AI In Pharmaceutical R & D Market Size and Share
AI In Pharmaceutical R & D Market Analysis by Mordor Intelligence
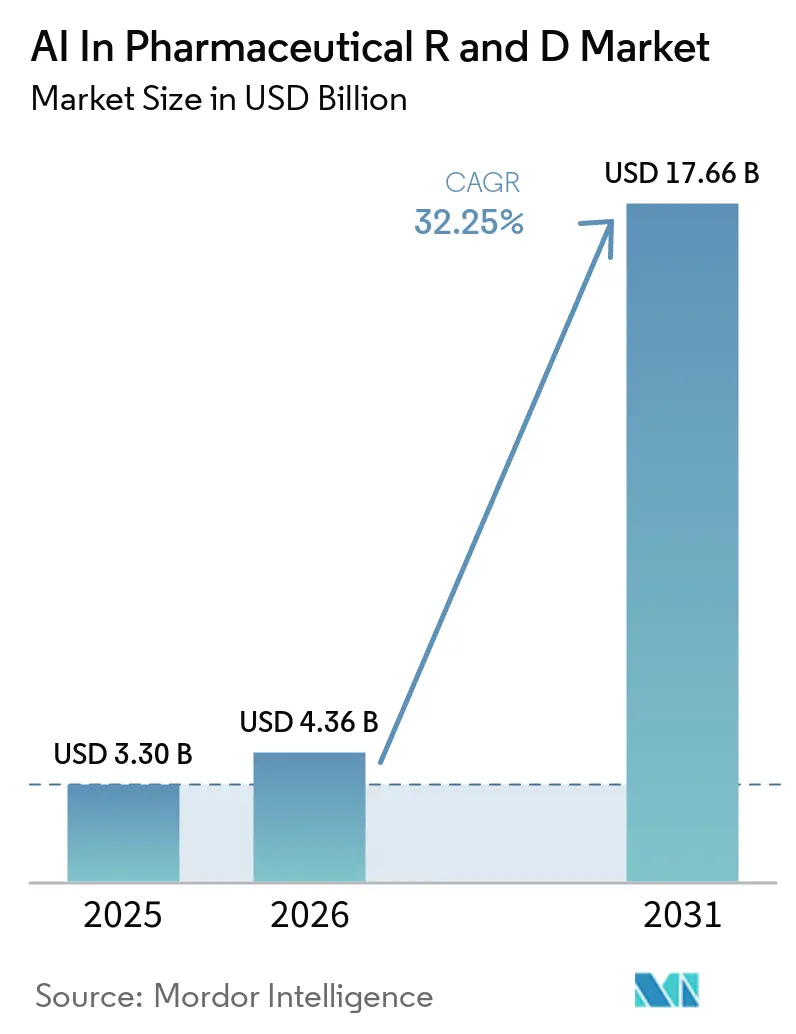
The AI In Pharmaceutical R & D Market size was valued at USD 3.30 billion in 2025 and is estimated to grow from USD 4.36 billion in 2026 to reach USD 17.66 billion by 2031, at a CAGR of 32.25% during the forecast period (2026-2031).
In January 2025, a JAMA study reported that the average cost of discovering and approving a drug has risen to a capitalized USD 1.31 billion. These increasing expenses are putting pressure on profit margins and driving sponsors to adopt automation in early-stage workflows. Regulatory authorities are reinforcing this shift. In January 2026, the FDA and EMA introduced ten joint guiding principles for AI practices, signaling their formal support for AI-driven drug discovery. Additionally, the FDA highlighted its growing focus on AI by reviewing 170 AI-related submissions in 2022, a significant increase from just 14 in 2020, reflecting the rapid adoption of this technology. Investor confidence aligns with this trend, as twelve life-sciences AI deals exceeded USD 200 million in 2024, indicating strong expectations for the rapid growth of AI in pharmaceutical R&D.
Key Report Takeaways
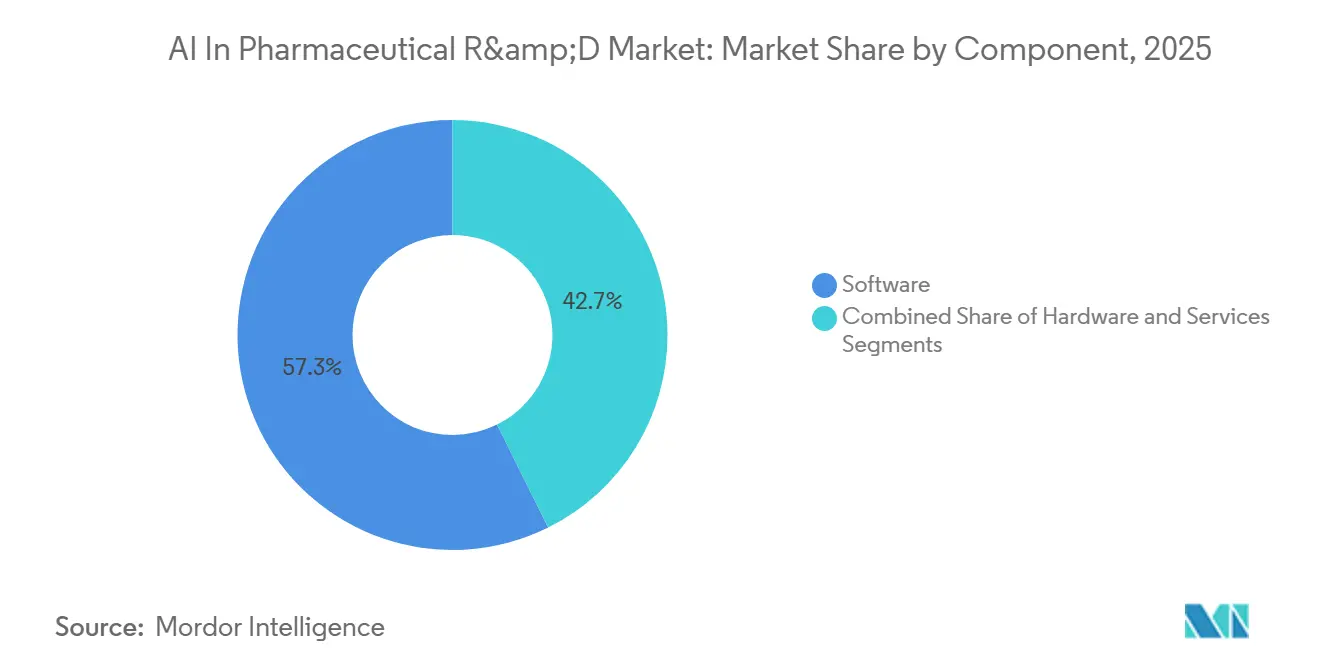
- By component, software led with 57.34% of the AI in pharmaceutical R&D market share in 2025. Services is forecast to expand at a 32.55% CAGR through 2031, the fastest pace among components.
- By technology, machine learning commanded 45.45% share of the AI in pharmaceutical R&D market size in 2025. Deep and generative learning is advancing at a 32.79% CAGR to 2031, the highest growth within technologies.
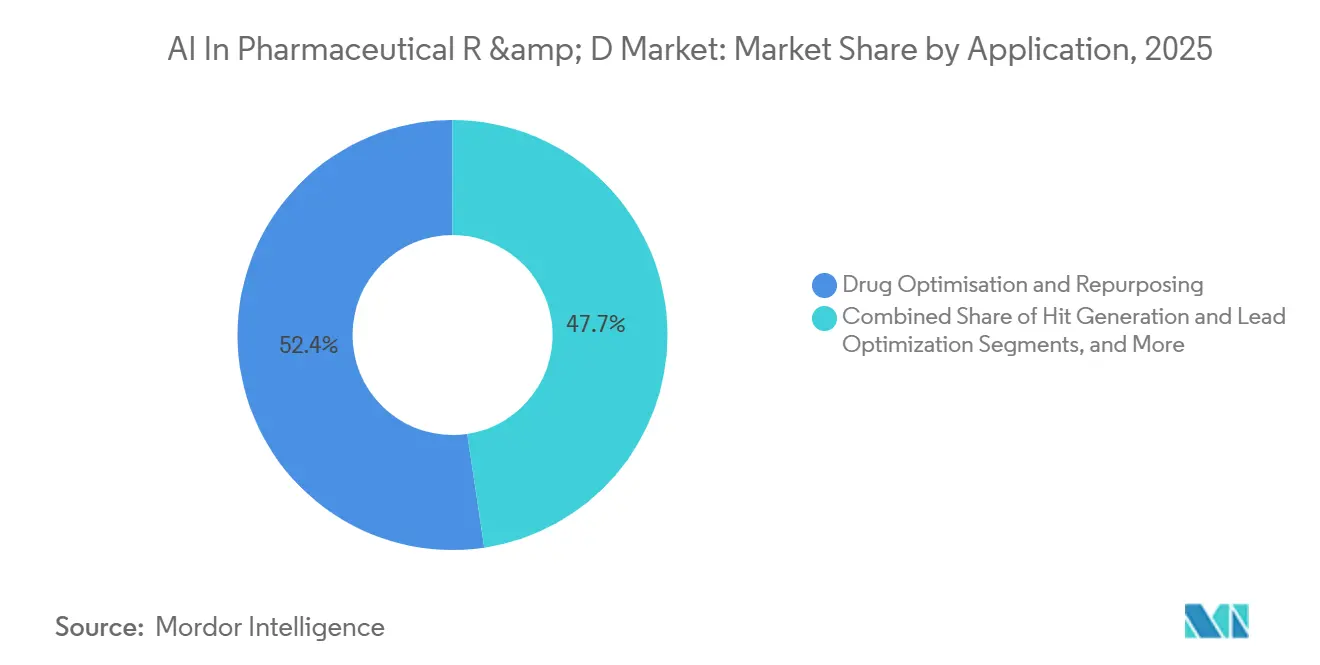
- By application, drug optimization and repurposing captured 52.35% revenue in 2025. Hit generation and lead optimization is projected to grow at a 32.98% CAGR through 2031.
- By end user, pharmaceutical and biotechnology companies held 59.45% spend in 2025. Contract research organizations are set to post a 33.15% CAGR during 2026-2031.
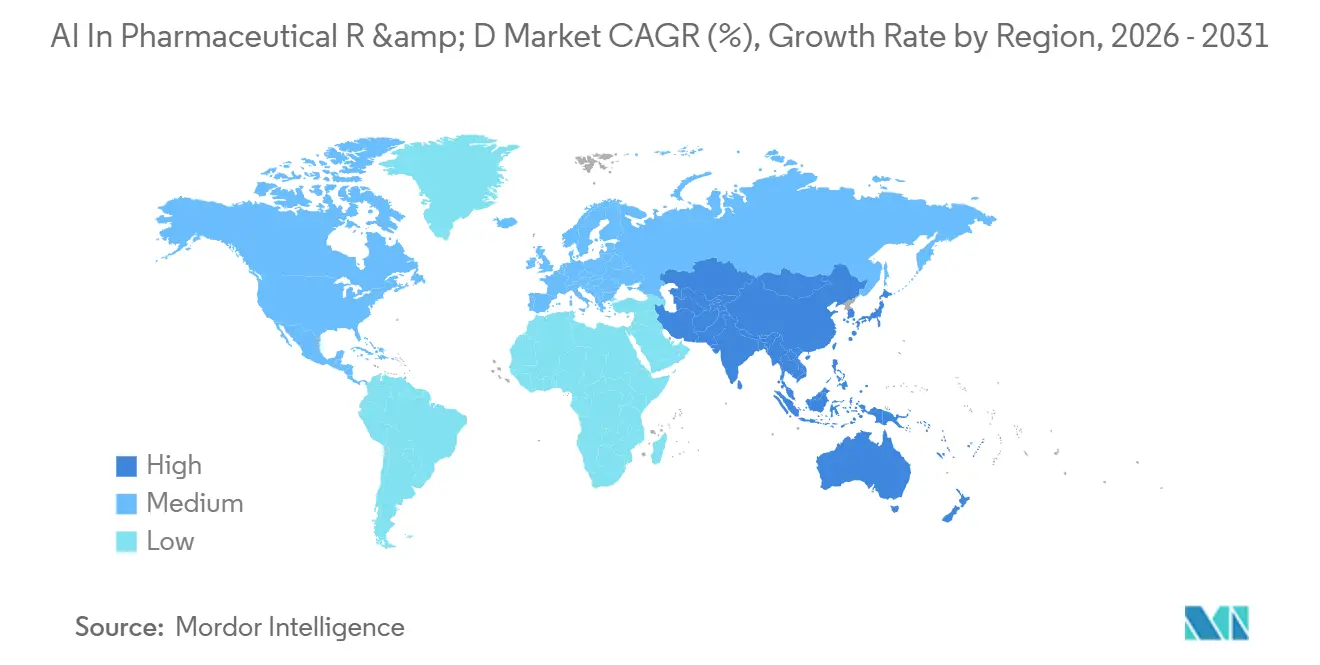
- By geography, North America accounted for 48.45% of the AI in pharmaceutical R&D market share in 2025. Asia-Pacific is on track for a 34.00% CAGR to 2031, the quickest regional expansion.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global AI In Pharmaceutical R & D Market Trends and Insights
Drivers Impact Analysis
| DRIVER | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| Escalating biopharma R&D costs | +6.8% | Global, concentrated in North America and Europe | Medium term (2-4 years) |
| Expanding biomedical data volumes | +5.2% | Global, with APAC data generation accelerating | Long term (≥ 4 years) |
| Pharma–AI partnerships & funding surge | +7.1% | Global, led by North America; APAC partnerships rising | Short term (≤ 2 years) |
| Regulatory openness to AI in R&D | +4.9% | North America, Europe; China regulatory alignment emerging | Medium term (2-4 years) |
| Emergence of foundation bio-models | +5.5% | Global, with North America and Europe leading deployment | Medium term (2-4 years) |
| Growth of federated‐learning data corridors | +2.8% | Europe and North America consortia; APAC nascent | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Soaring Costs in Biopharma R&D
JAMA estimates that the average cost of bringing a drug to market is USD 1.31 billion, accounting for capital costs and program attrition. Smaller biopharma firms face a 37.6% cost premium compared to the top 20 companies, highlighting the critical need to reduce drug development timelines.[1]JAMA Network, Mulcahy et al., “Use of Clinical Trial Characteristics to Estimate Costs,” jamanetwork.com AI platforms are addressing this challenge by improving phase-transition success rates and shortening patient trial durations. For instance, Insilico Medicine identified a preclinical candidate in just eight months, significantly faster than the traditional 2.5 to 4-year timeframe. These advancements are increasing confidence in AI's ability to reduce costs in pharmaceutical R&D, with deal structures now frequently incorporating milestones tied to accelerated timelines.[2]European Medicines Agency, “Reflection Paper on the Use of Artificial Intelligence,” ema.europa.eu
Surge in Biomedical Data Volumes
Foundation models depend on diverse data sets, including genomics, proteomics, imaging, and electronic health records (EHRs). Recursion, for example, has gained exclusive access to 20 petabytes of oncology data, increasing its total data holdings to approximately 50 petabytes. Similarly, IBM Research trains its models on over a billion small molecules and protein sequences, while Xaira’s X-Cell leverages 4.9 billion parameters derived from 25.6 million single-cell transcriptomes.[3]IBM Research, “Biomedical Foundation Models,” ibm.com The expansion and diversity of these data sets enhance model generalization, enabling predictions for previously untested targets or pathways. This trend is driving continuous investments in cloud computing, further expanding opportunities for specialized infrastructure providers in the AI-driven pharmaceutical R&D market.
Surge in Pharma–AI Collaborations & Funding
In 2024, twelve transactions exceeded USD 200 million, with Isomorphic Labs leading through agreements with Eli Lilly (USD 1.745 billion) and Novartis (USD 1.238 billion). Insilico Medicine secured an USD 888 million oncology deal with Servier in January 2026, which included USD 32 million in immediate research payments. Absci also expanded its partnership with Almirall to USD 650 million, focusing on two dermatology targets. The dominant risk-sharing model assigns discovery responsibilities to AI specialists, while pharmaceutical companies manage clinical and commercial risks. These financial inflows are fueling platform growth and reinforcing the momentum of the AI in pharmaceutical R&D market.
Regulatory Embrace of AI in R&D
In January 2026, the FDA and EMA introduced joint principles emphasizing data governance, model lifecycle management, and transparent communication of algorithmic limitations. These guidelines align with established practices in medical-device AI, providing drug developers with a clear compliance framework. The EMA's 2024 reflection paper outlines high-risk use cases and specifies testing requirements. Similarly, China's NMPA issued directives in March 2026 advocating for collaborative human-AI reviews and advanced risk-surveillance systems. Harmonizing regulatory principles across regions reduces duplication and facilitates global deployments, significantly expanding the addressable market for AI in pharmaceutical R&D.
Restraints Impact Analysis
| RESTRAINT | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| Data quality & standardization gaps | -3.2% | Global, acute in emerging markets and rare-disease datasets | Medium term (2-4 years) |
| Shortage of AI-skilled life-science talent | -2.9% | Global, most severe in North America and Europe | Short term (≤ 2 years) |
| IP ambiguity for AI-generated molecules | -2.1% | Global, regulatory clarity evolving in US and EU | Long term (≥ 4 years) |
| Rising GPU / cloud compute costs | -1.8% | Global, disproportionate impact on smaller biotechs | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Data Quality & Standardization Gaps
Inconsistent annotations, missing metadata, and non-standard identifiers weaken the generalizability of models. Regulatory bodies now require traceable documentation for every data-processing step and explicit acknowledgment of limitations. Collaborative initiatives, such as federated projects, are addressing these gaps by standardizing ontologies across partners. Additionally, national strategies, like China's digital-intelligent transformation plan for 2025, aim to establish unified data standards and regional data hubs to improve data quality. However, challenges such as outdated assays and underrepresented populations remain significant obstacles that could limit the growth of the AI in pharmaceutical R&D market until adequately addressed.
Shortage of AI-Skilled Life-Science Talent
The demand for professionals skilled in both deep learning and medicinal chemistry significantly outpaces the available supply. The lack of "dual-fluency" talent is identified as a critical constraint for the deployment of AI in the sector. While some companies have reduced their workforce, they have prioritized retaining core algorithm experts to maintain progress in their pipelines. National initiatives, such as China's transformation agenda for 2025, emphasize cross-disciplinary education to expand the talent pool. Partnerships between sponsors and technology providers are also increasing, with companies securing strategic investments to access specialized talent and hardware. Without rapid expansion of training programs, the AI in pharmaceutical R&D market may face short-term challenges that could slow its growth trajectory.
Segment Analysis
Software Takes the Lead, Services Gain Momentum
In 2025, software captured a dominant 57.34% share of the AI in pharmaceutical R&D market. Integrated platforms like RecursionOS manage processes ranging from target discovery to clinical-trial simulations, processing millions of phenomics images weekly and storing 36 petabytes of proprietary data. Exscientia’s platform integrates primary human-tissue assays into design loops, which management asserts enhances clinical relevance compared to traditional animal models. This strong platform loyalty drives recurring license revenue and supports the growth trajectory of the AI in pharmaceutical R&D market.
While services currently hold a smaller market share, they are projected to grow at a robust 32.55% CAGR through 2031. Contract Research Organizations (CROs) are differentiating their offerings by incorporating AI-driven patient-recruitment modules and adaptive-trial designs. As sponsors increasingly prefer variable costs over fixed ones, the adoption of outsourced AI capabilities is expected to expand, deepening their penetration in the AI in pharmaceutical R&D market.

Machine Learning Leads the Way, Generative Models on the Rise
In 2025, machine learning accounted for 45.45% of technology revenue, driving key processes such as ADME-Tox prediction, biomarker discovery, and patient stratification. Supervised algorithms continue to be dependable, particularly when labeled data is abundant.
Generative learning is emerging as the fastest-growing segment, with a remarkable 32.79% CAGR projected through 2031. AlphaFold 3’s innovative diffusion method has significantly enhanced protein-ligand predictions, improving accuracy by 50%. Exscientia’s sixth AI-generated molecule entered clinical trials in 2023, marking a pivotal moment for the technology. With advancements like transformer and diffusion networks now crafting antibodies, RNA therapeutics, and PROTACs, generative frameworks are set to redefine the AI in pharmaceutical R&D market landscape.
Repurposing Takes the Lead, Hit-to-Lead Gains Traction
In 2025, drug optimization and repurposing accounted for a substantial 52.35% of market expenditures. For example, BenevolentAI identified PDE10 as a potential treatment for ulcerative colitis, achieving positive Phase Ia results in 2024. The combination of reduced biological risk and shorter regulatory pathways bolsters investment in this area, solidifying its role as a growth pillar for the AI in pharmaceutical R&D market.
However, hit generation and lead optimization are witnessing even faster growth, expanding at a notable 32.98% CAGR. Companies like Absci and Almirall have successfully produced functional antibodies against challenging targets, showcasing generative AI’s capability to address complex biological issues. As an increasing number of AI-designed molecules navigate the IND process, there is a growing inclination toward this stage, further energizing the AI in pharmaceutical R&D market.

Pharma Leads the Charge, CROs Capture More Market Share
In 2025, pharmaceutical and biotechnology firms accounted for a commanding 59.45% of market spending. Notable collaborations include Recursion’s partnerships with Roche-Genentech, Bayer, and Merck KGaA, which could collectively surpass USD 2 billion in milestones. Additionally, Sanofi’s substantial USD 5.2 billion agreement with Exscientia encompasses up to 15 programs, underscoring big pharma's growing enthusiasm for expansive AI platforms.
Contract research organizations (CROs) are poised for a robust 33.15% CAGR through 2031. Sponsors increasingly regard CROs as comprehensive solutions for AI-driven trial designs, real-world evidence analytics, and patient stratification. With an expanded array of services, CROs are solidifying their role as pivotal gatekeepers in the AI in pharmaceutical R&D market's value chain.
Geography Analysis
In 2025, North America accounted for 48.45% of the revenue, driven by biotech clusters spanning Boston to San Diego and early FDA guidance on AI credibility. In 2024, venture investors directed nearly USD 10 billion into AI drug-discovery deals, with five transactions exceeding USD 1 billion. Demonstrating the region's computational capabilities, Recursion’s BioHive-2 supercomputer features 504 NVIDIA H100 GPUs. These factors collectively establish North America as the benchmark market for AI-driven innovation in pharmaceutical R&D.
Europe, while following North America, demonstrates strong regulatory involvement. Federated learning initiatives, such as MELLODDY, emphasize compliance with GDPR requirements. The EMA’s reflection paper provides detailed guidance across the product life cycle. Additionally, public-private partnerships and Horizon Europe funding foster early-stage ventures, maintaining Europe’s relevance in the AI pharmaceutical R&D market.
Asia-Pacific is the fastest-growing region, with a projected CAGR of 34.00% through 2031. China’s digital-intelligent transformation plan aims for full implementation by 2030, including 100 pilot digital drug factories and 10 large-model innovation hubs. India offers cost-efficient data-science talent for Contract Research Organizations (CROs), while Japan utilizes precision-medicine grants to modernize its clinical infrastructure. Although Latin America and the Middle East & Africa (MEA) lag in computational resources and regulatory frameworks, pilot projects in Brazil and the UAE indicate gradual progress, expanding the global footprint of AI in pharmaceutical R&D.
Competitive Landscape
AI-native biotechs, traditional discovery platforms, and hyperscalers vie for dominance in overlapping segments, leading to a moderately fragmented market. However, this fragmentation is being swiftly addressed through mergers, acquisitions, and strategic partnerships. In a significant move, Recursion and Exscientia finalized a merger agreement in August 2024. This merger combines Recursion's expertise in high-throughput phenomics and computational biology, supported by its BioHive-2 supercomputer, with Exscientia's capabilities in automated small-molecule synthesis and precision oncology. The newly formed entity anticipates annual synergies exceeding USD 100 million and targets approximately 10 clinical readouts within the next 18 months.
There are untapped opportunities in areas like rare diseases and overlooked patient demographics, where federated learning can unify fragmented datasets. Additionally, veterinary biologics present potential, particularly in AI-driven half-life engineering and species-specific optimization, which remain underutilized. Regulatory AI applications also offer growth prospects, especially with China's NMPA guidance in March 2026, emphasizing automated dossier reviews and pharmacovigilance signal triage.
Emerging disruptors include companies integrating AI with synthetic biology. For instance, Absci's generative AI combined with scalable wet labs enables weekly screening of billions of cells and six-week design-to-validation cycles. Another example is the use of patient-derived organoids and spatial multi-omics to enhance translational fidelity, as demonstrated by a recent partnership. As technology advances, the focus is shifting toward comprehensive integration from target identification to clinical candidate nomination. Demonstrating regulatory-grade validation and clinical translatability is becoming critical to reduce risks in pharmaceutical collaborations.
AI In Pharmaceutical R & D Industry Leaders
Insilico Medicine IP Limited
Recursion Pharmaceuticals
NVIDIA
XtalPi
Exscientia plc
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: Insilico Medicine signed a USD 888 million oncology discovery pact with Servier, earning USD 32 million in near-term R&D payments.
- January 2026: Insilico Medicine and Hisun Pharma nominated a preclinical candidate only eight months after initiating collaboration, leveraging the Pharma.AI platform.
- January 2026: The FDA and EMA issued ten guiding principles for good AI practice in drug development.
- August 2025: Almirall and Absci expanded their dermatology collaboration, lifting potential milestones to USD 650 million.
Global AI In Pharmaceutical R & D Market Report Scope
As per the scope of the report, artificial intelligence (AI) in pharmaceutical R&D refers to the application of machine learning (ML), deep learning (DL), and advanced data analytics to accelerate the discovery, development, and manufacturing of new medicines.
The AI in Pharmaceutical R&D market is segmented by component, technology, application, end-user, and geography. By component, the market includes software, services, and hardware. By technology, the market is segmented into machine learning, natural language processing, and deep/generative learning. By application, the market is categorized into target identification & validation, hit generation & lead optimization, preclinical/clinical testing, and drug optimization & repurposing. By end-user, the market is segmented into pharmaceutical & biotechnology companies, contract research organizations, academic & research institutes, and others. By geography, the market is analyzed across North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the market sizes and forecasts in terms of value (USD) for the above segments.
| Software |
| Services |
| Hardware |
| Machine Learning |
| Natural Language Processing |
| Deep / Generative Learning |
| Target Identification & Validation |
| Hit Generation & Lead Optimisation |
| Preclinical / Clinical Testing |
| Drug Optimisation & Repurposing |
| Pharmaceutical & Biotechnology Companies |
| Contract Research Organisations |
| Academic & Research Institutes |
| Others |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Component | Software | |
| Services | ||
| Hardware | ||
| By Technology | Machine Learning | |
| Natural Language Processing | ||
| Deep / Generative Learning | ||
| By Application | Target Identification & Validation | |
| Hit Generation & Lead Optimisation | ||
| Preclinical / Clinical Testing | ||
| Drug Optimisation & Repurposing | ||
| By End User | Pharmaceutical & Biotechnology Companies | |
| Contract Research Organisations | ||
| Academic & Research Institutes | ||
| Others | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large will spending on AI-driven discovery be by 2031?
The AI in pharmaceutical R&D market size is forecast to reach USD 17.66 billion by 2031.
What compound annual growth is expected for AI platforms in drug R&D?
Global revenue is projected to rise at a 32.25% CAGR over 2026-2031.
Which region is expanding the fastest?
Asia-Pacific leads with a 34.00% CAGR, propelled by China's digital-intelligent transformation mandate.
Which application currently attracts the most investment?
Drug optimization and repurposing captured 52.35% of 2025 spending in the AI in pharmaceutical R&D market.
Which technology category is growing quickest?
Deep and generative learning is expected to post a 32.79% CAGR through 2031, outpacing all other technologies.
How are regulators supporting AI adoption?
The FDA and EMA published joint guiding principles in 2026 that outline risk-based requirements for trustworthy AI in drug development.
Page last updated on: