Market Trends of Hunter Syndrome Treatment Industry
This section covers the major market trends shaping the Hunter Syndrome Treatment Market according to our research experts:
The Enzyme Replacement Therapy (ERT) Segment is Expected to Witness Significant Growth in the Hunter Syndrome Treatment Market During the Forecast Period
The enzyme replacement therapy is expected to witness significant growth in the hunter syndrome treatment market over the forecast period due to the rising availability of enzyme replacement therapies, increasing awareness, and less stringent guidelines for rare diseases.
Enzyme replacement therapy (ERT) is a standard of care for several types of rare diseases and consists of replacing the deficient or absent enzyme with a functional recombinant version through intravenous administration. For instance, as per an article published in October 2021, it has been observed that pabinafusp alfa was effective against both the somatic and CNS symptoms of patients with MPS-II as it can pass through the blood-brain barrier to reach the central nervous tissues. Thus, the high efficacy and safety of the novel enzyme drug are expected to increase its adoption for treating patients with hunter syndrome, thereby propelling the market's growth.
As per an article published in PLOS One, in May 2021, it has been observed that enzyme replacement therapy (ERT) is the primary treatment for mucopolysaccharidosis type-II (hunter syndrome) that helps in improving the course of diseases by reducing the somatic symptoms, including hepatomegaly and splenomegaly. In addition, as per the same source, ERT using human recombinant iduronate 2-sulphatase administered intravenously is considered the current standard of care for patients with hunter syndrome. Thus, such studies are expected to increase the demand for ERT and propel the segment's growth over the forecast period.
The growing development of enzyme replacement therapy products by companies are also likely to increase the availability of ERT therapies in the market, further augmenting its growth. For instance, in March 2021, the MHLW approved IZCARGO (pabinafusp alfa 10 mL, intravenous drip infusion) for the treatment of MPS II in Japan. It is a recombinant iduronate-2-sulfatase enzyme replacement therapy (ERT) developed using J-Brain Cargo, a proprietary technology developed by JCR, to deliver therapeutics across the blood-brain barrier (BBB).
Thus, owing to the high efficacy and safety of ERT, the rising number of research studies, and increasing product approvals, the studied segment is expected to grow over the forecast period.
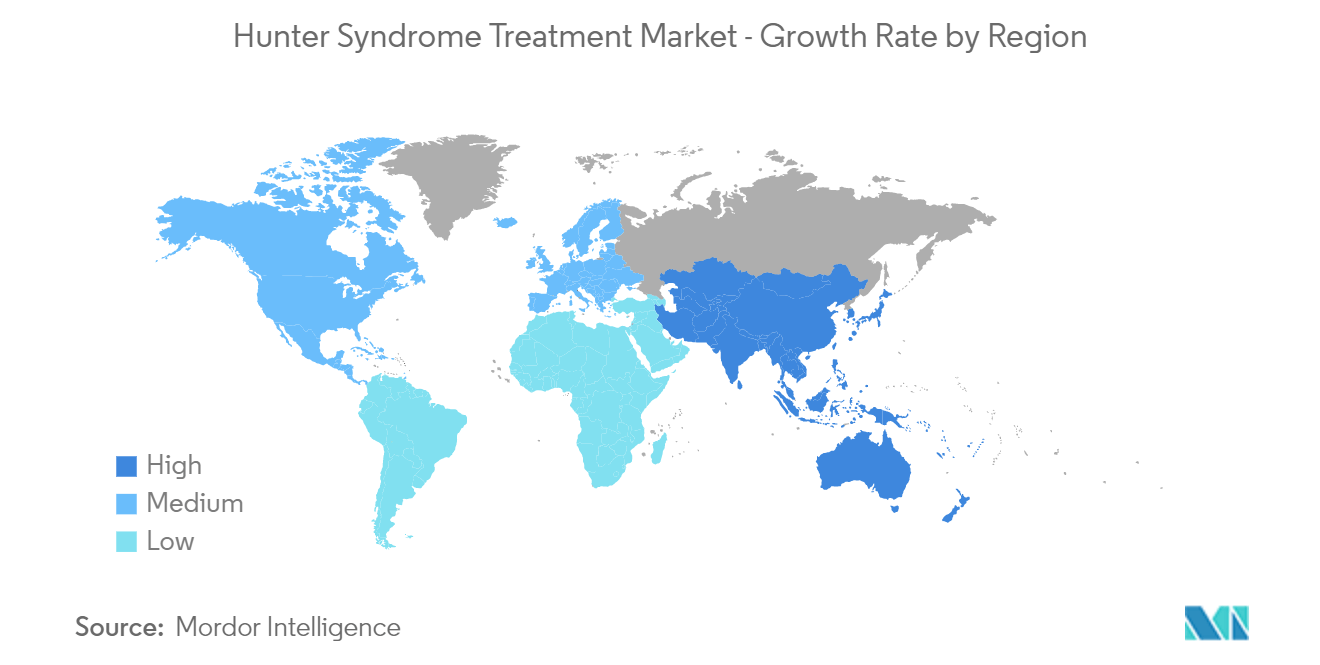
North America is Expected to Hold a Significant Share in the Hunter Syndrome Treatment Market During the Forecast Period
North America is expected to witness healthy growth over the forecast period due to the rising awareness regarding rare diseases, favorable regulations for orphan drug development, growing healthcare expenditure, and favorable reimbursement policies for expensive drugs in the region. The presence of well-established healthcare infrastructure, increasing adoption of novel therapies, and technological advancements are also expected to fuel the hunter syndrome treatment market.
The increasing focus on developing advanced products by companies and rising product launches and approvals in the region are expected to boost the market over the forecast period. For instance, in August 2022, REGENXBIO Inc. announced its plans to file a Biologics License Application (BLA) in 2024 using the FDA's accelerated approval pathway for RGX-121 for the treatment of mucopolysaccharidosis Type II (MPS II). In February 2022, Homology Medicines, Inc. presented data on HMI-203 gene therapy candidate for the treatment of hunter syndrome (MPS II), under phase I dose-escalation clinical study in adults with hunter syndrome, at the Annual WorldSymposium Meeting.
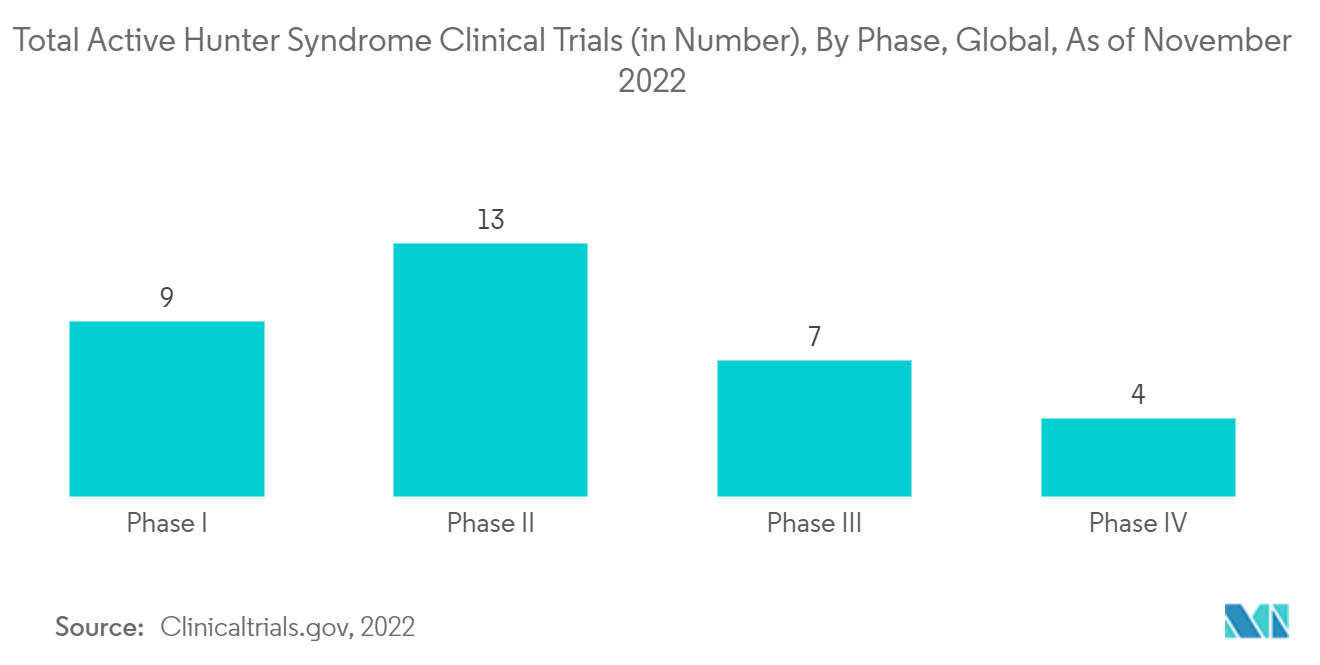
The increasing focus of key players in conducting clinical trials for evaluating the safety and effectiveness of enzyme drugs and therapies for treating patients with hunter syndrome may also contribute to the market's growth. For instance, as per Clinicaltrials.gov, as of September 2022, REGENXBIO Inc. was conducting a phase II/III clinical study to determine the safety and efficacy of RGX-121, a gene therapy, in children aged five and over with severe MPS II. As per the same source, 23 active clinical studies for hunter syndrome are ongoing in the United States, out of which nine are in phase I clinical trials, 10 in phase II, three in phase III, and one in phase IV. Hence, the high number of clinical trials in the country may boost the growth of the hunter syndrome treatment market over the forecast period.
The rising focus on developing various safe treatment options for patients with hunter syndrome and increasing product launches in the country are expected to boost the market's growth during the forecast period.