Market Size of Pharmaceutical Contract Development and Manufacturing Organization (CDMO) Industry
| Study Period | 2019 - 2029 |
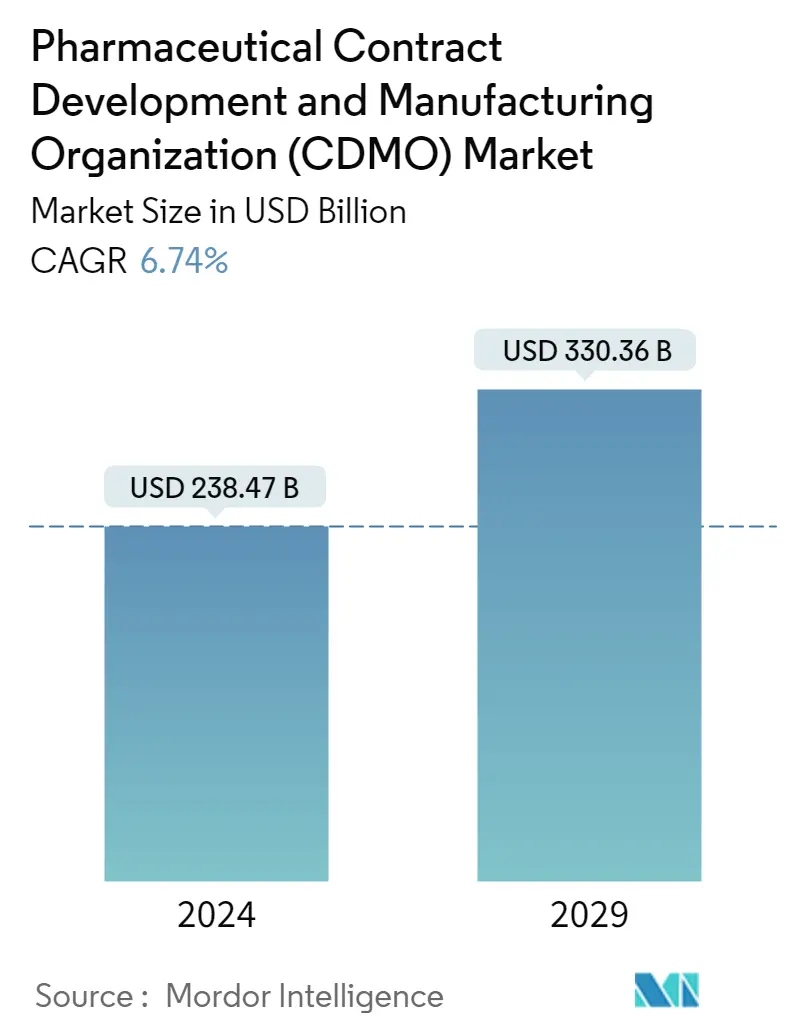
| Market Size (2024) | USD 238.47 Billion |
| Market Size (2029) | USD 330.36 Billion |
| CAGR (2024 - 2029) | 6.74 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Contract Development and Manufacturing Organization (CDMO) Market Analysis
The Pharmaceutical Contract Development and Manufacturing Organization Market size is estimated at USD 238.47 billion in 2024, and is expected to reach USD 330.36 billion by 2029, growing at a CAGR of 6.74% during the forecast period (2024-2029).
- Advanced manufacturing techniques and processes are projected to help the CMO market grow. CMOs are expected to improve the efficiency of their manufacturing processes, minimizing waste and lowering costs, owing to new operational strategies, such as continuous manufacturing. The growth of small and mid-sized pharmaceutical firms, which are in charge of an expanding portion of new drug approvals and frequently lack manufacturing capacity, is anticipated to be a driving force behind CMOs adopting new manufacturing technologies.
- A growing number of pharmaceutical companies recognized the potential profitability of working with a CMO for clinical and commercial-stage manufacturing due to the increasing demand for generic drugs and biologics, the capital-intensive nature of the industry, and the complex manufacturing requirements. Pharmaceutical innovator companies must fill their pipelines with fresh medications in light of the pharmaceutical industry's continued expansion, particularly from the onset of the COVID-19 pandemic. They lack the funding to research, create, and produce goods. Therefore, the need for CMOs is crucial.
- Growth of the generic drugs market, high uptake of small molecule drugs across a wide range of diseases, patent expiration of various drugs, advanced manufacturing techniques for active pharmaceutical ingredients (API), finished dosage formulations (FDF), growing requirements for advanced processes and production technologies to meet regulatory requirements, rising deals, such as mergers, acquisitions, and other investments, increased demand for generic injectables, growing pipeline of COVID-19 vaccines and biologics production, and increasing geriatric population are contributing drivers of CMO market.
- With the rise of personalized medicine, the one-size-fits-all model is rendered obsolete. Since the industry is trying to make clinical trials more accessible and patient-friendly, technology has become a key element in contract research organizations. To maintain a competitive edge and ensure they can provide customers with the full range of solutions, CROs are at the forefront of implementing the newest technologies and tools. Adopting these new technologies has been helping CROs to be more effective and increase the speed of research, thereby driving CRO market growth.
- The costs invested in R&D are continuously increasing, yet the valuable results from these processes are becoming rarer. Many pharmaceutical companies have invested in AP biologics and injectable capabilities projects. Also, pharmaceutical companies have been seeking vendors who provide contract manufacturing, contract packaging, and quality testing services. In addition, third-party logistic providers, like DHL, have been extending their service capabilities to include contract packaging services. Furthermore, as the turnaround in formulations and technology increases, CMOs will need to invest more significant amounts to achieve agility and accommodate quicker changes in the manufacturing setup. All these factors may restrict the growth of the studied market.
- The CMO/CDMO service industry is in a unique position to help drug developers overcome some of the difficulties brought on by the COVID-19 pandemic. The pharma and biopharma industries were impacted in many ways by this pandemic, including supply chain logistics, drug development, clinical trials, supplies, and manufacturing. The COVID-19 pandemic spurred digital transformation initiatives across the pharmaceutical industry, particularly in clinical development. The need for quicker clinical development increased as COVID-19, non-COVID-19 drugs, and treatments continued to develop during that period.
Pharmaceutical Contract Development and Manufacturing Organization (CDMO) Market Report Snapshots
- Pharmaceutical Contract Development and Manufacturing Organization (CDMO) Market Size
- Pharmaceutical Contract Development and Manufacturing Organization (CDMO) Market Share
- Pharmaceutical Contract Development and Manufacturing Organization (CDMO) Market Trends
- Pharmaceutical Contract Development and Manufacturing Organization (CDMO) Companies
