Antimicrobial Ingredients Market Size and Share
Antimicrobial Ingredients Market Analysis by Mordor Intelligence
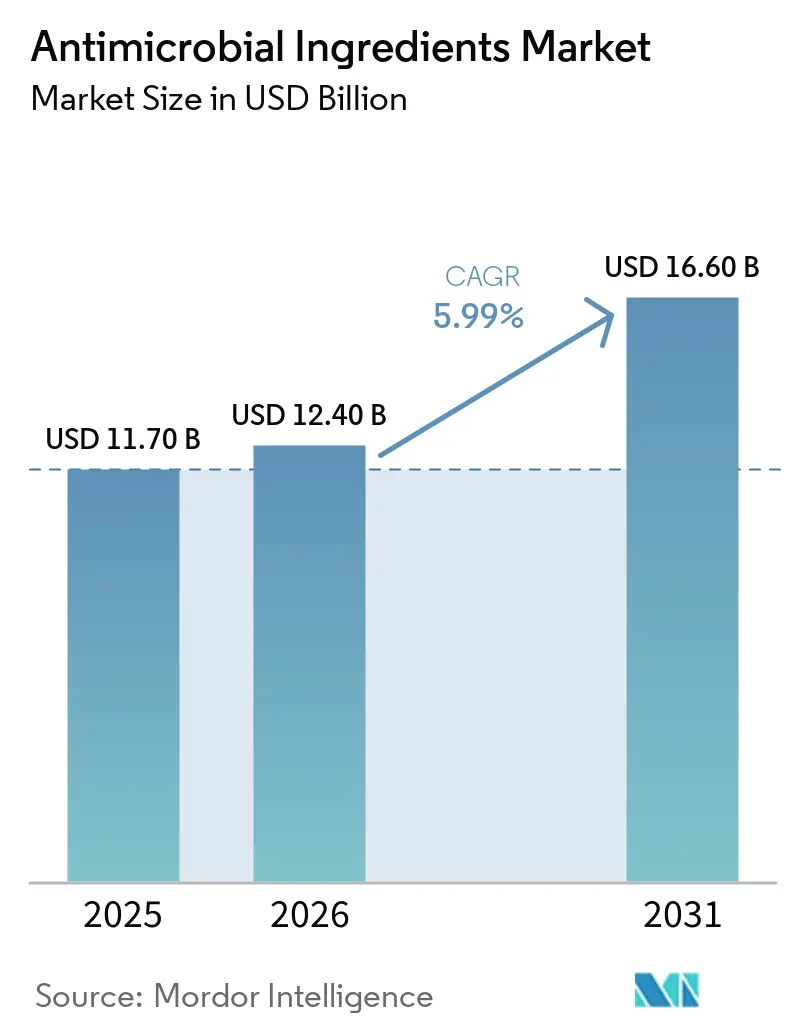
The Antimicrobial Ingredients Market size is expected to increase from USD 11.70 billion in 2025 to USD 12.40 billion in 2026 and reach USD 16.60 billion by 2031, growing at a CAGR of 5.99% over 2026-2031.
Adoption is accelerating because infection-control budgets are shifting from episodic cleaning toward built-in prevention, especially in operating rooms and intensive-care units where every healthcare-associated infection can trigger reimbursement penalties[1]Centers for Medicare & Medicaid Services, “Hospital-Acquired Condition Reduction Program,” cms.gov. Hospitals are embedding silver, copper, and quaternary ammonium compounds directly into devices and high-touch surfaces to cut labor hours tied to daily disinfection rounds. Procurement teams also favor antimicrobial coatings that sustain efficacy for multiple weeks, reducing chemical use and nurse workload. Suppliers that bundle dossier support under the U.S. EPA, EU BPR, and FDA 510(k) pathways are consolidating share because compliance costs are rising faster than raw-material prices. Collectively, these trends position the Antimicrobial ingredients market for steady expansion through the decade while creating white space for peptide and cold-plasma technologies.
Key Report Takeaways
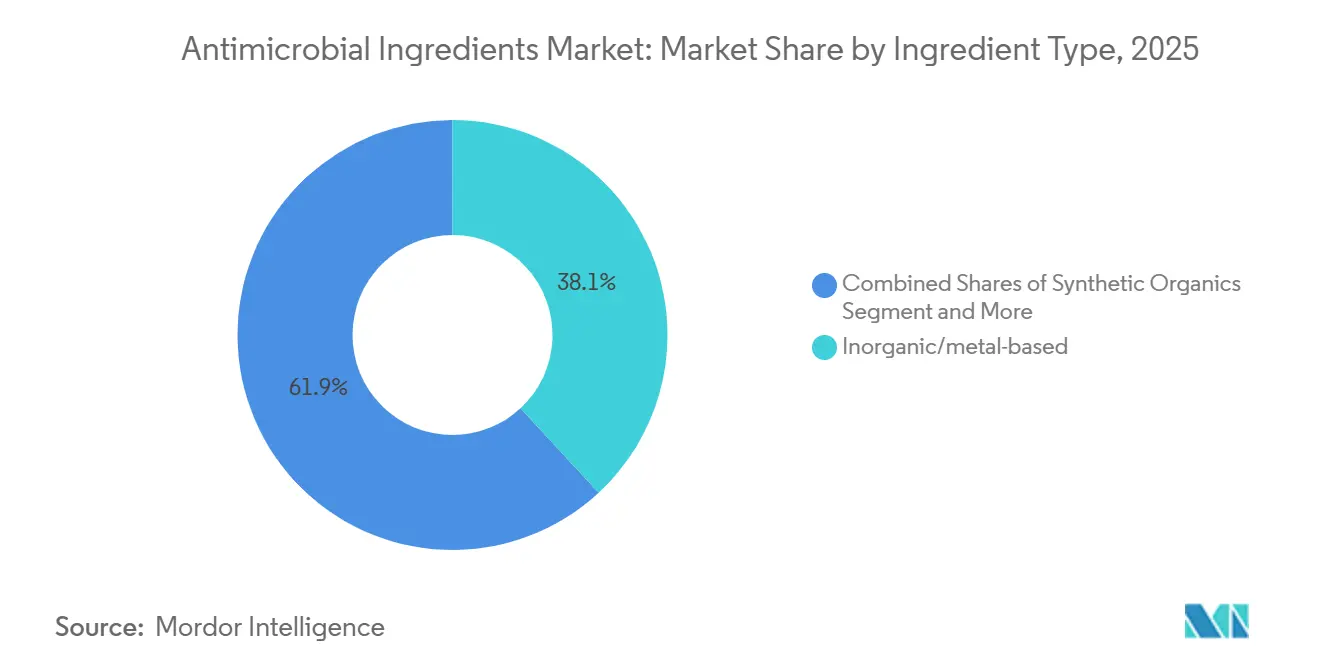
- By ingredient type, inorganic and metal-based chemistries held 38.10% of the antimicrobial ingredients market share in 2025. However, natural and bio-based alternatives are projected to expand at a 7.89% CAGR through 2031.
- By application, medical devices and implants captured 27.81% share of the antimicrobial ingredients market size in 2025. Whereas hospital hygiene and disinfectants are advancing at an 8.15% CAGR through 2031.
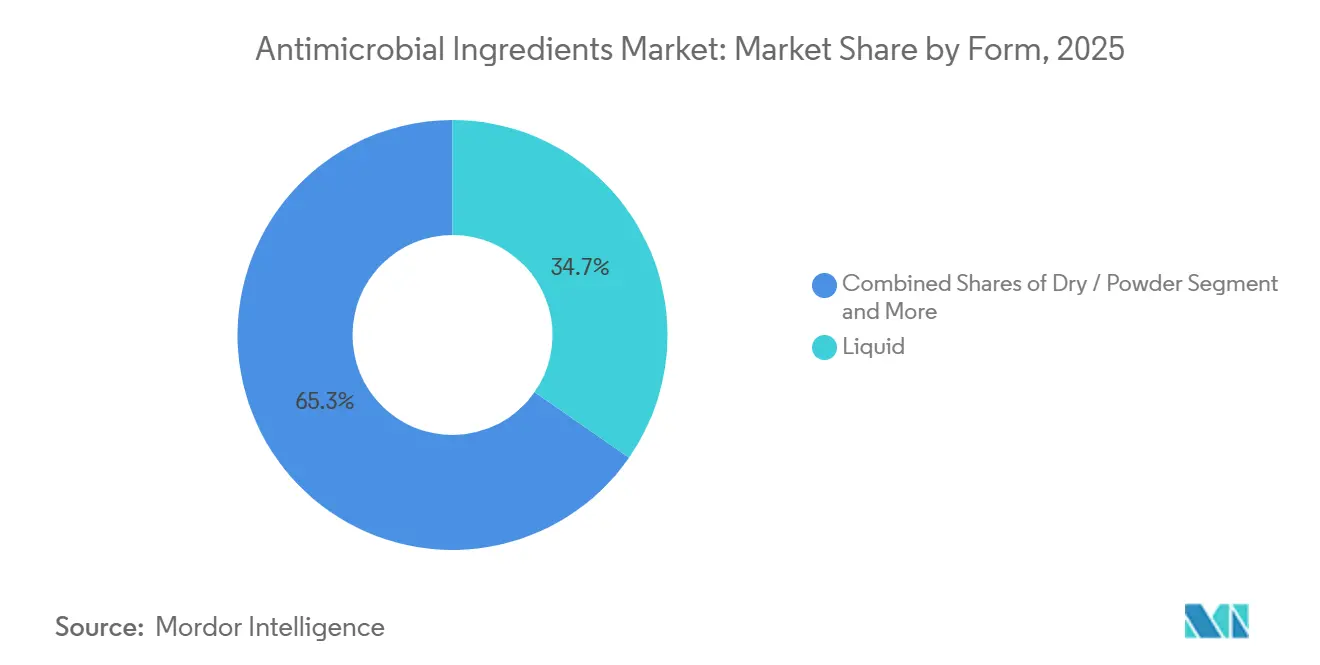
- By form, liquid formulations led with 34.67% share of the antimicrobial ingredients market size in 2025. Yet encapsulated and controlled-release systems are forecast to expand at a 7.98% CAGR through 2031.
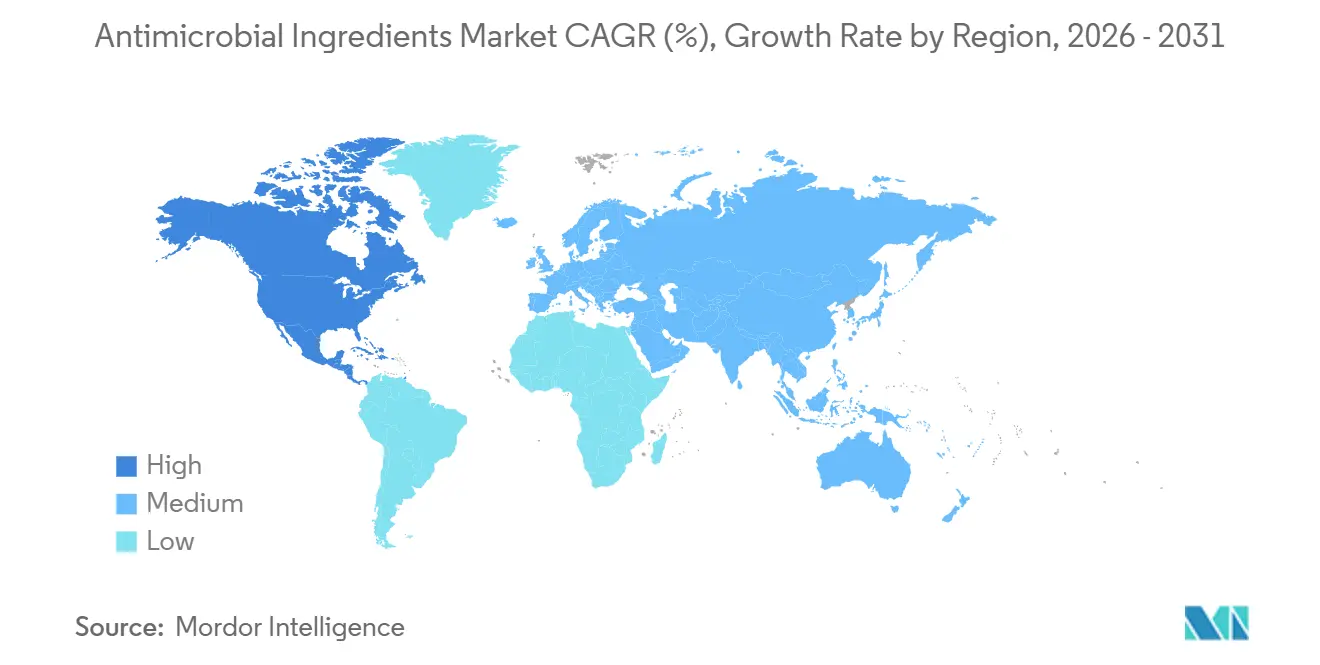
- By geography, the Asia Pacific commanded 35.18% of the antimicrobial ingredients market share in 2025. However, North America is the fastest-growing region at a 7.67% CAGR over 2026-2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Antimicrobial Ingredients Market Trends and Insights
Driver Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Healthcare-Associated Infections | +1.2% | Global, acute in North America and the EU | Medium term (2-4 years) |
| Stringent Infection Control Regulations | +1.0% | North America and the EU, expanding to APAC | Long term (≥ 4 years) |
| Growth in Medical Device and Implant Usage | +0.9% | Global, led by APAC manufacturing hubs | Medium term (2-4 years) |
| Expansion of Hospital Infrastructure | +0.8% | APAC core, spill-over to MEA and South America | Long term (≥ 4 years) |
| Increased Focus on Surface Hygiene Post-2020 | +1.1% | Global, heightened in North America and EU | Short term (≤ 2 years) |
| Advancements in Antimicrobial Technologies | +0.7% | North America and EU R&D centers, APAC scale-up | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Healthcare-Associated Infections
Healthcare-associated infections impose USD 28 billion to USD 45 billion in annual costs on U.S. hospitals and still affect roughly 1 in 31 patients on any given day. Although bloodstream and urinary-tract infection rates declined modestly between 2023 and 2024, absolute incidence remains high, steering administrators toward catheters, ventilator tubing, and bed linens impregnated with silver and quaternary ammonium compounds. Medicare payment penalties under the Hospital-Acquired Condition Reduction Program further motivate adoption. Peer-reviewed studies show copper-silver textiles retain 99.9% efficacy against ESKAPE pathogens after 50 wash cycles, reinforcing return-on-investment arguments.
Stringent Infection Control Regulations
The FDA’s 2024 guidance requires any device marketed with an infection-prevention claim to demonstrate clinical efficacy under ISO 22196 or ASTM E2180. In the EU, the Biocidal Products Regulation demands nanomaterial-specific dossiers that can take five years to clear[2]European Chemicals Agency, “Biocidal Products Regulation,” echa.europa.eu. These hurdles raise the cost of market entry but also reward suppliers that offer turnkey regulatory files across multiple jurisdictions. To meet both safety and efficacy targets, manufacturers turn to controlled-release capsules that deliver steady antimicrobial levels while limiting cytotoxic peaks. EPA registration of copper-alloy touch surfaces in 2023 exemplifies how material advances and dossier depth intersect.
Growth in Medical Device and Implant Usage
Orthopedic plates, cardiovascular stents, and urinary catheters collectively represent a USD 15 billion sub-segment within the Antimicrobial ingredients market. Silver-based coatings dominate, but enzyme and peptide systems such as lysozyme and ε-polylysine are emerging because they kill bacteria without harming mammalian cells. Medline’s SilvaSorb dressing maintains antimicrobial action for seven days, cutting nursing time and consumable waste. Research published in 2025 showed chitosan-polylysine coatings reduced Staphylococcus aureus biofilm by 98.5% while supporting osteoblast growth, signaling regulatory readiness for first peptide-coated implants later this decade.
Expansion of Hospital Infrastructure
China earmarked RMB 1.2 trillion (USD 170 billion) for hospital construction through 2025, while India plans 150,000 Health and Wellness Centers by 2027. New builds adopt antimicrobial flooring, wall cladding, and HVAC filters from day one, opening volume opportunities for epoxy and PVC formulations. Stonhard’s epoxy floors meet FDA food-contact rules and resist fungal growth, appealing to facilities that combine surgical, pharmacy, and dietary operations. Middle-East hospital beds are set to grow 12% by 2030 under Vision 2030 reforms.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Regulatory and Safety Concerns | -0.6% | Global, acute in EU and North America | Long term (≥ 4 years) |
| Risk of Antimicrobial Resistance | -0.5% | Global, heightened scrutiny in EU | Medium term (2-4 years) |
| High Cost of Advanced Materials | -0.4% | APAC, MEA, South America price-sensitive users | Medium term (2-4 years) |
| Limited Long-Term Durability in High-Touch | -0.3% | Global, especially surgical wards | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Regulatory and Safety Concerns
EU dossiers for nanomaterials can cost EUR 1 million per active substance and extend launch cycles to five years. FDA guidance denies accelerated reviews for combination products that claim antimicrobial benefits, forcing full 510(k) or PMA submissions. Hospitals, therefore, hedge with legacy alcohols and chlorine because decades of toxicology data exist. Cytotoxic thresholds also cap silver loading in wound dressings at 10 ppm, limiting efficacy windows and prompting some buyers to stick with cheaper single-use gauze.
Risk of Antimicrobial Resistance
WHO warns that indiscriminate biocide use can foster cross-resistance in key pathogens such as carbapenem-resistant Acinetobacter. Laboratory work in 2025 showed quaternary ammonium exposure up-regulated efflux pumps that also expel fluoroquinolones. EU regulators now mandate six-month resistance surveillance for antimicrobial-coated devices, adding USD 300,000 in annual compliance costs [3]European Medicines Agency, “AMR Surveillance Requirements,” ema.europa.eu. These concerns push suppliers to multi-action metals and peptides that lower single-gene adaptation odds but still meet hospital kill-time targets.
Segment Analysis
By Ingredient Type: Metal Dominance Meets Bio-Based Momentum
In 2025, inorganic and metal chemistries captured 38.10% revenue as silver and copper delivered broad-spectrum kill without fostering quick resistance. Copper alloys achieve a 4-log reduction of MRSA within two hours, driving uptake on bed rails and call buttons. Synthetic organics such as quaternary ammonium compounds remain workhorses in sprays and wipes but face eco-toxicity scrutiny. Natural and bio-based options are growing at 7.89% CAGR, supported by sustainability mandates and patient preference for low-toxicity rooms. Essential oils appeal in low-risk zones, while chitosan serves wound dressings needing hemostasis and antimicrobial action. High peptide prices confine their use to premium implants, yet regulatory fast-tracks for GRAS ingredients like nisin could offset cost barriers by 2028.
Surging R&D dollars are steering toward enzymes that disrupt biofilms rather than kill planktonic cells, reducing resistance pressure. DSM-Firmenich is piloting Delvo Nis peptide coatings for catheters, betting that FDA familiarity with the molecule in food applications will streamline device approval.

By Application: Devices Anchor Revenue, Hygiene Leads Growth
Medical devices and implants generated 27.81% revenue in 2025, reflecting steady demand for coated catheters, dressings, and fracture plates. Hospitals favor silver hydrocolloids that stay active for a week, lowering change frequency and nursing burden. Central line kits with antimicrobial lumens cost more upfront but avert bloodstream infections that can add USD 45,000 per case, making payback clear in value-based care models.
Hospital hygiene and disinfectants are slated for the fastest expansion at 8.15% CAGR. Arxada’s NUGEN HLD-CD solves C. difficile turnover bottlenecks, freeing ORs faster on busy surgical days. Textile firms now weave copper or zinc into privacy curtains to trim laundering cycles, and Microban’s 2026 deal with Yunus USA brings similar technology to home bedrooms, indicating spillover beyond healthcare. Cleanroom flooring, GMP wallboards, and contamination-control mats round out the “other” bucket, each carving niche revenue that still feeds the broader Antimicrobial ingredients market.
By Form: Liquids Hold Volume, Encapsulated Systems Accelerate
Liquids retained 34.67% share in 2025 because they integrate seamlessly into spray and mop routines. Brenntag’s 2024 pact with Arxada widened Nordic access to quaternary ammonium concentrates, underlining logistical advantage. Powders serve plastic extruders that prefer solvent-free masterbatch, while gels stake out wound and vertical-surface niches.
Encapsulated formats are advancing at 7.98% CAGR as administrators calculate lifetime cost, not sticker price. Layer-by-layer smart coatings leak peptides only when bacterial pH rises, extending efficacy to 14 days without resistance pressure. Cerium oxide and silver micro-capsules inside polyurethane paints promise 10-year durability for air-handling units. As hospitals decontaminate entire HVAC runs during retrofits, encapsulated paints may capture additional Antimicrobial ingredients market demand that liquids cannot reach.

Geography Analysis
Asia Pacific accounted for 35.18% revenue in 2025, underpinned by China’s device factories and India’s hospital build-out. Beijing’s 14th Five-Year Plan channeled USD 170 billion into provincial facilities, each specifying antimicrobial epoxy floors and copper-alloy handles. Japan obliges antimicrobial coatings on public hospital fixtures, making it a high-value niche despite slower population growth. South Korea enforces JIS Z 2801 testing, which smaller suppliers often lack, giving incumbents an edge.
North America is the quickest-growing geography at 7.67% CAGR because CMS links reimbursement to infection scores. U.S. hospitals willingly pay List N premiums for disinfectants that guarantee SARS-CoV-2 efficacy. Device makers also rely on antimicrobial masterbatch to secure 510(k) clearance for coated catheters. Canada’s alignment under USMCA reduces dossier duplication, helping mid-sized formulators expand. Though volumes lag Asia, higher average selling prices deliver outsized profit, reinforcing regional importance to the antimicrobial ingredients market.
Europe, the Middle East, and South America fill the remainder. EU BPR timelines stretch launch cycles, but Germany, France, and the U.K. remain innovation hubs demanding data-rich dossiers. Middle-East bed growth under Vision 2030 stipulates ISO 14644 clean zones, a tailwind for antimicrobial HVAC coatings. South America lags because of budget constraints, yet Mercosur's work on unified biocide rules could shorten approval windows and unlock demand in Brazil and Argentina before 2031.
Competitive Landscape
Top suppliers, LANXESS, BASF, Arxada, Dow, and Clariant, control a notable share of global revenue, underscoring moderate concentration. LANXESS bought IFF’s Microbial Control arm for USD 1.3 billion in 2022, adding USD 450 million in quaternary ammonium and isothiazolinone sales. Arxada posted USD 2 billion in revenue in 2024, doubling patent filings on sustainable formulations. The big five compete on dossier depth, vertical integration, and full-range portfolios that span metals, QACs, acids, and peptides.
Specialists fill gaps. Microban’s AkoTech embeds antimicrobial plus hydrophobic traits for touchscreen OEMs, differentiating on multi-functionality. BioCote and Sanitized focus on masterbatch additives for plastics, signing OEM agreements that bake infection control into product design. Kismet Technologies and Dycem exploit nanoparticle and cold-plasma niches that giant incumbents still evaluate but have not scaled.
Future rivalry will tilt toward engineered systems, not commodity activities. Patent spikes in cerium oxide, ε-polylysine, and smart carriers suggest emphasis on balancing efficacy, durability, safety, and environmental fate. Suppliers that can provide real-world surveillance data and life-cycle analyses will outpace commodity competitors, especially as reimbursement models penalize infections rather than reward volume purchases. The Antimicrobial ingredients market therefore remains ripe for both consolidation and disruptive entry.
Antimicrobial Ingredients Industry Leaders
LANXESS AG
BASF SE
Arxada AG
Dow Inc.
Clariant AG
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: Revance introduced PanOxyl Antimicrobial Purifying Mist, a feather-light spray that helps clear, purify, and calm the skin while keeping its natural barrier intact.
- April 2025: Arxada launched NUGEN HLD-CD disinfectant, achieving five-minute C. difficile kill times and securing EU MDR certification.
- March 2025: Microban International unveiled AkoTech coating that combines antimicrobial, hydrophobic, and anti-fingerprint effects for floors and touchscreens.
Global Antimicrobial Ingredients Market Report Scope
As per the scope of the report, antimicrobial ingredients are specialized substances designed to destroy or inhibit the growth of a wide range of microorganisms, including bacteria, viruses, fungi, and parasites. These agents are broadly classified into several categories based on their target and application: antibiotics are used internally to treat bacterial infections; antiseptics are applied to living tissue to prevent infection; and disinfectants are utilized to sanitize non-living surfaces.
The antimicrobial ingredients market is segmented by ingredient type, application, form, and geography. Based on ingredient type, the market is segmented into inorganic/metal-based, synthetic organics, organic acids & salts, natural/bio-based, and enzymatic & antimicrobial peptides. By applications, the market is segmented into medical devices & implants, healthcare surfaces & coatings, wound care products, healthcare textiles, hospital hygiene & disinfectants, pharmaceutical & biotechnology manufacturing, and others. By form, the market is segmented into dry / powder, liquid, gel/foam, masterbatch/carrier for polymers, and encapsulated / controlled-release systems.
Geographically, the market is segmented into North America, Europe, Asia-Pacific, the Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. For each segment, the market size and forecast are provided in terms of value (USD).
| Inorganic/metal-based |
| Synthetic organics |
| Organic acids & salts |
| Natural/bio-based |
| Enzymatic & antimicrobial peptides |
| Medical Devices & Implants |
| Healthcare Surfaces & Coatings |
| Wound Care Products |
| Healthcare Textiles |
| Hospital Hygiene & Disinfectants |
| Pharmaceutical & Biotechnology Manufacturing |
| Others |
| Dry / Powder |
| Liquid |
| Gel / Foam |
| Masterbatch / Carrier for polymers |
| Encapsulated / Controlled-release systems |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Ingredient Type | Inorganic/metal-based | |
| Synthetic organics | ||
| Organic acids & salts | ||
| Natural/bio-based | ||
| Enzymatic & antimicrobial peptides | ||
| By Application | Medical Devices & Implants | |
| Healthcare Surfaces & Coatings | ||
| Wound Care Products | ||
| Healthcare Textiles | ||
| Hospital Hygiene & Disinfectants | ||
| Pharmaceutical & Biotechnology Manufacturing | ||
| Others | ||
| By Form | Dry / Powder | |
| Liquid | ||
| Gel / Foam | ||
| Masterbatch / Carrier for polymers | ||
| Encapsulated / Controlled-release systems | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the 2026 value of the antimicrobial ingredients market?
The Antimicrobial ingredients market size stands at USD 12.4 billion in 2026, and Mordor Intelligence projects will reach USD 16.6 billion by 2031.
Which segment leads in the antimicrobial ingredients market share?
In 2025, inorganic and metal-based chemistries held 38.10% of the antimicrobial ingredients market share, led by silver and copper actives
Which application is growing fastest within the antimicrobial ingredients industry?
Hospital hygiene and disinfectants are forecast to rise at an 8.15% CAGR through 2031 as facilities prioritize rapid-kill, surface-compatible products.
Which region offers the highest growth potential through 2031?
North America is set to expand at a 7.67% CAGR through 2031, driven by reimbursement penalties tied to infection metrics and EPA List N procurement standards.
Page last updated on: