaneous immunotherapy remains central in moderate-to-severe cat allergy because it can change disease trajectory and reduce downstream medication use in adherent cohorts. Spanish real-world study using a depigmented, polymerized extract reported large gains in quality-of-life and asthma control over 24 months, while an independent safety cohort found treatment well-tolerated with few discontinuations. U.S. labels for standardized cat hair and cat pelt extracts require in-clinic dosing and post-injection observation, which anchors SCIT inside provider settings with staff and equipment for reaction management. Non-interchangeability between hair and pelt extracts and explicit instructions for dose reduction when switching lots also encourage continuity and careful supervision, further consolidating SCIT within clinic-centered workflows. This risk-managed model sustains physician confidence because it blends reproducible outcomes with practical safety guardrails that can be executed in regular practice. These features keep SCIT as a durable pillar within the cat allergy in humans market, even as newer modalities enter the mix.
Emerging biologics enlarging addressable pool
Late-stage programs for allergen-blocking monoclonal antibodies have produced strong reductions in ocular symptoms and skin reactivity in controlled allergen-challenge settings one week after a single subcutaneous dose. The same platform has advanced to present further Phase 3 analyses in 2026, and sponsors have outlined plans to begin additional registration-enabling trials, which sustain the forward path to regulatory submissions. Exploratory subgroup data suggest stronger responses in patients with Fel d 1–driven disease, which aligns with component-informed selection and highlights how diagnostics can support targeted biologic use. Adjacent research on TSLP blockade as an adjunct to immunotherapy has shown sustained biomarker and symptom improvements after treatment stops, which supports multipronged biologic strategies for a subset of difficult cases. These studies broaden clinical options for needle-averse patients who decline multi-year injection schedules and for those seeking rapid relief that does not rely on daily pills. As a result, the cat allergy in humans market is positioned to layer biologics into care algorithms when evidence and coverage align
Restraints Impact Analysis
| Restraint | % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| No FDA-approved SLIT Tablets for Cat; SLIT Drops not FDA-approved in the U.S. | -1.4% | United States | Short term (≤ 2 years) |
| SCIT Safety/logistics Burden (anaphylaxis risk, in-clinic time) Limits Uptake and Adherence | -0.9% | Global, pronounced in regions with limited allergist density (rural U.S., Southeast Asia) | Medium term (2-4 years) |
| Multi-year Adherence Challenges and Discontinuations Reduce Realized Outcomes | -0.7% | Global | Long term (≥ 4 years) |
| Extract Variability and Standardization Differences (hair vs pelt; Manufacturer/Region) Complicate Consistency | -0.5% | Global, regulatory friction highest in fragmented EU markets | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
No FDA-Approved SLIT Tablets For Cat, And SLIT Drops Not FDA-Approved In The U.S.
Sublingual tablets are FDA approved for grasses, short ragweed, and dust mite, but no cat dander tablet is licensed and liquid drops for cat remain off-label in the United States. This limits broad reimbursement and pushes many patients who prefer home administration toward SCIT or cash-pay sublingual drops prescribed in select practices that accept off-label use. Europe has demonstrated the process integrity for pediatric tablet expansion in other allergens, as seen in the 2024 approval of ACARIZAX for children, which shows that tablet platforms can progress when sponsors invest in the necessary dossiers and trials even if cat tablets are not yet available[4]Sublingual Immunotherapy Treatment for Allergic Rhinitis,” American Academy of Allergy, Asthma & Immunology, aaaai.org. Until a sponsor completes a cat-specific tablet program, the gap will continue to slow convenience-driven adoption in the United States relative to tablet-established allergens. This has a practical effect on channel dynamics because clinic-centered SCIT remains the most reimbursable pathway while sublingual drops scale more readily where national authorities already accept cat SLIT as standard of care. As a result, the cat allergy in humans market in the United States continues to rely on clinic-based models despite growing patient interest in home-use options.
SCIT Safety/Logistics Burden Limits Uptake And Adherence
Standardized cat hair and pelt extracts require administration under conditions that can manage anaphylaxis, and labels specify monitoring after injection, which creates time and travel costs for patients and operational needs for providers. Real-world depigmented cat allergoid data in Germany underscore a favorable safety profile with low discontinuation for adverse events, but anxiety about systemic reactions and the cadence of build-up and maintenance visits can still influence willingness to start or remain on therapy. Spanish data over 24 months show disease control benefits that justify the visit burden for many patients, although continued engagement over multiple years requires structured follow-up to maintain outcomes. These realities keep SCIT tied to facilities with observation capabilities and trained personnel, which helps patient safety while reinforcing in-clinic workflows. They also open the door for modalities that require fewer visits or switch to home use, especially in settings where allergist availability is limited. These logistics contribute to patient preference for convenience when efficacy is competitive and safety is predictable, which keeps interest in sublingual and biologic options active within the cat allergy in humans market.
Segment Analysis
By Product Type: Antihistamines Anchor Volume, Yet Allergen-Blocking mAbs Capture Innovation Premium
Antihistamines accounted for 38.43% of 2025 volume and remain the default starting point for many patients because they offer rapid symptom relief and easy access through retail aisles without a physician visit. Their longevity in practice and multiple oral formats support adherence for mild to moderate cases and sustain broad reach among first-line users who value predictable dosing. Intranasal corticosteroids address persistent congestion and nasal inflammation when oral antihistamines underperform, expanding physician-directed choices for patients who require stronger local anti-inflammatory control.
Allergen-blocking monoclonal antibodies are projected to expand at an 11.87% CAGR through 2031 as sponsors progress confirmatory trials and define coverage strategies for launch. Their fit is clearest for patients who want fast relief without daily pills or clinic visits every month, while immunotherapy remains the choice for those seeking a disease-modifying course of care over several years with physician oversight. Source-control nutrition that neutralizes Fel d 1 at the cat, including IgY-based diets that showed meaningful reductions in active allergen on hair by week three, can complement both drug therapy and immunotherapy inside homes that prefer to retain pets. This product mix, spanning consumer retail, prescription channels, and specialty biologics, helps the cat allergy in humans market serve heterogenous patient needs while keeping options open as evidence and labeling evolve.
Note: Segment shares of all individual segments available upon report purchase
By Route of Administration: Oral Convenience Leads, Subcutaneous Growth Signals Premium Shift
Oral formulations held 45.34% share in 2025 on the strength of second-generation antihistamines that people can use without clinic visits. Oral tablets and liquids meet consumer expectations for immediacy and predictability, supporting seasonal use and daily maintenance for milder phenotypes. Intranasal corticosteroids occupy a distinct role because they suppress nasal inflammation and congestion that oral agents often fail to resolve completely, which keeps the nasal route attractive when upper-airway symptoms dominate.
The cat allergy in humans market size for subcutaneous administration is projected to expand at a 9.87% CAGR over 2026-2031 as clinics continue to standardize SCIT pathways and as biologics target cohorts that want fewer visits with sustained relief. Biologic programs that offer three-month durability after a single subcutaneous dose may further validate the injection route for convenience-focused patients if efficacy and safety remain consistent in confirmatory studies.
By Prescription Status: OTC Dominance Meets Rx Innovation at the Biologics Frontier
Over-the-counter products accounted for 63.42% in 2025 as consumers favored immediate retail access to second-generation antihistamines that require no physician visit. This position reflects long familiarity with oral tablets and liquids, repeated seasonal use during high-exposure months, and a clear consumer pathway that begins in pharmacy aisles and continues online. Prescription-only therapies continue to serve patients who need stronger or longer-acting control, including intranasal corticosteroids for persistent rhinitis and immunotherapy for multi-year reduction in symptoms and medication reliance supported by real-world evidence. The OTC segment is projected to expand at an 9.65% CAGR through 2031 as awareness and availability remain high across regions and as people seek predictable relief with simple dosing.
As prescribers match therapy to phenotype, the prescription tier benefits from component-informed decisions that distinguish Fel d 1–driven cases from more complex sensitization profiles and from explicit extract-label instructions that structure dosing and switching. Adjunct source reduction with IgY-based cat diets can also reduce hair allergen load within three weeks and align with both OTC and prescription regimens to ease symptom burden at home. This interplay keeps the cat allergy in humans market adaptive and resilient as patient needs change over time.
By Distribution Channel: Retail Pharmacy Infrastructure Meets Online’s Digital Lift
Retail pharmacies held 52.34% of 2025 distribution because OTC antihistamines, nasal sprays, and eye drops dominate everyday use cases and because pharmacists provide guidance that supports safe and effective self-care. These outlets also serve as pick-up points for prescriptions, which encourages bundling of OTC and Rx purchases when patients collect intranasal sprays or start maintenance regimens. Hospital and specialty pharmacies focus on subcutaneous immunotherapy and biologics that need supervision, observation, or cold-chain management, including standardized cat extracts whose labels require post-injection monitoring.
Online pharmacies are expected to grow at an 11.51% CAGR through 2031 within the cat allergy in humans market as digital habits persist, and e-prescribing supports direct-to-home fulfillment for appropriate therapies. Telehealth also normalizes remote consultations, which lets clinicians order component-resolved tests through national reference labs and align therapy with patient goals without in-person visits when clinically appropriate. Growth will be strongest for products that do not require clinic infrastructure and that can be shipped without strict observation protocols, while clinic-only treatments continue to reinforce the hospital channel. These complementary flows support a durable multi-channel model that serves different levels of treatment intensity inside the cat allergy in humans market.
Geography Analysis
North America held 42.44% in 2025 in the cat allergy in humans market, supported by a dense allergist network, established SCIT pathways, and consistent use of component-informed testing that improves patient selection and dosing decisions. Cat ownership surged by 23% in 2024 to reach 49 million U.S. households, which increased exposure among sensitized individuals and lifted demand across OTC and prescription therapies as well as specialty services. Regional innovation density adds momentum because Fel d 1–blocking monoclonal antibodies continue to generate positive results in late-stage studies, and sponsors have committed to registration-enabling trials in 2026.
Europe maintains a substantial footprint shaped by national formularies that support immunotherapy and by a long history of sublingual use across other allergens, even as there is no approved cat tablet. Recent pediatric tablet approvals in dust mite show that sponsor-led clinical programs can scale successfully once efficacy and safety are clear, which offers a template for future cat-specific filings if pursued. Real-world evidence from Spain confirms SCIT effectiveness in routine practice and supports allergists’ confidence in depigmented, polymerized extracts as part of long-term care for appropriate patients.
Asia-Pacific is the fastest-growing region with an 11.95% forecast CAGR as urbanization, rising pet ownership, and increasing access to diagnostics and immunotherapy combine to expand treatment. In China, clinical cohorts show higher cat-dander sensitization in recent years, which supports the need for therapies and reinforces the role for component-resolved testing when prescribers consider multi-year plans. China’s prior experience with standardized sublingual products for other respiratory allergens also provides a platform for future cat-specific pathways if sponsors dedicate programs and trials. As testing and therapy broaden across urban centers, patient flow into structured, component-informed care is expected to accelerate and reinforce the cat allergy in humans market trajectory.
Competitive Landscape
The cat allergy in humans market shows moderate consolidation, bringing together established suppliers of standardized extracts, diagnostics companies that enable component-informed prescribing, and biologics developers that target faster relief or new adjunct mechanisms. Extract manufacturers operate with vertical control over source materials and extraction chemistries, and reinforce continuity with clinician education that reflects label requirements for dosing, observation, and non-interchangeability between hair and pelt extracts. Method advances like human monoclonal IgE–based immunoenzymetric assays for Fel d 1 content measurement support more precise potency reporting, which can help align dosing interpretations across products. This combination of supply capability, clear labeling, and diagnostics integration provides a stable backbone as new entrants seek to differentiate.
Recent strategic moves underscore this balance between established categories and novel modalities. Regeneron advanced Fel d 1–blocking antibodies through positive Phase 3 results and outlined plans for additional registration-enabling work in 2026, anchoring the pathway for allergen-blocking biologics that can complement or substitute for multi-year extract courses in selected patients. ALK reported robust 2025 financial performance with double-digit revenue growth and margins reflective of sustained demand for immunotherapy tablets in other respiratory allergens, highlighting the operational readiness that could transfer to cat when evidence and filings converge. This mix of steps gives the cat allergy in humans market multiple near- and mid-term innovation levers across biologics, immunotherapy, diagnostics, and home-based adjuncts.
Cat Allergy In Humans Industry Leaders
ALK‑Abelló A/S
Bayer AG
Haleon
Sanofi
Kenvue
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- February 2026: Regeneron presented 36 abstracts at AAAAI, including first-time Phase 3 data for REGN1908 and REGN1909 that target Fel d 1, and announced plans to initiate additional registration-enabling trials in H1 2026.
- October 2025: Cadila Pharmaceuticals launched Dlorfast‑M tablets, a fixed‑dose combination of desloratadine and montelukast, for the treatment of allergic rhinitis and urticaria. The dual‑action therapy targets both histamine and leukotriene pathways and is designed as a once‑daily oral treatment to improve symptom control and patient compliance.
Global Cat Allergy In Humans Market Report Scope
The cat allergy in humans market comprises pharmaceutical and immunological therapies used to prevent, manage, or treat allergic reactions caused by exposure to cat allergens such as dander, saliva, and urine proteins. The market includes symptomatic treatments and disease‑modifying options delivered through various routes and accessed via over‑the‑counter or prescription channels across clinical and home‑care settings worldwide.
The cat allergy in humans market is segmented by product type, including antihistamines, corticosteroids, allergen‑blocking monoclonal antibodies, immunotherapy, and other therapies; by route of administration, comprising oral, intranasal, ophthalmic, subcutaneous, and other routes; by prescription status, covering over‑the‑counter and prescription medications; and by distribution channel, including hospital pharmacies, retail pharmacies, and online pharmacies. By geography, the market is segmented into North America, Europe, Asia-Pacific, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Antihistamines |
| Corticosteroids |
| Allergen-blocking Monoclonal Antibodies |
| Immunotherapy |
| Others (Nasal Decongestant, etc) |
| Oral |
| Intranasal |
| Ophthalmic |
| Subcutaneous |
| Others |
| Over-the-Counter (OTC) |
| Prescription (Rx) |
| Hospital Pharmacy |
| Retail Pharmacy |
| Online Pharmacy |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Antihistamines | |
| Corticosteroids | ||
| Allergen-blocking Monoclonal Antibodies | ||
| Immunotherapy | ||
| Others (Nasal Decongestant, etc) | ||
| By Route of Administration | Oral | |
| Intranasal | ||
| Ophthalmic | ||
| Subcutaneous | ||
| Others | ||
| By Prescription Status | Over-the-Counter (OTC) | |
| Prescription (Rx) | ||
| By Distribution Channel | Hospital Pharmacy | |
| Retail Pharmacy | ||
| Online Pharmacy | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
Key Questions Answered in the Report
What is the current outlook for the cat allergy in humans market?
The cat allergy in humans market size was USD 6.45 billion in 2025 and is projected to reach USD 10.78 billion by 2031 at a 9.22% CAGR over 2026-2031.
Which product type leads and which grows fastest in cat allergy care?
Antihistamines led with 38.43% in 2025, while allergen-blocking monoclonal antibodies are projected to grow at 11.87% CAGR through 2031.
How do routes of administration compare for cat allergy therapies?
Oral formulations held 45.34% in 2025, while subcutaneous routes are forecast to expand at 9.87% CAGR through 2031 as immunotherapy and biologics scale.
What late-stage innovations may change treatment selection?
Fel d 1-blocking monoclonal antibodies have demonstrated sizable symptom reductions in Phase 3 challenge models and are advancing in registration-enabling work during 2026.
Which region currently leads and which is growing fastest?
North America held 42.44% in 2025, while Asia-Pacific is the fastest-growing region with a CAGR of 11.95% through 2031.




