Benzodiazepine APIs Market Size and Share
Benzodiazepine APIs Market Analysis by Mordor Intelligence
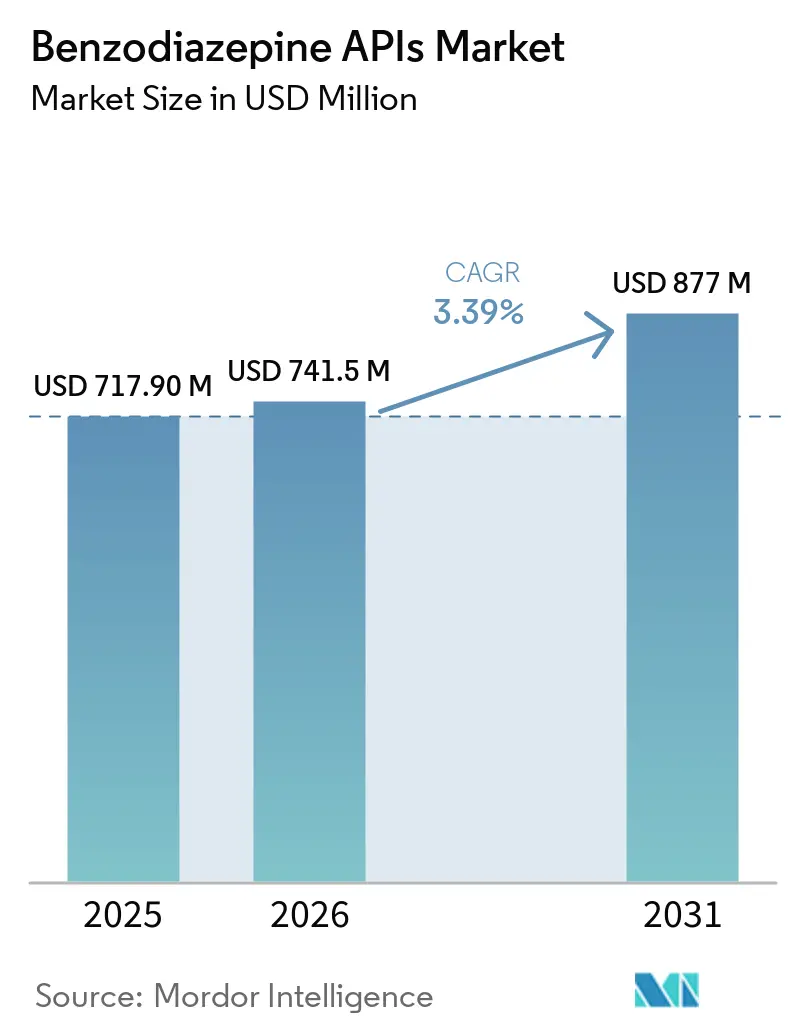
The Benzodiazepine APIs Market size is expected to increase from USD 717.90 million in 2025 to USD 741.5 million in 2026 and reach USD 877 million by 2031, growing at a CAGR of 3.39% over 2026-2031.
Steady therapy demand for anxiety, seizure rescue, and short-acting procedural sedation balances against more restrictive labeling, trade-permit delays, and diversion controls. Ongoing approvals for intranasal diazepam and widening veterinary indications are nudging volumes upward even as chronic outpatient scripts plateau. Capital commitments exceeding USD 150 million apiece by Cambrex, Piramal, and Seqens demonstrate a pivot toward high-compliance capacity with DEA vault infrastructure that meets essential-medicine stockpiling mandates in the United States and Europe. Reshoring incentives under the EU Critical Medicines Act are starting to relocate limited volumes from India and China to Western Europe, but Asia-Pacific retains cost leadership through integrated precursor supply and labor arbitrage.
Key Report Takeaways
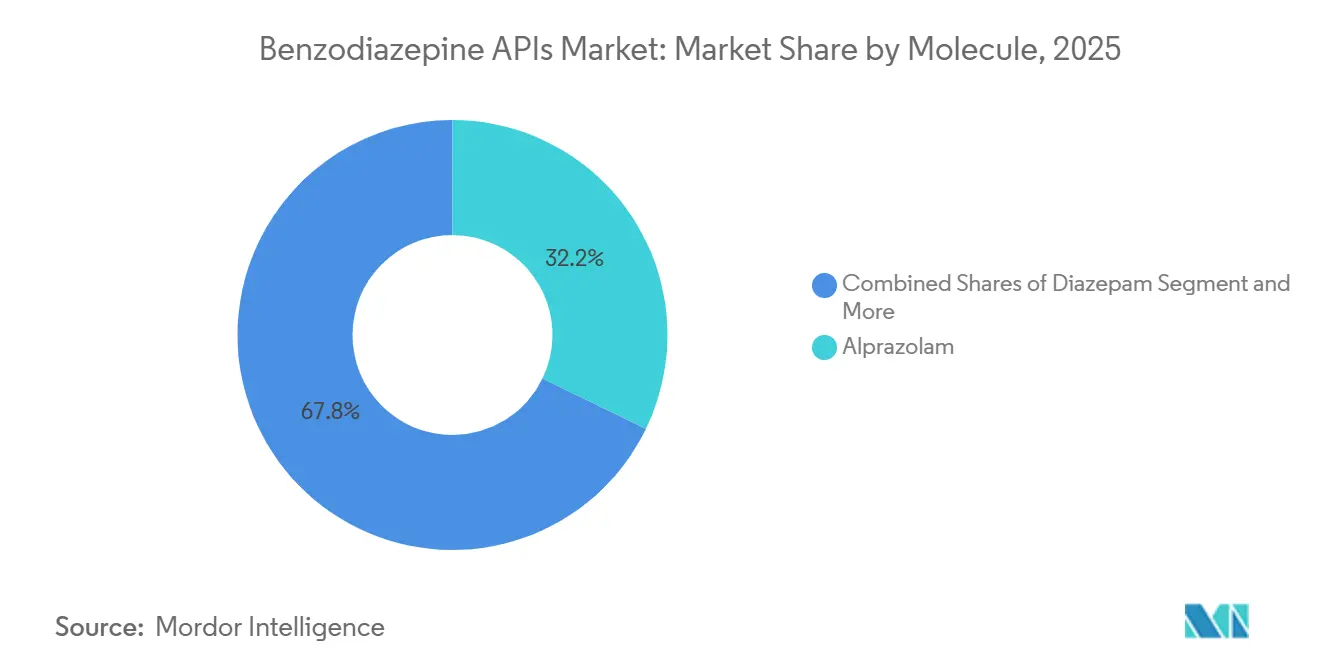
- By molecule, alprazolam led with 32.18% of the benzodiazepine active pharmaceutical ingredients (APIs) market share in 2025, while Midazolam is projected to post the highest 3.87% CAGR through 2031.
- By route of administration, oral dosage forms captured 43.18% of demand in 2025; intranasal delivery is forecast to be the fastest growing at 3.77% CAGR over the same horizon.
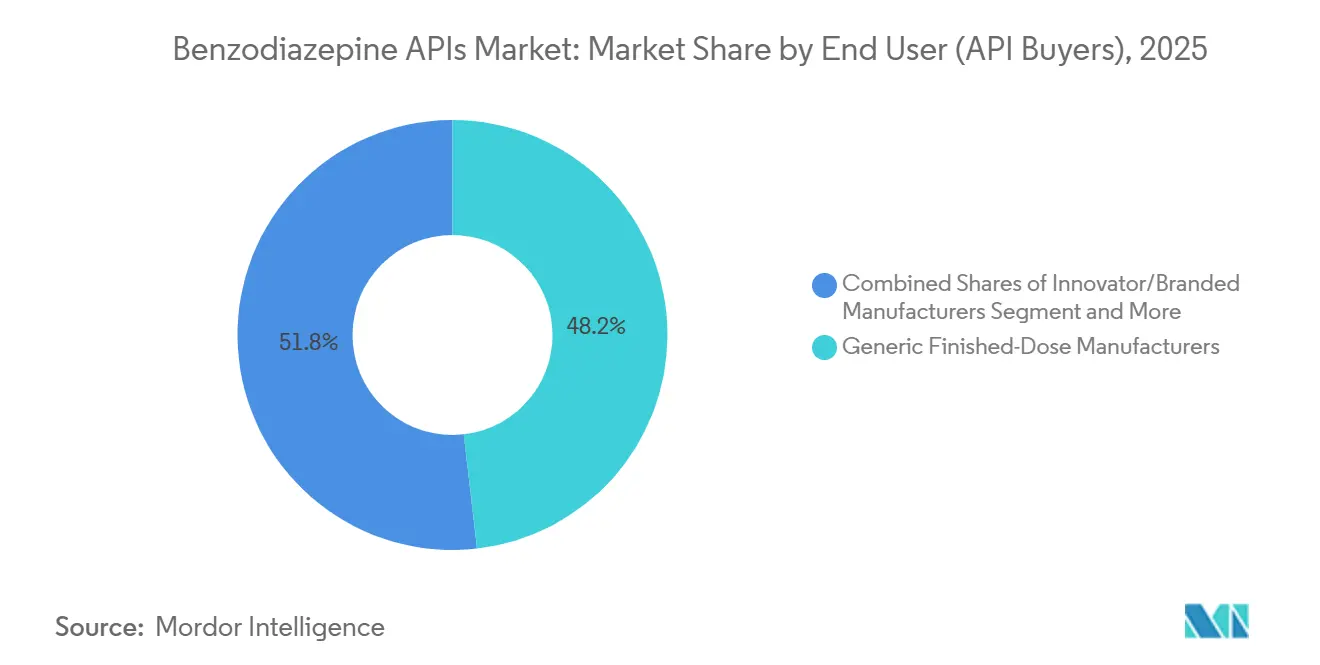
- By end user, generic finished-dose manufacturers accounted for 48.19% of purchases in 2025, yet CDMOs with captive lines are advancing at a 3.75% CAGR to 2031.
- By therapeutic indication, anxiety disorders made up 43.12% of 2025 volumes and will continue to dominate, expanding at 3.63% CAGR to 2031.
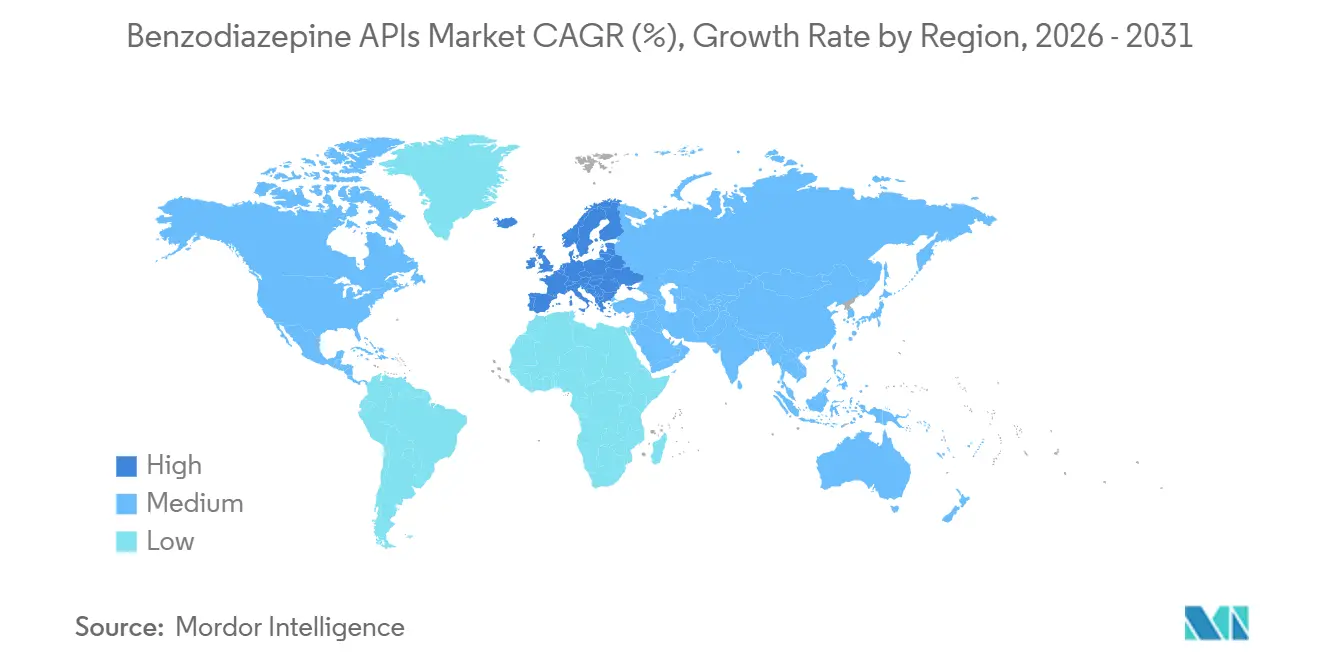
- By geography, Asia-Pacific held 47.15% revenue share in 2025, whereas Europe is on course for the quickest regional expansion at 3.68% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Benzodiazepine APIs Market Trends and Insights
Driver Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Anxiety and seizure burden sustaining core molecules | +0.9% | Global, with concentration in North America, Europe, and urban Asia-Pacific | Long term (≥ 4 years) |
| ICU and procedural sedation need supporting injectable APIs | +0.7% | North America, Europe, Middle East hospital networks | Medium term (2–4 years) |
| Expansion of generic manufacturing and API outsourcing | +0.8% | Asia-Pacific production hubs (India, China), with demand from North America and Europe | Medium term (2–4 years) |
| Veterinary neurology/sedation use cases maintaining niche volumes | +0.3% | North America, Europe, Australia (companion-animal markets) | Short term (≤ 2 years) |
| Essential-medicine stockpiling and reshoring programs are stabilizing demand | +0.5% | Europe (EU Critical Medicines Act), select African and Pacific island nations | Long term (≥ 4 years) |
| Intranasal and rescue-therapy reformulations lifting select APIs | +0.4% | North America (FDA approvals), Europe (EMA pathways), and emerging in the Asia-Pacific | Medium term (2–4 years) |
| Source: Mordor Intelligence | |||
Anxiety and Seizure Burden Sustaining Core Molecules
Anxiety disorders generated major revenue in 2025 demand and remain pivotal as the WHO’s 2025 policy note urged every member state to ring-fence continuous alprazolam and diazepam supplies on essential-medicine lists[1]World Health Organization, “Ensuring Balance in National Policies on Controlled Substances,” who.int. INCB import statistics show 8,766 kg of alprazolam and 33,092 kg of diazepam distributed to 141 countries in 2024, underscoring entrenched clinical reliance. Pediatric seizure rescue is widening the base further; buccal midazolam and rectal diazepam were added to formularies from Zambia to Seychelles last year. ASAM’s 2025 tapering guideline simultaneously preserved the role of short-acting benzodiazepines for acute crises while discouraging months-long regimens. The dual narrative of access and caution is therefore channeling more active pharmaceutical ingredients (APIs) into hospital emergency kits, public stockpiles, and veterinary neurology while tempering large-volume chronic tablet prescriptions.
ICU and Procedural Sedation Needs Supporting Injectable APIs
Midazolam’s projected significant growth stems from its indispensability where seconds matter, bronchoscopy, cardioversion, dental surgery, and alcohol-withdrawal delirium. SCCM’s 2025 critical-care guideline shifted ventilated patients toward propofol or dexmedetomidine, yet kept IV midazolam in first position for refractory agitation[2]Society of Critical Care Medicine, “Pain, Agitation/Sedation, Delirium Guideline,” sccm.org. FDA clearance of ready-to-use intranasal midazolam devices for seizure clusters has spilled over into procedural suites, especially pediatrics, where needle-free access improves throughput and patient comfort. Piramal’s USD 90 million sterile fill-finish upgrade in Michigan and Kentucky directly targets this hospital pipeline, affirming that parenteral benzodiazepines will not exit formularies despite broader sedative substitution.
Expansion of Generic Manufacturing and API Outsourcing
India shipped 84% of its benzodiazepine output in 2025, but 74% of its raw materials crossed the other direction from China, highlighting a fragile precursor loop. CDMOs that can internalize synthesis, formulation, and vault storage are winning tenders from innovators exiting legacy volumes. Cambrex invested USD 120 million in DEA-compliant suites across Iowa and Milan during 2026 to onshore supply for U.S. and EU clients. Supriya Lifescience’s debt-free Ambernath plant came online in January 2026 with 15 U.S. DMFs filed and aims for USD 192 million revenue by 2029, proving that smaller Indian players can capture wallet share by coupling HPAPI know-how with compliance credentials.
Veterinary Neurology/Sedation Use Cases Maintaining Niche Volumes
ACVIM’s 2024 consensus proclaimed intranasal midazolam 70% effective in canine status epilepticus versus 20% for rectal diazepam, accelerating clinic adoption. North American and European pet-ownership rates above 60% are spurring neurologic work-ups that rely on benzodiazepines for both diagnosis and rescue. SeQuent Scientific recorded USD 40.5 million in veterinary API sales for FY2025, supported by a dedicated USFDA-audited Vizag block. Absence of INCB import certificates for most veterinary lines shortens lead times, enabling modest but reliable 4-5% annual volume gains.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Tighter safety warnings and prescribing controls curbing chronic use | -0.6% | North America, Europe (FDA/EMA boxed warnings, ASAM guidelines) | Medium term (2–4 years) |
| International trade/permit frictions for psychotropics | -0.4% | Global, with an acute impact on India-China trade and Africa/Latin America imports | Short term (≤ 2 years) |
| Diversion/abuse risk prompting regional restrictions | -0.3% | North America, Europe, Australia (PDMP monitoring, Schedule IV enforcement) | Long term (≥ 4 years) |
| Supply-chain fragility and precursor constraints | -0.4% | Asia-Pacific (China precursor dominance), Europe (reshoring bottlenecks) | Medium term (2–4 years) |
| Source: Mordor Intelligence | |||
Tighter Safety Warnings and Prescribing Controls Curbing Chronic Use
The 2024 FDA boxed warning on benzodiazepine-opioid co-prescription and ASAM’s 2025 tapering algorithm are compressing refill volumes for alprazolam tablets, previously the workhorse of primary-care anxiety therapy[3]U.S. Food and Drug Administration, “Drug Safety Communication: Benzodiazepine-Opioid Boxed Warning,” fda.gov. Europe followed with prior-authorization hurdles in Spain and France, pushing patients toward SSRIs or SNRIs for generalized anxiety. PDMP systems in 47 U.S. states now flag prescriptions longer than 30 days, disincentivizing family physicians from large scripts. Net impact is a measurable decline in chronic outpatient usage, partially compensated by growth in high-acuity settings.
International Trade and Permit Frictions for Psychotropics
Every cross-border shipment of a Schedule IV benzodiazepine requires an INCB import and export permit. Lags grew to 12 weeks in 2024 when 23 countries failed to file complete statistics, leading to suspended allowances. China’s Ministry of Commerce additionally placed export caps on select aminobenzophenone precursors, lengthening India’s inbound cycle to 14 weeks. DEA’s Operation Zodiac intensified scrutiny of Mexican imports, delaying legal consignments alongside clandestine flows. Smaller generic companies lacking in-house regulatory teams struggle most, conceding volume to CDMOs that maintain dedicated permit desks.
Segment Analysis
By Molecule: Alprazolam Anchors, Midazolam Accelerates
Alprazolam generated the largest 32.18% benzodiazepine active pharmaceutical ingredients (APIs) market share in 2025, yet its growth is flattening under prescribing caps. Continuous-flow chemistry from Purdue has slashed diazepam cost-of-goods by 25%, but commercial adoption remains at pilot scale. Midazolam, aided by intranasal launches, is forecast for a 3.87% pace, the fastest in the set. Diazepam and lorazepam together made up a notable portion of volume, but grapple with intermittent shortages that push hospitals toward midazolam substitutes. Specialty epilepsy molecules such as clobazam are edging upward in orphan-drug niches, whereas older insomnia actives like flurazepam languish amid non-benzodiazepine hypnotic competition.
Commercial economics differ sharply: high-volume tablets carry razor-thin margins, whereas rescue sprays or sterile syringes command premiums for micronized or sterile-filtered intermediates. That bifurcation is visible in investment patterns, Cambrex added controlled-substance flow reactors in Iowa for specialty lots while Indian exporters chase tablet-grade demand. Continuous-flow patents filed during 2024-2026 for lorazepam and clonazepam underscore a race to greener, lower-solvent routes that can satisfy EU sustainability audits once the Critical Medicines Act’s environmental reporting clauses activate in 2027.
Note: Segment shares of all individual segments available upon report purchase
By Therapeutic Indication: Anxiety Dominates, Procedural Sedation Gains
At 43.12% of 2025 usage, anxiety remains the principal indication, buoyed by WHO-endorsed essential-medicine programs even as chronic scripts contract. Epilepsy and cluster seizure rescue represent the next-largest slice, swelling since the FDA widened Valtoco’s label to children aged 2–5 in April 2025. ICU sedation volumes are plateauing because of guideline shifts, yet midazolam holds irreplaceable status for alcohol-withdrawal delirium, keeping a floor under injectable purchases. Procedural sedation, colonoscopy, dental, and minor orthopedic procedures have emerged as the fastest-advancing clinical bucket at 4% plus, thanks to intranasal adoption that circumvents IV lines in day-surgery centers. Muscle spasm, insomnia, and veterinary neurology round out demand at single-digit percentages but carry distinct formulation needs, rectal gels, buccal solutions, flavor-masked liquids, that bolster API premiums.
By End User (API Buyers): CDMOs With Captive Lines Outpace Generics
Generic finished-dose manufacturers still bought the largest slice of benzodiazepine APIs in 2025 at 48.19%, yet contract development and manufacturing organizations (CDMOs) that also run their own finished-dose lines are expanding faster, growing 3.75% a year as sponsors look for one-stop partners that shorten Drug Enforcement Administration paperwork and simplify logistics. Benuvia illustrates the model: the U.S. specialist operates an 83,000-square-foot site that houses DEA-rated vaults and supports synthesis, formulation, and fill-finish capabilities that have already backed more than 500 FDA filings.
Hospitals and 503B compounding centers, which bought notable volumes of APIs in 2025, are also steering volume toward CDMOs that can certify sterile injectables to United States Pharmacopeia standards as regulators tighten oversight of in-house compounding. Innovator companies accounted for notable share of demand, focused on niche products such as Valtoco intranasal diazepam and Onfi clobazam, where patent or orphan status supports premium pricing.
Note: Segment shares of all individual segments available upon report purchase
By Route of Administration Served: Oral Plateaus, Nasal Surges
Demand is shifting toward intranasal delivery, a segment forecast to rise 3.77% a year through 2031 as producers master the micronized powders and high-purity concentrates required for spray devices, expertise that again favors formulation-savvy CDMOs over bulk API traders. Oral products still commanded 43.18% of 2025 volume, led by alprazolam tablets and diazepam capsules, but renewed prescribing limits and the growing use of SSRIs are keeping this channel flat. Parenteral formats, IV and IM injections made up a significant share of demand last year, buoyed by procedural sedation and intensive-care use; Piramal’s USD 90 million expansion in Michigan and Kentucky targets precisely these sterile controlled-substance lines. Nasal therapies are gaining momentum in real-world success stories. Valtoco, Neurelis’s intranasal diazepam, secured FDA clearance in 2020 and added a pediatric label extension down to two years of age in April 2025, an update that helped many families move away from rectal gels thanks to 97% bioavailability and friendlier administration.
Alternative options, such as rectal, buccal, and sublingual routes, hold a modest share of demand, mainly in pediatric or resource-limited settings. Rectal diazepam, once the default rescue agent, is steadily losing share because of slower absorption and lower caregiver acceptance, while European approval of buccal midazolam has prompted the World Health Organization to recommend the dosage form for essential-medicine lists across parts of Africa and the Pacific
Geography Analysis
Asia-Pacific remained the engine at 47.15% of worldwide revenue in 2025, rooted in India’s manufacturing heft and China’s precursor stranglehold. Still, Europe’s 3.68% CAGR is set to outpace every region as reshoring aid and six-month buffer stocks stimulate localized output. The Benzodiazepine APIs market share for Europe is expected to grow significantly under the Critical Medicines Act’s direct procurement clauses.
North America held a notable share in 2025 revenue, sustained by hospital procedural sedation and federally funded emergency reserves. Persistent diazepam and lorazepam shortages in 2024-2026 nudged hospitals to midazolam, triggering Siegfried’s multi-site U.S. acquisition spree in May 2026. Canada and Mexico trail in volume but face heightened border checks under DEA diversion crackdowns.
The Middle East & Africa cluster shows patchy yet vital growth tied to tertiary-care upgrades in Gulf states and expanded essential-medicine lists in Southern Africa. South America’s dependence on Indian imports exposes it to permit oscillations; Argentina endured three months of stock-outs in 2025 after incomplete INCB filings. WHO’s 2025 balance guideline is nudging ministries to execute firmer demand forecasts, smoothing but not eliminating periodic shortages.
Competitive Landscape
A significant share of 2025 sales accrued to the top 10 suppliers, confirming moderate concentration. Cambrex, Piramal, and Seqens each allocated more than USD 150 million during 2025-2026 to controlled-substance suites, indicating a strategic bet on compliance barriers as a durable moat. Indian specialists such as Supriya Lifescience and Solara are pivoting toward HPAPIs to escape commodity margin squeeze, while Europe’s EuroAPI is pruning undifferentiated molecules to focus on opiates and inhalation steroids. Innovator divestitures are flowing into CDMOs that can cradle projects from synthesis through fill-finish under one roof. Benuvia and Veranova exemplify this model, having processed more than 500 FDA filings combined while housing DEA Schedule I–V vaults.
Benzodiazepine APIs Industry Leaders
Cambrex Corporation
Piramal Pharma Limited
SEQENS Group
Supriya Lifescience Ltd.
EUROAPI S.A.S.
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- March 2026: Cambrex started advancing a USD 150 million dual-continent investment to expand its manufacturing capabilities, focusing on DEA-regulated controlled substances, highly potent APIs (HPAPI), and peptide manufacturing.
- January 2025: Supriya Lifescience brought its Ambernath HPAPI block online, aiming for Rs 1,600 crore (USD 192 million) revenue within three years.
- April 2025: FDA widened Valtoco’s indication to cluster-seizure patients aged 2–5, boosting intranasal diazepam demand
Global Benzodiazepine APIs Market Report Scope
As per the scope of the report, benzodiazepine active pharmaceutical ingredients (APIs) are a critical class of synthetic psychoactive substances used to manufacture medications that act as central nervous system (CNS) depressants. Chemically, these APIs are defined by a core structure featuring a benzene ring fused to a seven-membered diazepine ring.
The benzodiazepine APIs market is segmented by molecules, by therapeutic indication served, by end user (API Buyers), route of administration, and by geography. Based on molecules, the market is segmented into alprazolam, diazepam, lorazepam, midazolam, and others. By therapeutic indication served, the market is segmented into anxiety disorders, epilepsy and seizure rescue, ICU sedation and agitation management, procedural sedation/anesthesia, insomnia, muscle spasm and spasticity, preoperative anxiolysis, and veterinary indications. By end users, the market is generic finished-dose manufacturers, innovator/branded manufacturers, hospital outsourcing and compounding centers, CDMOs with captive FDF lines, and veterinary pharmaceuticals manufacturers. By route of administration, the market is segmented into oral, parenteral, nasal, and others
Geographically, the market is segmented into North America, Europe, Asia-Pacific, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Alprazolam |
| Diazepam |
| Lorazepam |
| Midazolam |
| Others |
| Anxiety disorders |
| Epilepsy and seizure rescue (incl. cluster seizures) |
| ICU sedation and agitation management |
| Procedural sedation/anesthesia |
| Insomnia |
| Muscle spasm and spasticity |
| Preoperative anxiolysis |
| Other Therapeutic Indications |
| Generic finished-dose manufacturers |
| Innovator/branded manufacturers |
| Hospital outsourcing and compounding centers |
| CDMOs with captive FDF lines |
| Veterinary pharmaceuticals manufacturers |
| Oral |
| Parenteral |
| Nasal |
| Others |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Molecule | Alprazolam | |
| Diazepam | ||
| Lorazepam | ||
| Midazolam | ||
| Others | ||
| By Therapeutic Indication Served | Anxiety disorders | |
| Epilepsy and seizure rescue (incl. cluster seizures) | ||
| ICU sedation and agitation management | ||
| Procedural sedation/anesthesia | ||
| Insomnia | ||
| Muscle spasm and spasticity | ||
| Preoperative anxiolysis | ||
| Other Therapeutic Indications | ||
| By End User (API Buyers) | Generic finished-dose manufacturers | |
| Innovator/branded manufacturers | ||
| Hospital outsourcing and compounding centers | ||
| CDMOs with captive FDF lines | ||
| Veterinary pharmaceuticals manufacturers | ||
| By Route of Administration | Oral | |
| Parenteral | ||
| Nasal | ||
| Others | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
Key Questions Answered in the Report
What is the current size of the Benzodiazepine APIs market?
The sector was worth USD 741.5 million in 2026 and is projected to reach USD 877 million by 2031, delivering a 3.39% CAGR over 2026-2031
Which molecule accounts for the largest share?
Alprazolam led with 32.18% of 2025 revenue thanks to its widespread use for anxiety, while midazolam is the fastest-growing on the back of intranasal seizure-rescue products
What factors are driving demand for these APIs?
Rising anxiety and seizure cases, steady procedural-sedation volumes, essential-medicine stockpiles, and new intranasal formulations together keep volumes edging higher despite tighter prescribing rules
Which region is expanding most quickly?
Europe shows the briskest pace at 3.68% CAGR through 2031, supported by the EU Critical Medicines Act and related incentives for local production




