Market Size of Non-Invasive Prenatal Testing Industry
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
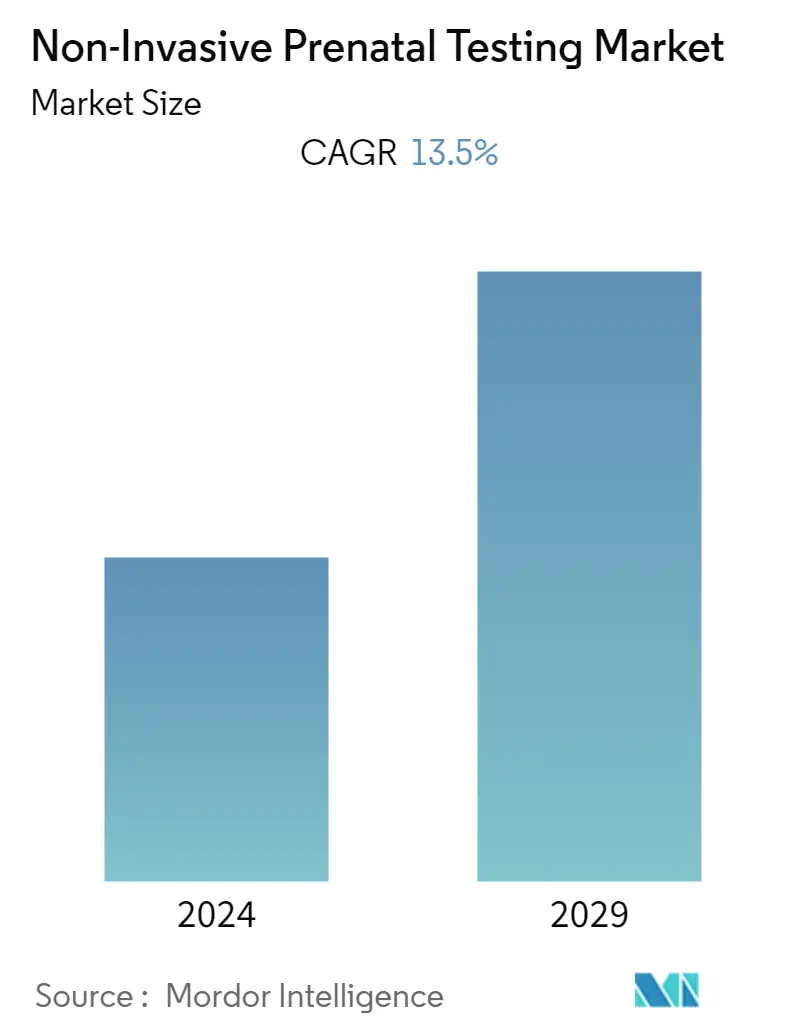
| CAGR | 13.50 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Non-invasive Prenatal Testing Market Analysis
The Non-Invasive Prenatal Testing Market (NIPT) size is expected to grow from USD 3.03 billion in 2023 to USD 5.71 billion by 2028, at a CAGR of 13.50% during the forecast period.
The COVID-19 pandemic impacted the non-invasive prenatal testing market due to the hold on non-invasive prenatal screening services to prevent the spread of the COVID-19 virus. The tremendous pressure on hospitals to provide services to COVID-19 patients was also one of the factors inhibiting hospitals and diagnostic centers from providing non-invasive prenatal screening (NIPS) services. According to the study published in the Journal of Clinical Medicine in March 2022, in Poland, the number of patients enrolled in the prenatal testing program fell compared to the previous year. In Poland, the number of integrated screening tests declined along with the number of newborns, while the percentage of triple tests increased. The number of invasive diagnostic tests also reduced significantly. Hence, the COVID-19 had a significant impact on the market growth.
Factors propelling the growth of the market are the increasing number of babies with chromosomal disorders owing to the increasing number of late pregnancies, increasing demand for early and non-invasive fetal diagnosis, and favorable reimbursement policies. The increasing number of stillbirth cases across the world is also expected to offer a sizeable growth opportunity for early disease diagnosis. For instance, in April 2022, a research study published in the National Library of Medicine stated that approximately 0.4% to 0.9% of newborns have chromosomal abnormalities, and about half have an abnormal phenotype. According to the same source, approximately 1 in 5,000 to 1 in 16,000 live births have the third most common autosomal trisomy. Hence, with such cases of stillbirths, the demand for testing increases, which, in turn, is expected to aid the overall market growth during the forecast period.
Additionally, the advantages of non-invasive prenatal tests over other prenatal tests, such as their non-invasive nature, better accuracy, and short duration of the procedure, are anticipated to hasten the growth of the NIPT market during the forecast period. Furthermore, awareness programs by public organizations are another factor propelling the market growth. For instance, in April 2022, the US Food and Drug Administration alerted the public about the risk of misleading results, improper use, and inappropriate interpretation of results (NIPT).
However, the lack of skilled professionals, stringent regulations, and ethical concerns are expected to hinder market growth.
