Asthma Biologics Market Size and Share
Asthma Biologics Market Analysis by Mordor Intelligence
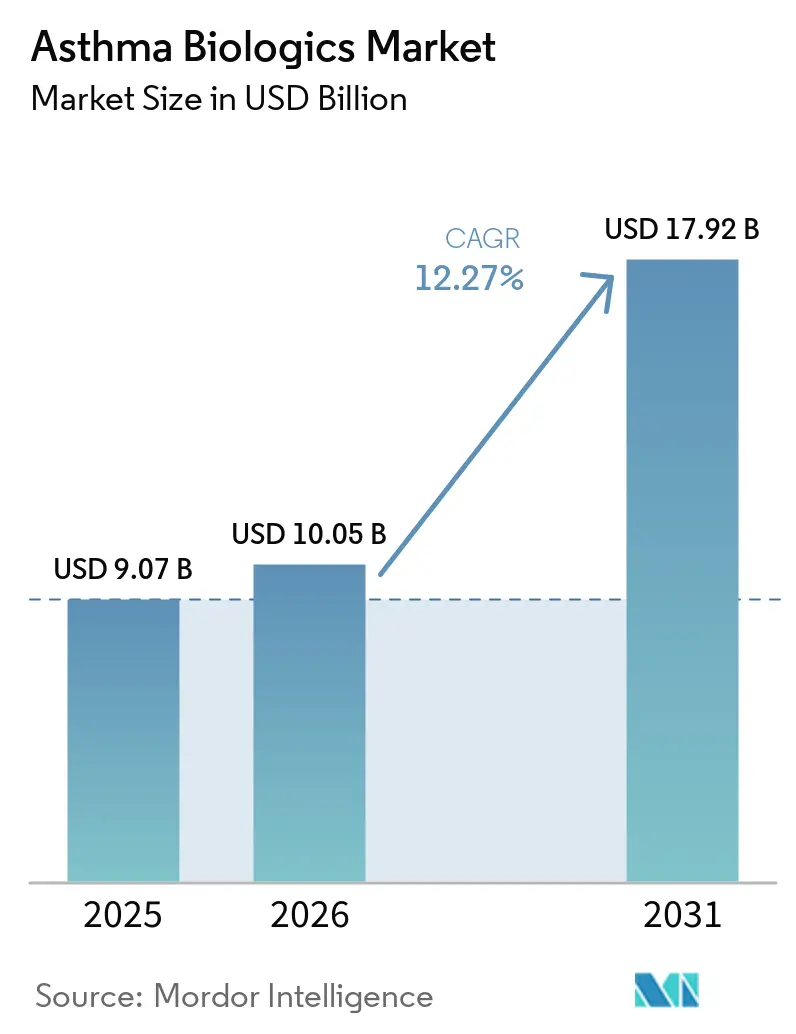
The Asthma Biologics Market size was valued at USD 9.07 billion in 2025 and is estimated to grow from USD 10.05 billion in 2026 to reach USD 17.92 billion by 2031, at a CAGR of 12.27% during the forecast period (2026-2031).
Record specialist adoption follows the 2024-2025 Global Initiative for Asthma (GINA) updates that moved biologics to Step 5, legitimizing earlier use and shortening the time patients spend on chronic oral corticosteroids [1]Global Initiative for Asthma, “GINA 2025 Report,” ginastma.org. Convenience engineering is now a core battleground: GSK’s twice-yearly depemokimab and Celltrion’s interchangeable omalizumab biosimilar illustrate how longer dosing intervals and lower prices can rival efficacy as sources of competitive differentiation. Multi-indication labeling, illustrated by AstraZeneca’s tezepelumab expansion into nasal polyps, is widening prescribing beyond pulmonology and accelerating volume growth. Real-world studies such as REALITI-A and RAPID confirm significant exacerbation reductions, strengthening payer confidence and supporting broader reimbursement.
Key Report Takeaways
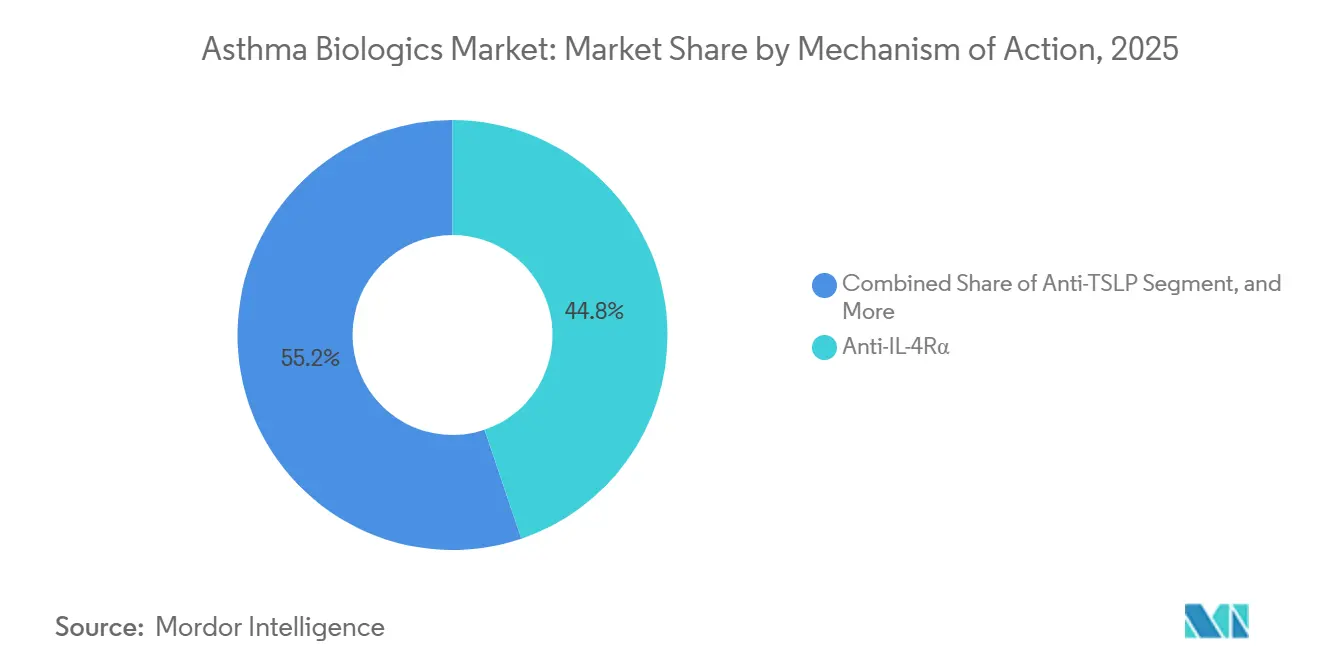
- By mechanism of action, anti-IL-4Rα agents led the asthma biologics market with 44.80% market share in 2025, and Anti-TSLP is expected to advance at a 14.49% CAGR through 2031.
- By phenotype, eosinophilic asthma accounted for 38.65% of the asthma biologics market size in 2025 and is advancing at a 13.65% CAGR through 2031.
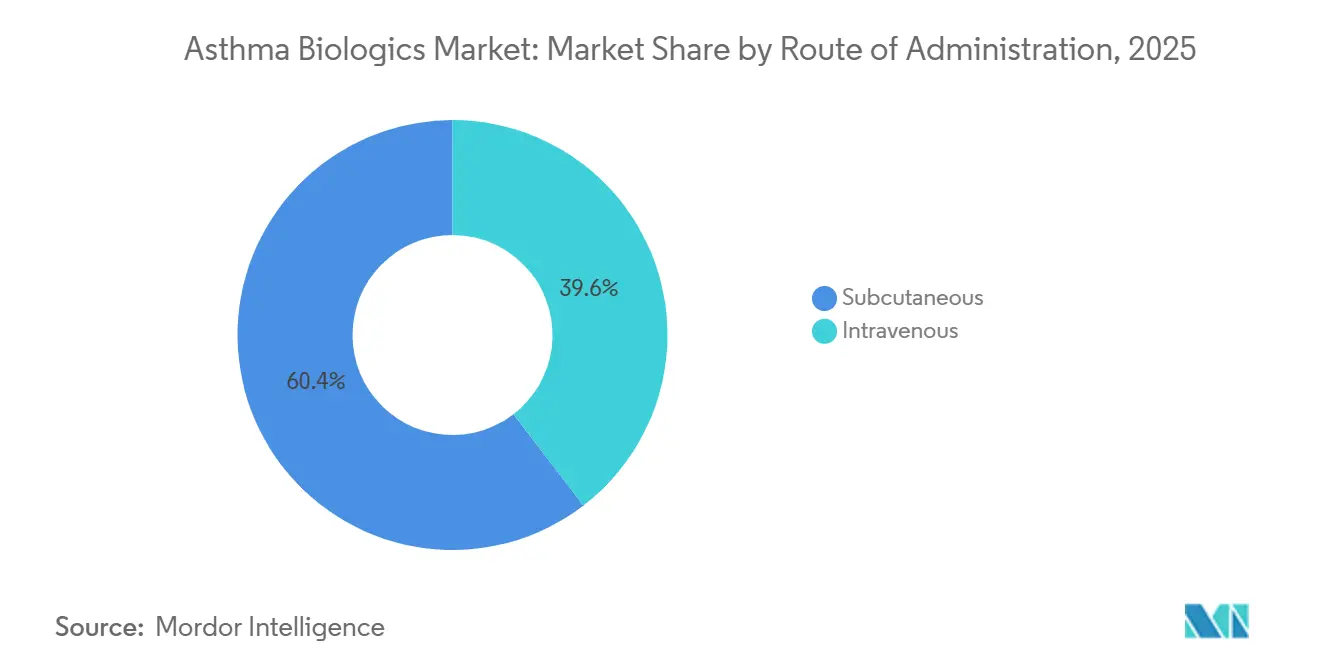
- By route of administration, subcutaneous delivery accounted for 60.45% of the asthma biologics market in 2025 and is expanding at the same 15.11% CAGR through 2031.
- By end user, hospitals accounted for 49.03% of revenue share in 2025, while Specialty Clinics recorded the highest projected growth with 14.23% CAGR.
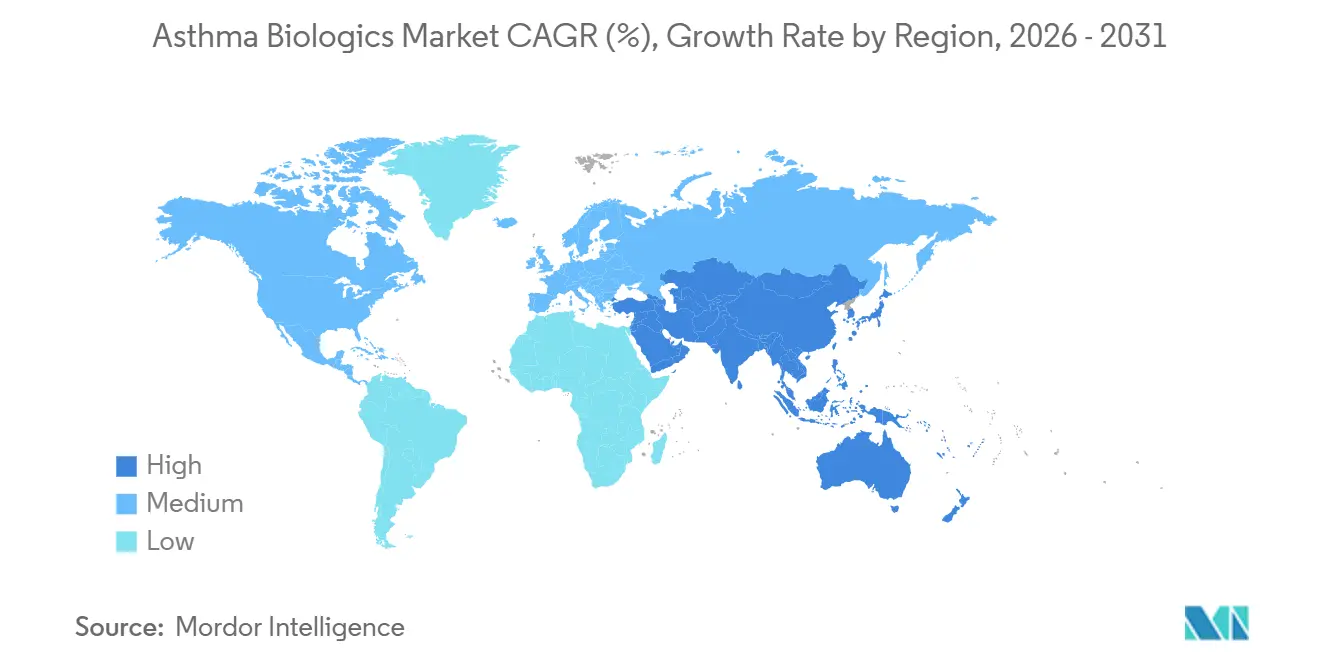
- By geography, North America accounted for 45.12% of revenue in 2025; Asia-Pacific is forecast to grow fastest at a 14.12% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Asthma Biologics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| GINA-endorsed add-on biologics for severe uncontrolled asthma | +2.8% | Global, strongest in North America & Europe | Medium term (2-4 years) |
| Broad-eligibility agents (TSLP pathway) expanding treatable pool | +2.3% | Global, fastest in Asia-Pacific | Medium term (2-4 years) |
| Pediatric labels and home-use pens advancing uptake | +1.9% | North America & Europe lead | Short term (≤ 2 years) |
| Real-world evidence reinforcing payer confidence | +1.7% | Global | Medium term (2-4 years) |
| Ultra-long-acting IL-5 biologics cutting injection burden | +1.4% | North America & Europe first | Long term (≥ 4 years) |
| Co-morbidity overlap driving multi-specialty prescribing | +1.2% | Global | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
GINA-Endorsed Add-On Biologics for Severe Uncontrolled Asthma
GINA’s 2024-2025 revisions positioned biologics as the standard Step 5 option, giving pulmonologists a guideline-backed rationale to start biologics sooner and decrease prolonged oral corticosteroid exposure. Registry data show 50-70% exacerbation drops within six months of mepolizumab or dupilumab initiation [2]European Respiratory Journal Editors, “REALITI-A Registry Outcomes,” erj.ersjournals.com. FDA approval of benralizumab for eosinophilic granulomatosis with polyangiitis in 2024 reinforced class benefits across multiple Type 2 diseases. Payers interpret these outcomes as cost offsets because emergency visits decline when exacerbations fall. However, GINA’s biomarker emphasis favors markets with reliable eosinophil and FeNO testing, slowing penetration in regions with limited laboratory capacity.
Broad-Eligibility Agents Expanding Treatable Population Beyond Biomarker-High Patients
Tezepelumab’s thymic stromal lymphopoietin blockade demonstrated 56% overall and 41% biomarker-low exacerbation reduction in the NAVIGATOR trial, creating the first biologic option for 25-30% of severe asthma patients who lack elevated eosinophils or IgE [3]New England Journal of Medicine Staff, “Tezepelumab in Severe Asthma—NAVIGATOR Study,” nejm.org. The October 2025 FDA label for chronic rhinosinusitis with nasal polyps further broadens use among ENT specialists. Absence of a predictive biomarker complicates payer value models, prompting six-month reassessment rules, yet early uptake data suggest clinicians are willing to trial the drug in non-eosinophilic cases when alternatives are limited. Growth in Asia-Pacific is poised to accelerate once phenotyping infrastructure becomes more accessible and regional payers adopt value-based coverage.
Pediatric Label Expansions and Self/At-Home Administration Improving Uptake
Between 2024 and 2025, regulators cleared dupilumab down to age six months for atopic dermatitis and six years for asthma; mepolizumab and benralizumab won approvals for children aged six and above. Early biologic intervention reduces growth-stunting from systemic steroids. Parallel device innovation—pre-filled pens and autoinjectors allow after-training home dosing, shrinking clinic visit time by more than three hours per injection. A 2025 UK study found 18% higher 12-month persistence with autoinjectors than vial-and-syringe use. Regulations in the US and EU facilitate home use after initial monitoring, but Japan and several Southeast Asian countries still require clinic administration, limiting convenience-driven growth in those markets.
Real-World Evidence Strengthening Payer Confidence
Registries such as REALITI-A for mepolizumab reported fewer clinically significant exacerbations and significantly lower maintenance steroid doses at 12 months. Dupilumab’s RAPID registry showed 70% of patients halving exacerbation rates, with 40% eliminating oral steroids. The Institute for Clinical and Economic Review classified three biologics as “high value” for patients with eosinophil counts above 300 cells/μL in its 2025 update. Several US commercial payers responded by easing prior authorization when biomarker thresholds are met. Insurers in Europe mirror this trend, citing lower hospitalization costs and improved productivity.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High prices and payer step edits/prior authorization | -1.8% | Global, most acute in US & emerging markets | Short term (≤ 2 years) |
| Safety monitoring requirements curb site-of-care flexibility | -1.3% | Global | Medium term (2-4 years) |
| Limited biomarker infrastructure in resource-constrained regions | -0.9% | Asia-Pacific, Latin America, MEA | Long term (≥ 4 years) |
| IV infusion burden versus subcutaneous options | -0.7% | Global | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
High Prices and Payer Step Edits Restrict Access and Delay Initiation
Annual biologic costs span USD 30,000-40,000, prompting US insurers to demand documented trials of inhaled steroids and often oral corticosteroids before approval. A 2024 claims review showed 22-28% initial denial rates, extending treatment start by an average of 45 days. Each extra month on steroids lifts fracture risk by 12% and diabetes incidence by 8%. Medicaid applies even tighter rules, while emerging markets rely on out-of-pocket spending except where price-discounted reimbursement.
Safety and Monitoring Constraints Limit Site-of-Care Flexibility
FDA and EMA mandate supervised first doses with 30-60-minute observation owing to anaphylaxis risk. Reslizumab’s IV infusion requires observation at every dose, limiting its appeal relative to subcutaneous peers and contributing to double-digit sales decline in 2024. Some US pulmonologists still prefer clinic-based dosing for all biologics, citing liability and technique concerns. These policies add indirect costs and dissuade site-of-care migration, though pressure from payers seeking lower infusion fees is prompting gradual liberalization.
*Our forecasts treat driver/restraint impacts as directional, not additive. The impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Mechanism of Action: TSLP Pathway Disrupts Biomarker-Dependent Paradigm
Anti-IL-4Rα agents retained the largest asthma biologics market share at 44.80% in 2025, yet Anti-TSLP agents are positioned to expand at a 14.49% CAGR through 2031, the fastest among all classes. Dupilumab’s broad efficacy across asthma, atopic dermatitis, and CRSwNP underpinned USD 11.6 billion in global sales in 2024, with asthma accounting for roughly 35% of revenue, consolidating the drug’s position as a platform therapy. By contrast, tezepelumab’s upstream TSLP blockade treats the 25-30% of severe patients who present with low eosinophils, normal FeNO, and minimal IgE, a cohort historically limited to systemic corticosteroids. In NAVIGATOR, tezepelumab cut exacerbations 41% in biomarker-low patients, a feat still unmatched by rival mechanisms. AstraZeneca’s October 2025 CRSwNP label expansion now allows ENT specialists to initiate the drug, widening its prescriber base and expanding the asthma biologics market addressable by a single molecule.

By Phenotype: Eosinophilic Segment Retains Size Advantage While Biomarker-Low Gains Momentum
Eosinophilic asthma maintained 38.65% of 2025 revenue and remains pivotal to payer coverage algorithms that favor documented eosinophilia. Eosinophilic asthma is also expected to advance at 13.65% CAGR through 2031. This phenotype offers multiple therapeutic options—IL-5, IL-5Rα, and IL-4Rα inhibition—resulting in competitive contracting that keeps prices in check for payers while preserving physician choice.
Biomarker-low severe asthma, historically underserved, shows rapid patient conversion following the approval of tezepelumab, adding thousands of new candidates to the asthma biologics market. Absence of a clear predictive test requires trial-and-error strategies, and insurers frequently mandate six-month reassessment endpoints. Nevertheless, pulmonologists welcome the option because systemic steroids and bronchial thermoplasty offer modest benefit for this group.
By Route of Administration: Subcutaneous Delivery Dominates on Convenience and Cost
Subcutaneous formulations accounted for 60.45% of 2025 revenue and are projected to grow at a 15.11% CAGR through 2031, reshaping the trajectory of the asthma biologics market toward at-home care. Five leading products, dupilumab, mepolizumab, benralizumab, omalizumab, and tezepelumab, now offer prefilled pens or autoinjectors that require only an initial supervised dose before self-administration, cutting per-dose delivery costs by 30-40% compared with hospital infusion. A 2025 German time-motion study showed that patients saved 3.2 hours per subcutaneous dose compared with IV infusion, including travel and observation time.
Intravenous delivery, dominated by reslizumab, faces sustained headwinds as commercial payers escalate site-of-care steering and as new long-acting subcutaneous options, including depemokimab’s twice-yearly regimen, push convenience boundaries even further.
By End-User: Hospitals Anchor Initiation, Home Settings Accelerate Follow-Up Volumes
Hospitals generated 49.03% of 2025 revenue because initiation protocols still require supervised dosing. However, payer site-of-care programs have begun redirecting maintenance injections to specialty clinics and the home once safety is proven. Specialty Clinics is expected to grow the fastest, with a 14.23% CAGR through 2031. Specialty clinics appeal through flexible scheduling and reduced facility fees, capturing a growing slice of the asthma biologics market.
Home dosing is the fastest-growing channel as specialty pharmacies coordinate delivery, telehealth teaching, and adherence monitoring. Regions that relax self-administration rules will unlock further growth; conversely, Japan’s continued clinic mandate illustrates how regulation can slow channel migration despite patient interest.
Geography Analysis
North America retained 45.12% of the 2025 value, buoyed by payer coverage that reimburses biologics for biomarker-qualified patients despite high list prices. The region benefits from mature pulmonology networks, robust biomarker testing, and quick FDA approvals, making it the reference launch market for new agents like depemokimab. Canada widened access when provinces added mepolizumab and benralizumab to public formularies in 2025, narrowing gaps with US availability.
Europe contributed a significant share of global revenue. Germany’s flexible reimbursement fosters early adoption, while the UK’s NICE imposes tighter cost-effectiveness filters that slow uptake until negotiated discounts align with QALY thresholds. Southern European countries show variable hospital budgets but follow EMA guidance once national price negotiations conclude. Biosimilar entry is expected to temper spending growth without curtailing access, as seen with omalizumab.
Asia-Pacific posts the fastest growth at 9.48% CAGR, driven by China’s price-discounted duplication list entry and Japan’s premium innovation pricing for tezepelumab and depemokimab. South Korea and Australia exhibit mid-single-digit growth on the back of expanding private insurance coverage. India remains nascent but could pivot upward when local biosimilars arrive post-2027, lowering costs and enabling broader reach.
Competitive Landscape
A significant share of the asthma biologics market is held by four major players, Sanofi, GlaxoSmithKline, AstraZeneca, and Novartis AG, who together controlled the majority share of 2025 sales. Sanofi’s multi-indication strategy for Dupixent underpins its position; the drug now spans asthma, atopic dermatitis, CRSwNP, eosinophilic esophagitis, and prurigo nodularis, leveraging shared sales infrastructure across specialties. GSK differentiates through dosing interval leadership: Nucala’s autoinjector and the new twice-yearly depemokimab target adherence gaps. AstraZeneca’s tezepelumab addresses biomarker-low patients and ENT indications, expanding the prescriber universe.
Biosimilar dynamics are reshaping anti-IgE competition. Celltrion’s interchangeable Omlyclo, priced below Xolair, enables automatic substitution, raising the bar for originator retention tactics. Novartis counters with enhanced patient support and multi-year volume agreements with integrated delivery networks. Similar biosimilar challenges loom for IL-5 inhibitors between 2026 and 2027, prompting incumbents to emphasize device convenience and real-world adherence outcomes.
Pipeline diversity is growing. Anti-IL-33 antibodies in Phase 3 trials and oral TSLP antagonists in Phase 2 could offer efficacy similar to injectables with greater convenience or lower costs. If successful, these assets might shift the asthma biologics market toward oral maintenance, although regulators will scrutinize long-term safety given systemic exposure.
Asthma Biologics Industry Leaders
Sanofi
GlaxoSmithKline
Celltrion Inc.
AstraZeneca PLC
Novartis AG
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2026: Sanofi announced positive top-line results from Phase 2 trials of lunsekimig (SAR443765), targeting chronic respiratory diseases like asthma and chronic rhinosinusitis with nasal polyps (CRSwNP) by simultaneously blocking TSLP and IL-13.
- December 2025: EMA’s CHMP issued a positive opinion for depemokimab for severe asthma with type 2 inflammation and chronic rhinosinusitis with nasal polyps, starting national reimbursement processes in Europe.
- October 2025: The FDA approved tezepelumab for chronic rhinosinusitis with nasal polyps, expanding the drug’s label beyond asthma.
Global Asthma Biologics Market Report Scope
As per the scope of the report, asthma biologics are a class of precision medications used to treat moderate-to-severe asthma that remains uncontrolled despite the use of traditional high-dose inhaled steroids and long-acting bronchodilators.
The asthma biologics market is segmented by mechanism of action, phenotype, route of administration, end users, and geography. By mechanism of action, the market is segmented into Anti-IL-4Rα, Anti-TSLP, Anti-IL-5, Anti-IL-5Rα, and Anti-IgE. By phenotype, the market is segmented into eosinophilic asthma, allergic asthma, OCS-dependent severe asthma, and non-eosinophilic asthma. By route of administration, the market is segmented into subcutaneous and intravenous.
By end users, hospitals, specialty clinics, and home/at-home settings. Geographically, the market is segmented across North America, Europe, the Asia-Pacific region, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Anti-IL-4Rα |
| Anti-TSLP |
| Anti-IL-5 |
| Anti-IL-5Rα |
| Anti-IgE |
| Eosinophilic asthma |
| Allergic asthama |
| OCS-dependent severe asthma |
| Non-eosinophilic asthama |
| Subcutaneous |
| Intravenous |
| Hospitals |
| Specialty Clinics |
| Home/At-home Settings |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of APAC | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of MEA | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Mechanism of Action (MoA) | Anti-IL-4Rα | |
| Anti-TSLP | ||
| Anti-IL-5 | ||
| Anti-IL-5Rα | ||
| Anti-IgE | ||
| By Phenotype / Biomarker Segment | Eosinophilic asthma | |
| Allergic asthama | ||
| OCS-dependent severe asthma | ||
| Non-eosinophilic asthama | ||
| By Route of Administration | Subcutaneous | |
| Intravenous | ||
| By End-user | Hospitals | |
| Specialty Clinics | ||
| Home/At-home Settings | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of APAC | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of MEA | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large is the asthma biologics market in 2026?
It stands at USD 10.05 billion and is set to expand rapidly through 2031.
Which mechanism class holds the greatest revenue share?
Anti-IL-4Rα agents, led by dupilumab, controlled 44.80% of 2025 revenue.
What growth rate is forecast for Asia-Pacific?
Asia-Pacific is projected to post a 14.12% CAGR between 2026 and 2031.
How will biosimilars affect pricing?
The first interchangeable omalizumab biosimilar launched at discount, signaling broader price pressure as more follow-on products arrive.
Which product offers the longest dosing interval?
GSK’s depemokimab requires only two injections per year following its 2025 FDA approval.
Page last updated on: