Market Size of Global Neurology Devices Industry
| Study Period | 2019 - 2029 |
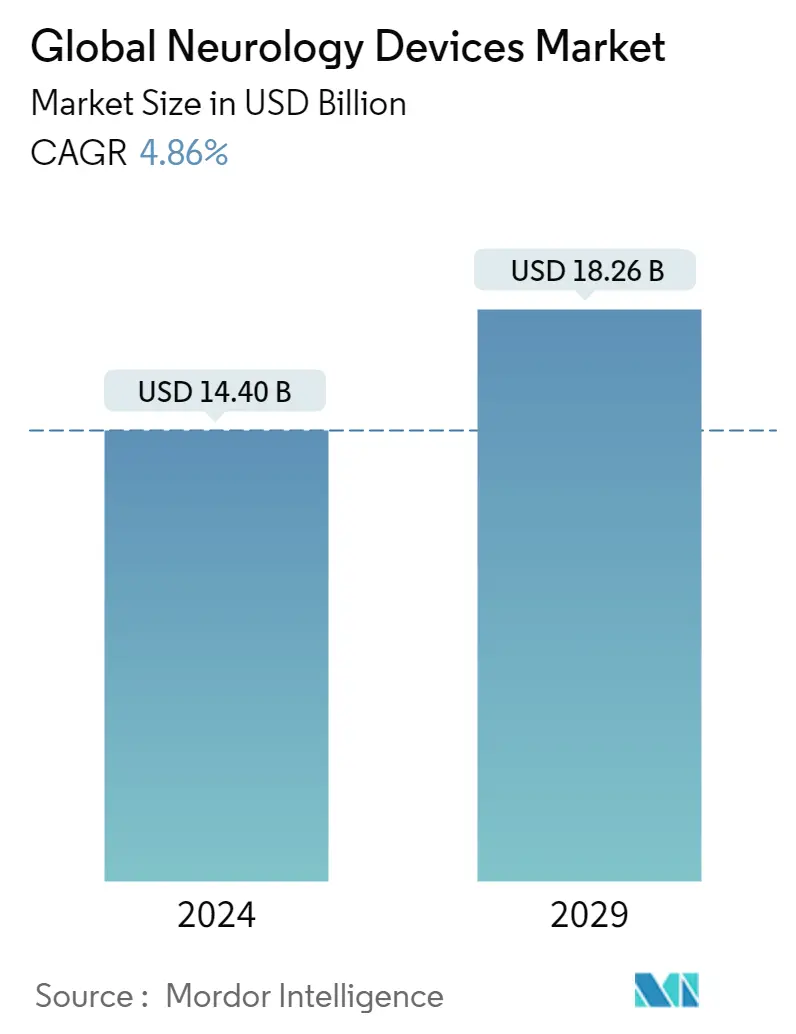
| Market Size (2024) | USD 14.40 Billion |
| Market Size (2029) | USD 18.26 Billion |
| CAGR (2024 - 2029) | 4.86 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Neurology Devices Market Analysis
The Global Neurology Devices Market size is estimated at USD 14.40 billion in 2024, and is expected to reach USD 18.26 billion by 2029, growing at a CAGR of 4.86% during the forecast period (2024-2029).
The COVID-19 pandemic had a significant impact on the global healthcare systems, with significant consequences not only for COVID-19-infected patients but also for others, resulting in the cancellation of treatment and surgical procedures to manage and reserve resources. For instance, in June 2020, the World Health Organization issued the results of a survey conducted by 155 countries for three weeks in May 2020, which showed that the prevention and treatment services of various chronic diseases, including various neurodegenerative disorders, such as Parkinson's, Alzheimer, and dementia, had been severely disrupted, and this has become a significant concern because people living with chronic diseases are at higher risk of severe COVID-19-related illness and death. In addition, 53% of the countries reported partial or complete disruption of services. This disruption in the management of different neurological conditions has impacted the lives of the patients. Moreover, the pandemic can also have a negative impact on new product launches, which are estimated to be the most prominent factor in the growth of the market studied. For instance, in April 2020, Biogen delayed Alzheimer's and dementia drug filing to Food and Drug Administration (FDA) while citing the complexity of data analysis and the impact of COVID-19 on its R&D resources. With the resumed services, treatment and surgical procedures and increased hospital visits are likely to increase the demand for neurology devices and treatment for various neurological disorders, thereby impacting the market growth.
The factors propelling the growth of the market are an increase in the incidence of neurological disorders, huge investments by private players in neurology devices, increase in research and development in the field of Neurotherapy.
The increasing burden of neurological disorders among the population is the key factor driving the demand for neurology devices over the forecast period. For instance, according to data published by the United Nations, in February 2021, about 1 billion people, nearly one in six of the world's population, suffer from neurological disorders, such as Alzheimer's and Parkinson's disease, strokes, multiple sclerosis, epilepsy migraine, brain injuries, and neuro infections in 2020. Additionally, as per the same source 62 million suffered from cerebrovascular disease, 50 million people suffered from epilepsy, 326 million from migraine, and 24 million from Alzheimer's disease and other dementias in 2019. Thus, the rising prevalence of neurological disorders among the population is expected to increase the demand for neurology devices to restore hearing and sight and provide an increased function for limb loss or congenital limb differences, thereby expected to augment the market growth.
Furthermore, the rising awareness among the population about neurological diseases and their treatment options is expected to drive market growth. For instance, as per a report published by World Health Organization (WHO), in April 2022, titled 'Draft Intersectoral Global Action Plan on Epilepsy and Other Neurological Disorders 2022-2031' 100% of countries are expected to have at least one functioning awareness campaign or advocacy program for neurological disorders by 2031. Such initiatives by the renowned organization are expected to drive the market in the coming future.
However, the high cost of equipment and stringent Food and Drug Administration validation and guidelines for new devices are likely to restrain the growth of the market.

