Back Table And Cart Covers Market Size and Share
Back Table And Cart Covers Market Analysis by Mordor Intelligence
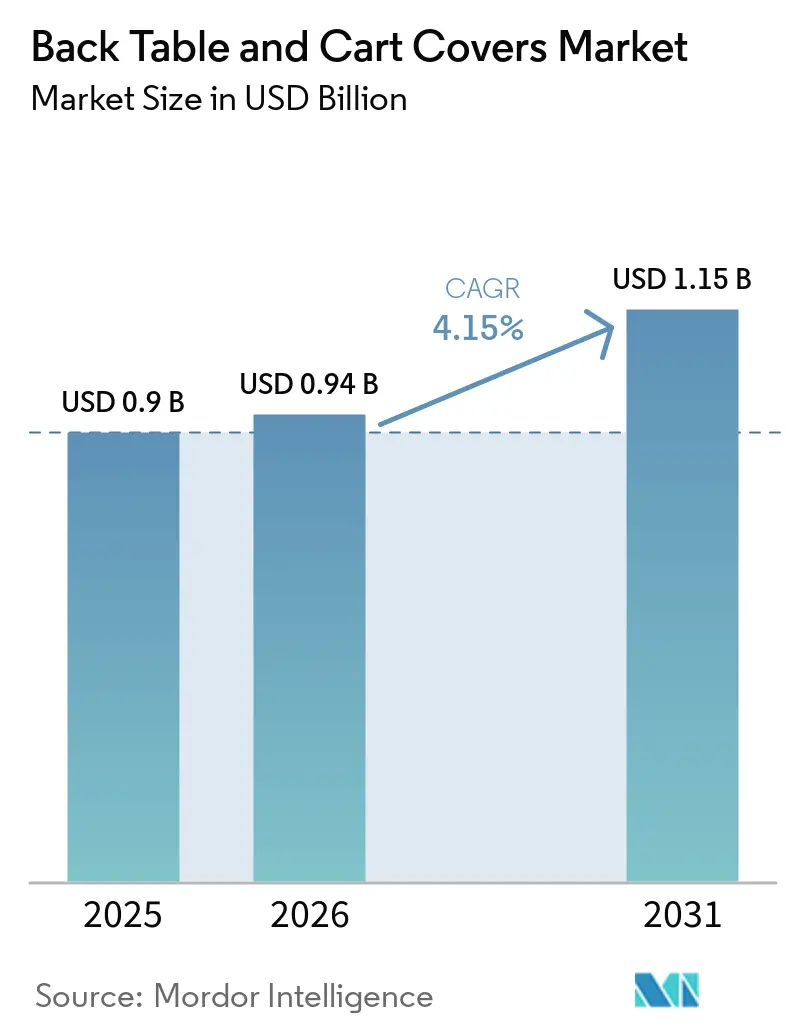
The Back Table And Cart Covers Market size is expected to grow from USD 0.9 billion in 2025 to USD 0.94 billion in 2026 and is forecast to reach USD 1.15 billion by 2031 at 4.15% CAGR over 2026-2031.
Evolving operating room protocols, certification refresh cycles, and sustained investment in sterile-field standardization are keeping demand steady in large hospital systems while sparking faster growth in ambulatory and specialty settings. European performance and safety specification updates, including EN 13795 and EU MDR 2017/745 compliance, continue to tighten product requirements and influence purchasing refreshes as older stock sunsets. Clinical pack vendors and drape specialists are positioning sustainability credentials and expand-at-scale logistics as competitive differentiators, which is supporting orderly market consolidation and multi-year contract awards. Digital procurement and group purchasing models remain central to price transparency and vendor qualification, and online platforms continue to expand their catalog depth and compliance toolsets. Manufacturers in Asia-Pacific are releasing lighter, laminate-backed barrier products targeted at cost-sensitive tenders, a product trend that aligns with broader hospital buildouts and rising procedural throughput in the region.
Key Report Takeaways
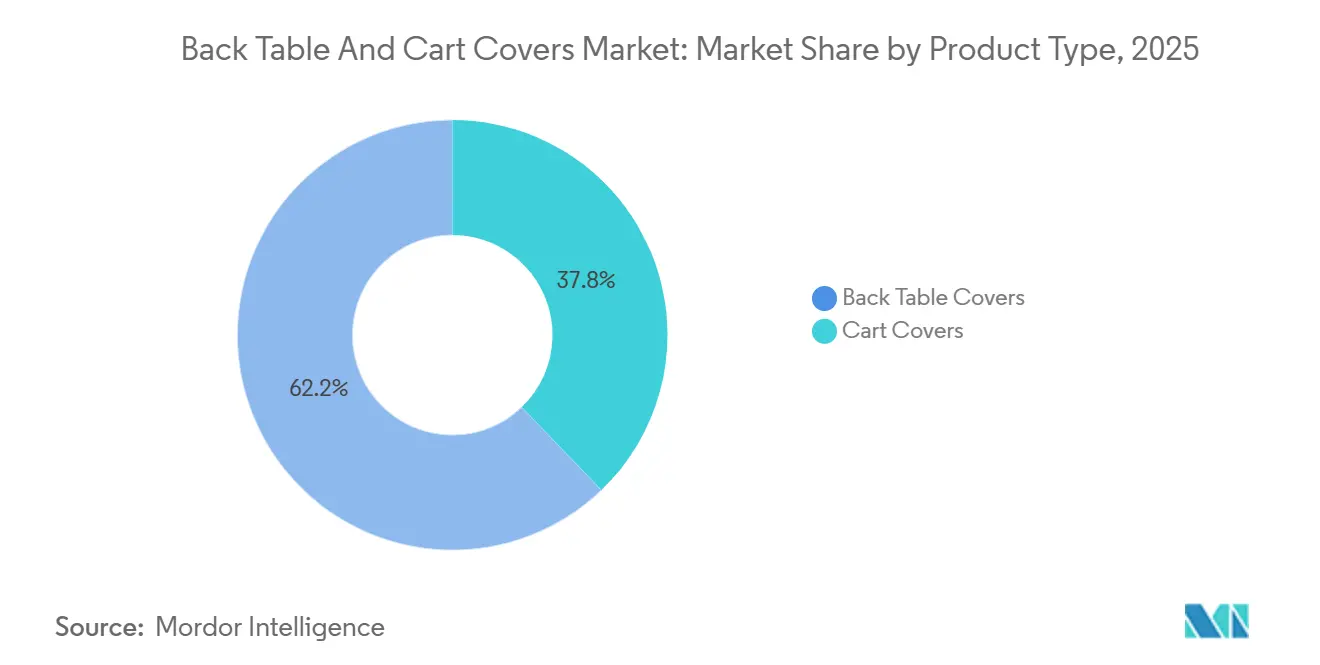
- By product type, back table covers led with 62.19% of the back table and cart covers market share in 2025. Cart covers recorded the highest projected CAGR at 6.45% through 2031.
- By sterility, sterile variants accounted for 65.89% of 2025 demand. The sterile segment is also the fastest growing at a 6.85% CAGR through 2031.
- By material, SMS or PP nonwoven fabric commanded 59.56% in 2025. PE film-laminate barrier materials are the fastest growing at a 5.89% CAGR.
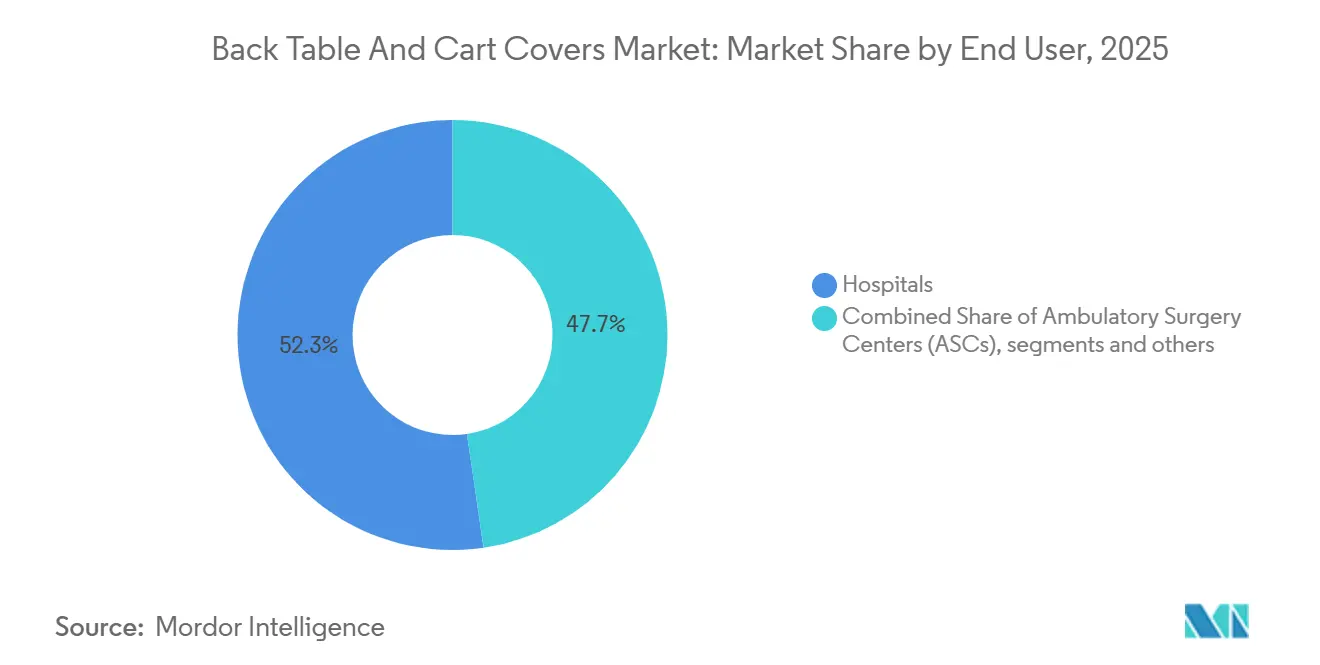
- By end user, hospitals held 52.31% in 2025. Specialty clinics and office-based surgery are growing fastest at a 6.54% CAGR.
- By distribution channel, direct tenders and GPO contracts retained 54.64% in 2025. Online procurement channels are scaling fastest at a 7.12% CAGR.
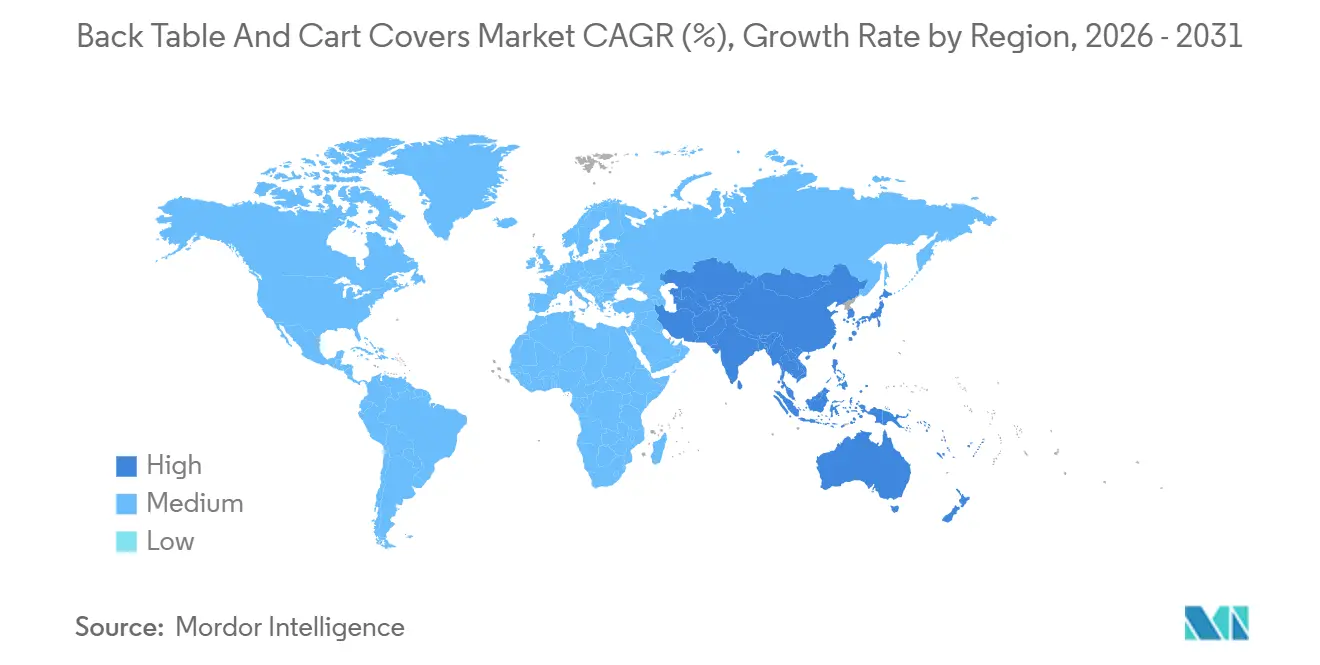
- By geography, North America held 43.44% in 2025. Asia-Pacific is the fastest growing at a 6.19% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Back Table And Cart Covers Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Stricter sterile-field protocols and table-cover use in OR workflows post-2019 guideline updates | +1.2% | Global, with early adoption in North America and Europe | Medium term (2-4 years) |
| Rising outpatient or ASC surgical procedure volumes shifting barrier product demand to high-turnover settings | +0.9% | North America core, expanding to Asia-Pacific | Short term (≤ 2 years) |
| Adoption of custom procedure packs that include back table and instrument table covers | +0.7% | Global, spillover from ASC to specialty clinics | Medium term (2-4 years) |
| Higher penetration of nonwoven SMS or laminate barrier materials in surgical drapes and covers | +0.6% | Global, led by APAC manufacturing hubs | Long term (≥ 4 years) |
| OR turnover-time optimization using standardized table or cart covers | +0.5% | North America and EU acute-care hospitals | Medium term (2-4 years) |
| Sustainability-led redesign with lighter, bio-based laminates catalyzing product refresh cycles | +0.3% | EU and North America, with early regulatory push | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Stricter Sterile-Field Protocols and Table-Cover Use in OR Workflows Post-2019 Guideline Updates
Hospitals and surgical centers are prioritizing barrier performance and packaging integrity in all sterile-field components, which is tightening specifications for back table and cart covers. European facilities are aligning to EN 13795 classifications, which codify standard and high-performance drape requirements and drive procurement toward products that demonstrate repeatable liquid barrier and microbiological protection. EU MDR 2017/745 has also accelerated stock refresh cycles as legacy inventory certified under older frameworks must be retired and replaced with MDR-conformant supplies. These dynamics elevate preference for validated sterile-pack configurations and standardized cover systems that simplify evidence collection for audits and clinical governance reviews. In fluid-intensive service lines, product selection is gravitating to higher-grade barriers and robust tensile performance profiles that maintain integrity over multi-hour procedures and repeated instrument transfers. Together, these trends are reinforcing premium performance tiers within the back table and cart covers market and concentrating spend with suppliers that can document compliance across international standards[1]Kingphar Medical Editorial Team, “Surgical Drapes Specifications: EN 13795 Standards & Procurement Guide 2026,” Kingphar Medical, kingphchina.com.
Rising Outpatient or ASC Surgical Procedure Volumes Shifting Barrier Product Demand to High-Turnover Settings
Procedure migration to ambulatory and office-based settings is rebalancing demand toward single-use cover solutions that enable quick room flips and reduce the need for complex reprocessing workflows. Facilities operating high-throughput schedules favor preconfigured carts and back table draping that keep equipment protected during transit and minimize touchpoints that could compromise sterility. As outpatient capacity expands, procurement teams are adopting digital marketplaces and direct-to-facility channels that streamline replenishment and shorten lead times for standardized barrier components. These channels also make it simpler to filter products by documentation requirements and quality management certifications when small teams manage purchasing alongside clinical tasks. Manufacturers that offer sterile cover packs designed for compact OR footprints and focused specialty workflows are well positioned to capture growth in these settings. This shift supports steady volume gains for disposable cover formats while pushing suppliers to provide reliable availability and elastic capacity for episodic surges.
Adoption of Custom Procedure Packs that Include Back Table and Instrument Table Covers
Custom procedure packs that bundle back table covers, instrument drapes, gowns, and accessories are gaining share because they compress setup time and reduce inventory handling. Vendor strategies increasingly pair sterilization and logistics capabilities under one roof to offer turnkey sterile-field solutions that fit predictable specialty workflows. A leading global drape and pack supplier reports that its pack solutions can cut theatre setup time and simplify administrative tasks, which helps perioperative teams focus on patient-facing work. Consolidation around integrated pack platforms is visible in recent acquisitions that bring sterilization, production, and distribution into a single operating model. Pack providers are also investing in modular, semi-custom configurations that share drape and cover elements across procedures to balance flexibility with compliance. As clinical pathways standardize, these packs become a lever for throughput and consistency, which sustains demand for sterile back table and cart cover components within the bundle.
Higher Penetration of Nonwoven SMS or Laminate Barrier Materials in Surgical Drapes and Covers
SMS or PP nonwoven constructions continue to dominate based on a favorable blend of fluid resistance, breathability, and cost control at the weights commonly specified for sterile covers. Product teams are also deploying lamination to achieve higher barrier performance at lower basis weights, which reduces material intensity and freight costs while preserving strike-through protection. Suppliers in Southeast Asia have released spunbond-laminated PE film materials that target regional hospitals with evolving barrier requirements and price-sensitive tenders. Sustainability features are gaining traction in premium portfolios, including renewable feedstock certification and redesigned components aimed at reduced lifecycle greenhouse gas impact. New cellulosic layers for spunlace platforms further diversify material options and enable alternative configurations for cover subsystems such as fluid-handling zones. Collectively, these materials innovations reinforce the performance spectrum within the back table, and cart covers market, and align with purchasing criteria that weigh barrier, comfort, and environmental attributes.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Environmental pressure favoring reusables or lower-footprint alternatives to single-use covers | -0.8% | EU and North America, driven by EPR mandates | Medium term (2-4 years) |
| Polypropylene or nonwoven raw-material price and availability volatility impacting costs | -0.6% | Global, acute in North America and Europe | Short term (≤ 2 years) |
| Practice variability and conflicting guidance on cover methods complicate adoption and training | -0.4% | Global, particularly in multi-site health systems | Medium term (2-4 years) |
| Growing waste-management and EPR pressures increasing lifecycle cost of disposables | -0.3% | EU and select U.S. states with textile EPR laws | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Environmental Pressure Favoring Reusables or Lower-Footprint Alternatives to Single-Use Covers
Extended Producer Responsibility policies are shaping purchasing strategies by adding per-unit cost signals to disposable textiles and setting the stage for broader lifecycle accounting. In the United States, California’s SB 707 established a statewide textile EPR framework that directly affects how hospitals evaluate single-use versus reusable pathways across defined product categories. Suppliers are responding with lighter-weight mono-material designs and bio-based content to reduce waste and align with health system sustainability targets. Large drape and pack providers now publish sustainability roadmaps and product-level documentation that address recycled or renewable content and greenhouse gas reduction. This evidence base supports hospital initiatives to balance clinical performance, logistics efficiency, and environmental impacts in operating room supply decisions. Over the forecast period, EPR implementation milestones and hospital climate-action commitments will continue to influence design direction and refresh timing for back table and cart cover portfolios.
Polypropylene or Nonwoven Raw-Material Price And Availability Volatility Impacting Costs
Back table and cart covers rely on polymer-based nonwovens and laminates, which link manufacturing costs to upstream petrochemical markets and regional feedstock availability. Periods of tight resin supply, processing outages, or logistics bottlenecks can pressure conversion margins and complicate price commitments on multi-year contracts. Suppliers mitigate exposure by diversifying resin sources, rebalancing basis weights, and applying lamination only where higher barrier performance is essential. Portfolio strategies also include alternative fiber layers and process innovations that maintain barrier targets while reducing polymer intensity at the component level. Distributors that serve hospitals and surgical centers buffer these swings through inventory programs and demand planning, yet price normalization can lag when supply chains remain constrained. Procurement teams, especially those operating through group contracts, continue to refine index-based adjustment clauses to share risk while ensuring continuity of sterile cover supply for high-acuity service lines.
Segment Analysis
By Product Type: Cart Covers Surge as OR Turnover Optimization Gains Traction
Back table covers held 62.19% in 2025 and remain foundational as facilities standardize sterile-field setup across general surgery, orthopedics, and cardiac programs. Cart covers are the fastest-growing product at a 6.45% CAGR through 2031, supported by pre-set sterile carts that move directly into operating rooms and reduce setup steps that can stretch room reset times. The back table and cart covers market benefits from this operational focus because streamlined draping lowers preparation variability and helps smaller teams sustain throughput during peak schedules. In settings that run compact rooms and rely on predictable case sequences, the appeal of dedicated cart covers is rising along with pre-bundled pack usage. Product specifications continue to converge toward international drape norms, and purchasing committees prioritize solutions with validated barrier and tensile ratings aligned to surgical risk categories.
As tendering cycles proceed, hospitals balance the unit price of single-use cart covers with the staffing and logistics savings associated with faster room flips. Product decisions also factor storage convenience, packaging integrity, and compatibility with sterile-pack programs that include related components. Suppliers that offer cart covers optimized for high-fluid specialties retain an edge when case mix demands higher-grade barrier performance. Regional sourcing strategies use laminate-backed or spunbond-heavy designs to meet diverse budgets and standards without complicating inventory. The back table and cart covers market size for cart covers is projected to expand in line with standardized turnover initiatives that favor preconfigured barrier solutions across ambulatory and inpatient sites.
By Sterility: Sterile Variants Dominate, Yet Non-Sterile Covers Serve Cost-Sensitive Preparatory Roles
Sterile back table and cart covers accounted for 65.89% in 2025 and lead growth at a 6.85% CAGR over the forecast period. This outcome reflects hospitals’ zero-tolerance approach to contamination risk in direct patient-contact zones and the administrative need for lot traceability. Non-sterile covers continue to address preparatory and transport tasks where immediate patient exposure is not present, which provides a cost lever during budget-constrained cycles. Clinical governance trends support sterile products for high-acuity procedures and encourage systematic documentation across draping and sterile barrier workflows. In markets where facility licensing references medical device classifications, sterile cover variants align with stricter oversight and structured conformity assessment steps under standard frameworks.
As EPR policies scale and lifecycle costs face scrutiny, procurement teams may evaluate partial shifts to reusable sterile textiles for select high-volume procedures. The policy direction in California is notable because it applies per-unit costs to covered single-use textiles while positioning reusables differently under the program’s fee structure. Product roadmaps from leading vendors show investment in packaging and materials aimed at lighter designs and renewable content, an approach that lowers disposal volumes and strengthens the clinical-economic case for single-use sterility in high-risk procedures. Over the forecast horizon, sterile back table and cart cover adoption is anchored by clinical policy, survey readiness, and pack standardization, while non-sterile cover demand remains stable in supporting roles. The back table and cart covers market continues to reflect this dichotomy as sites tune sterile allocations to case complexity and infection control targets.
By Material: SMS Nonwoven Leads, While PE Film-Laminate Gains on Fluid-Intensive Specialties
SMS or PP nonwoven fabric held 59.56% in 2025, consistent with widespread preference for breathable barrier solutions that meet target performance while maintaining affordability. Facilities deploying standard-performance drapes in moderate-fluid cases use SMS as a baseline, and purchasing teams align specs to operating room comfort and liquid resistance prerequisites. PE film-laminate covers are the fastest-growing material at a 5.89% CAGR through 2031 because their construction provides strike-through protection for high-fluid procedures at lower basis weights. Laminates bond a thin film to spunbond layers to improve barrier without unduly increasing stiffness, which supports handling during rapid instrument transfers on back tables and carts. Southeast Asian manufacturers have introduced new laminate-backed options that address regional tender requirements and enable budget-focused facilities to acquire higher barrier performance at competitive prices.
Premium suppliers are incorporating renewable feedstock into drape and cover materials to reduce greenhouse gas intensity and document sustainability performance to hospital buyers. New wetlaid cellulose layers designed for spunlace platforms expand the menu of complementary components that can be included within multi-layered cover systems. Product teams are also refining how laminate edges, seals, and folds are constructed to maintain barrier integrity while keeping packs compact for storage efficiency. In specialty procedures that entail large fluid volumes, hospitals lean toward laminate-backed options, while SMS remains the backbone for shorter and moderate cases. The back table and cart covers market size associated with SMS-based designs corresponds to the largest installed base, while laminate-backed designs capture a rising share due to performance-driven replacements and specialty program growth[2]WZXINYU Technical Team, “Innovative Applications of Laminated Non-Woven Fabric in Medical Textiles,” WZXINYU Non-woven, wzxinyu.com.
By End User: Hospitals Anchor Demand, Yet Specialty Clinics Accelerate on Outpatient Migration
Hospitals held 52.31% in 2025, which mirrors their case mix in high-acuity disciplines and continuous operating schedules that require dependable sterile cover availability. System-level procurement through group purchasing and committed contracts continues to drive SKU standardization and consistent pricing across affiliated facilities. Academic medical centers and integrated networks value vendor capabilities that fuse clinical documentation, sustainability attributes, and reliable distribution. Specialty clinics and office-based surgery are the fastest-growing end-user group at a 6.54% CAGR because clinicians shift defined procedures to lower-cost settings that prize rapid room turnover and simple supply logistics. For these sites, single-use back table and cart covers reduce the need for complex reprocessing programs and ease staff training burdens tied to sterile workflows.
Mid-market distributors and online platforms that connect manufacturers to verified care providers support this migration through responsive fulfillment and clear compliance documentation. Vendors that simplify ordering and provide evidence packets for audits maintain preferred status with small teams that juggle procurement and clinical duties. Hospital systems continue to rely on national distributors and prime-vendor arrangements that stabilize inventory and buffer demand surges. The back table and cart covers industry serves both ends of the spectrum by offering hospital-grade sterilization documentation and streamlined e-commerce models for smaller practices. Growth in hybrid facilities that co-locate inpatient and outpatient services further expands the use cases where standardized covers and packs create continuity across procedure settings.

By Distribution Channel: GPO Contracts Dominate, Online Channels Scale Rapidly
Direct tenders and group purchasing organization contracts retained 54.64% in 2025 as hospitals consolidate purchasing to secure predictable pricing and supply assurance from vetted vendors. These arrangements reinforce catalog standardization and deepen relationships with manufacturers that can service multi-year commitments and publish the required compliance documentation. Suppliers differentiate with service levels, sustainability profiles, and co-developed forecasting that reduce stockouts for critical sterile covers. Online procurement is the fastest-growing channel at a 7.12% CAGR over 2026-2031 as health systems digitize requisitions and centralize spend management. Growth in digital marketplaces is tied to product discovery, contract compliance controls, and integrations that reduce manual effort in sourcing and invoice matching.
Federal supply platforms and prime-vendor portals enhance visibility into approved items and help care providers maintain compliant purchasing across sites. As suppliers expand direct-to-facility participation, distributor roles evolve toward value-added services, including forecasting, backorder alternatives, and recall coordination. Enterprise procurement teams push for analytics that map utilization and contract adherence, and vendors respond by publishing product attributes and certification data for faster validation. The back table and cart covers market continues to benefit from tighter digital control of purchasing flows as hospitals compress rogue spend and ensure standardized sterile-field inputs. Technology pilots that centralize substitution approvals and automate communications demonstrate how digital procurement can lift resiliency for device categories that rely on consistent barrier performance.
Geography Analysis
North America held 43.44% in 2025 and maintains leadership based on a broad base of hospital infrastructure, a deep roster of acute-care and specialty programs, and long-standing group purchasing frameworks that enforce standardization. Updates to sterile-field expectations and multi-year contracting patterns combine to keep short-cycle replacement steady for back table and cart cover lines, while sustainability plans encourage material redesigns. Public-sector procurement and veteran health systems also reinforce vetted product lists and supply assurance through federal schedules and daily-updated contract catalogs. Over the forecast period, stable inpatient volumes and steady outpatient migration sustain balanced growth across hospital systems and independent facilities. The back table and cart covers market in North America continues to reward suppliers that pair manufacturing scale with documentation depth and timely distribution performance.
Asia-Pacific is the fastest-growing geography at a 6.19% CAGR over 2026-2031 as hospital capacity expansions, clinical safety adoption, and local production of nonwovens widen availability. Regional producers are releasing laminate-backed covers that match evolving barrier expectations while keeping unit costs aligned to public and private tender dynamics. These introductions allow facilities to align product selection to case complexity, with SMS formats for moderate procedures and laminate-backed covers for higher fluid volumes. As new hospitals come online, standardized pack approaches that bundle back table and cart covers are gaining acceptance because they simplify logistics and training. Growth is strongest where suppliers provide complete documentation sets and support qualification under multiple regulatory regimes across the region.
Europe remains steady as hospitals align to EN 13795 and sustain procurement programs that prioritize consistent barrier performance and documented conformity. MDR 2017/745 compliance deadlines have driven stock refresh and codified expectations around drape and cover performance, which benefits manufacturers with robust quality systems and audited production. Regional suppliers with vertically integrated capabilities are positioned to serve country-specific tendering and to hedge manufacturing footprints in support of resilience goals. Ongoing investments by premium producers in capacity, geographic reach, and sustainability attributes have supported continuity for sterile-field consumables across major EU health systems. The back table and cart covers market in Europe will continue to reflect these structural supports as procurement teams balance clinical outcomes, lifecycle documentation, and operational efficiency.
Competitive Landscape
The competitive field includes multi-national medical supply companies with broad drape and pack portfolios and regional specialists that focus on targeted cover formats or niche configurations. Global manufacturers emphasize sterile-pack completeness, performance documentation, and continuously improved sustainability attributes to meet hospital sourcing criteria. Regional challengers differentiate with price positions, laminate-backed innovations, and responsiveness to local tender documentation. Private investment activity has reshaped distribution and production footprints for device portfolios connected to surgical pack ecosystems, reflecting the value of vertically integrated sterilization-to-delivery models. Suppliers that integrate digital forecasting and purchasing collaboration tools into prime-vendor arrangements can tilt awards in their favor as health systems prioritize reliability and analytics capabilities. In this environment, the back table and cart covers market rewards platforms that unite manufacturing scale, quality management rigor, and service intensity.
Strategic moves illustrate the push toward consolidation and capability expansion. Platinum Equity completed the acquisition of a major medical products distribution business in December 2025, with plans to invest in fulfillment, proprietary products, and supply-chain enhancements that improve efficiency and resilience for hospitals and health systems. A European medical textiles and pack specialist acquired a vertically integrated custom pack manufacturer that unites production, sterilization, and distribution, highlighting the strategic importance of end-to-end sterile-field capabilities. Leading drape manufacturers have also announced multi-country capacity expansions and regional manufacturing initiatives to support their procedure-pack businesses and hedge supply chains. These actions enable suppliers to meet MDR-aligned documentation needs in Europe and to serve growth markets in the Middle East and Asia with local production.
Digital supply solutions and procurement partnerships are another axis of competition, as providers adopt tools that reduce sourcing friction and raise transparency. A major distributor and manufacturer introduced an AI-enabled control tower that centralizes product substitutions and approvals for prime-vendor customers, which drives contract compliance and accelerates service recovery during supply disruptions. Marketplace platforms connecting manufacturers directly to care providers are expanding catalogs and verification mechanisms that lower barriers to entry for smaller facilities. These digital features complement national and federal procurement platforms that list eligible products and assist buyers with daily-updated contract data. Vendors that plug into these networks with clear environmental attributes, audited certifications, and strong service levels are advantaged during renewals and new awards. The back table and cart covers market is likely to see continued digital differentiation layered on top of core product and logistics performance.
Back Table And Cart Covers Industry Leaders
Medline Industries
Owens & Minor (HALYARD)
Cardinal Health
Lohmann & Rauscher (L&R)
Paul Hartmann AG
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2026: Lohmann & Rauscher made a successful investment in Ethiopia through its subsidiary L&R Ethiopia, aimed at strengthening the company's global presence, crisis resilience, and social sustainability as part of its ongoing geographic expansion strategy.
- December 2025: Platinum Equity completed the acquisition of Owens & Minor's Products & Healthcare Services division for USD 375 million in cash, with the seller retaining a 5% equity stake. The transaction preserved tax attributes exceeding USD 150 million and repositioned Owens & Minor (rebranded Accendra Health) as a pure-play home-based care provider. Platinum plans to strengthen the acquired unit's fulfillment performance, accelerate proprietary product growth, and invest in supply-chain enhancements to improve efficiency and resiliency across its national medical/surgical distribution platform serving hospitals and health systems.
- September 2025: Medline announced a collaboration with Northwestern Medicine and Providence to develop Mpower™, a next-generation AI-powered supply-chain solution built on Microsoft 365 and Azure AI. The platform, which will be available at no cost to Medline Prime Vendor customers using Mpower Foundations, functions as an AI digital control tower for product substitutions, approvals, and communications, aiming to elevate supply-chain intelligence and resiliency while simplifying procurement workflows for caregivers.
Global Back Table And Cart Covers Market Report Scope
As per the report’s scope, back table and cart covers, often crafted from nonwoven materials, are single-use medical drapes utilized in operating rooms. Their primary purpose is to establish and maintain a sterile environment on operating tables, effectively preventing bacterial transfer and reducing the risk of surgical site infections (SSIs). Additionally, these covers provide protection for instrument trays, medical equipment, and sterile supply storage. The back table and cart covers is segmented into product type, sterility, end user, distribution channel, and geography.
Based product type, the market is segmented into back table covers and cart covers. Based on sterility, the market is segmented into sterile and non-sterile. Based on Material, the market is segmented into SMS/PP nonwoven, PE film-laminate, spunlace/rayon nonwovens, and reusable textile (polyester/cotton). Based on end user, the market is segmented into hospitals, ambulatory surgery centers (ASCs), and specialty clinics / office-based surgery. Based on distribution channel, the market is segmented into direct tenders / GPO contracts, distributor sales, and online. By geography, the market is segmented as North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| Back Table Covers |
| Cart Covers |
| Sterile |
| Non-Sterile |
| SMS/PP Nonwoven |
| PE Film-Laminate |
| Spunlace/Rayon Nonwovens |
| Reusable Textile (Polyester/Cotton) |
| Hospitals |
| Ambulatory Surgery Centers (ASCs) |
| Specialty Clinics / Office-based Surgery |
| Direct Tenders / GPO Contracts |
| Distributor Sales |
| Online |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Back Table Covers | |
| Cart Covers | ||
| By Sterility | Sterile | |
| Non-Sterile | ||
| By Material | SMS/PP Nonwoven | |
| PE Film-Laminate | ||
| Spunlace/Rayon Nonwovens | ||
| Reusable Textile (Polyester/Cotton) | ||
| By End User | Hospitals | |
| Ambulatory Surgery Centers (ASCs) | ||
| Specialty Clinics / Office-based Surgery | ||
| By Distribution Channel | Direct Tenders / GPO Contracts | |
| Distributor Sales | ||
| Online | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the expected growth trajectory for the back table and cart covers market through 2031?
The category is projected to grow from USD 0.90 billion in 2025 to USD 1.15 billion by 2031 at a 4.15% CAGR over 2026-2031.
Which product types are set to lead growth within this space?
Back table covers remain the largest by revenue, while cart covers are forecast to grow fastest at a 6.45% CAGR due to standardized pre-set carts that speed room turnover.
How do material choices affect performance and purchasing in the back table and cart covers market?
SMS or PP nonwovens lead on breathability and value, while PE film-laminates capture fluid-intensive procedures by delivering higher barrier performance at lower basis weights.
Where is regional growth strongest for back table and cart covers?
Asia-Pacific shows the fastest expansion at a 6.19% CAGR as hospitals add capacity and local suppliers release laminate-backed covers tuned to regional tender needs.
How are sustainability requirements influencing purchasing decisions?
Buyers are prioritizing lighter mono-material designs and renewable-content options that document greenhouse gas reductions, supported by supplier programs and EPR policy trends.
Which distribution channels are most influential for procurement?
Direct tenders and GPO contracts remain the largest share, while online procurement channels are scaling quickly with tools for contract compliance and faster replenishment.
Page last updated on:









