Chlamydia Infection Diagnostics Market Size and Share
Chlamydia Infection Diagnostics Market Analysis by Mordor Intelligence
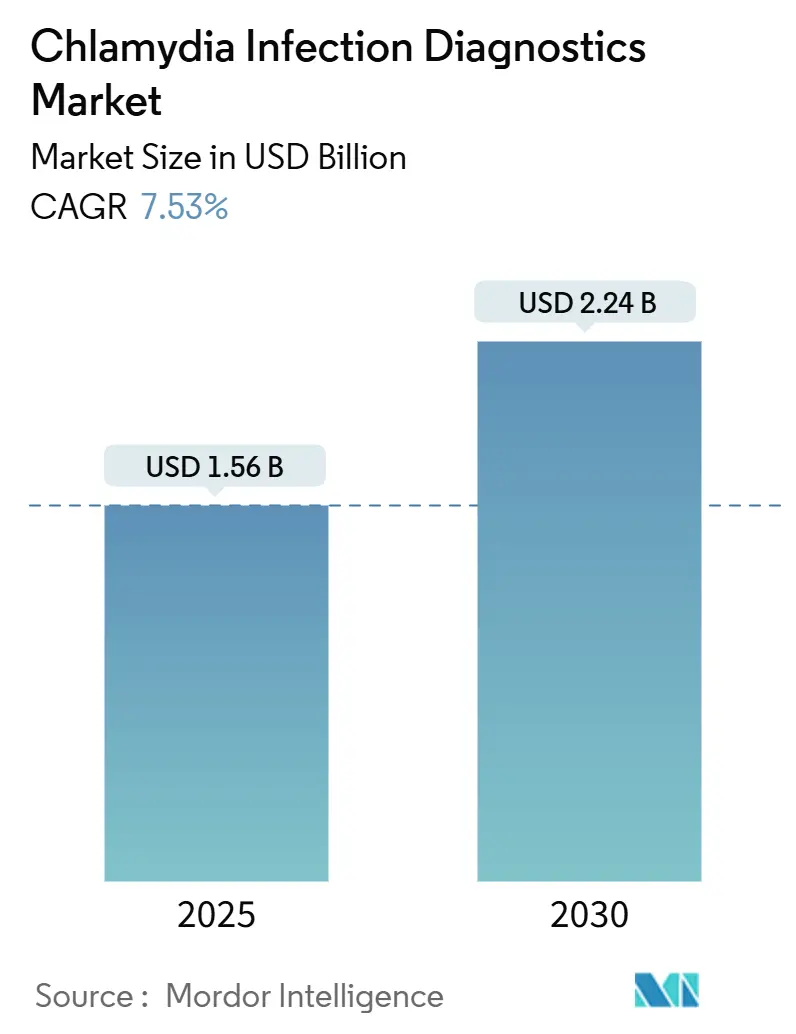
The Chlamydia Infection Diagnostics Market size is estimated at USD 1.56 billion in 2025, and is expected to reach USD 2.24 billion by 2030, at a CAGR of 7.53% during the forecast period (2025-2030).
Demand growth is anchored in the global rise of sexually transmitted infections, the high rate of asymptomatic cases, and expanding access to molecular platforms that deliver rapid, accurate results. Regulatory catalysts, most notably the FDA’s 2025 approval of the first over-the-counter chlamydia home test, are accelerating commercial timelines for similar products while shifting testing closer to consumers. Companies are prioritizing automation, multiplexing, and digital connectivity to shorten time-to-result, contain costs, and reduce loss-to-follow-up. In high-income countries, reimbursement frameworks and mandatory screening recommendations underpin steady test volumes, whereas emerging economies present volume upside once infrastructure bottlenecks are resolved.
Key Report Takeaways
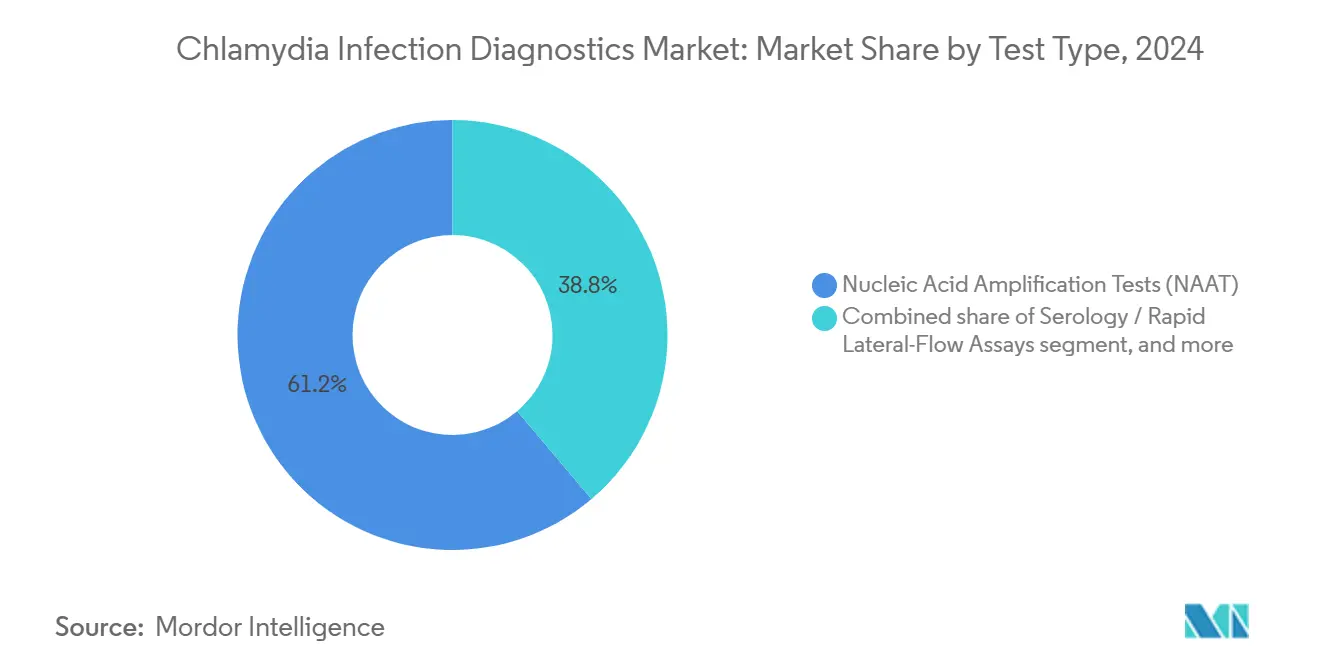
- By test type, Nucleic Acid Amplification Tests (NAAT) led with 61.23% of Chlamydia infection diagnostics market share in 2024, while Serology/Rapid Lateral-Flow Assays are projected to expand at a 4.67% CAGR to 2030.
- By specimen type, vaginal swabs accounted for 34.56% share of the Chlamydia infection diagnostics market size in 2024; urine samples are advancing at a 4.23% CAGR through 2030.
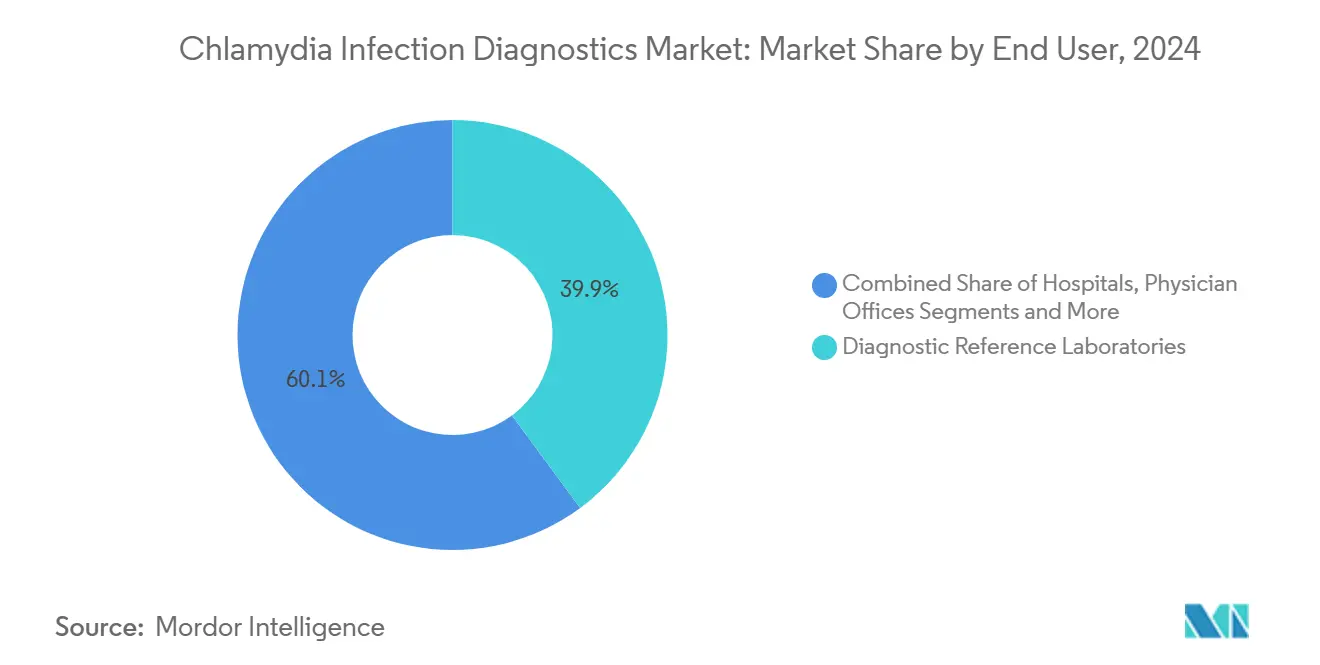
- By end user, diagnostic reference laboratories held 39.87% of the Chlamydia infection diagnostics market size in 2024, whereas home-care/direct-to-consumer channels exhibit the fastest 6.55% CAGR over the forecast horizon.
- By geography, North America dominated with 38.88% revenue share in 2024, but Asia-Pacific is set to post the highest 4.67% CAGR to 2030.
Global Chlamydia Infection Diagnostics Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Increasing global burden of sexually transmitted infections | +2.1% | Global; highest impact in Asia-Pacific & Sub-Saharan Africa | Long term (≥ 4 years) |
| Rising government funding for STI screening programs | +1.8% | North America & EU; expanding to Asia-Pacific | Medium term (2-4 years) |
| Technological advancements in molecular diagnostic platforms | +1.5% | Global; led by North America & Europe | Short term (≤ 2 years) |
| Growing acceptance of home-based self-testing solutions | +1.2% | North America & EU; early adoption in urban Asia-Pacific | Medium term (2-4 years) |
| Expansion of public–private partnerships in diagnostic infrastructure | +0.8% | Asia-Pacific core; spill-over to Middle East & Africa and Latin America | Long term (≥ 4 years) |
| Integration of point-of-care testing in primary healthcare settings | +0.9% | Global; accelerated adoption in rural and underserved areas | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Increasing Global Burden of Sexually Transmitted Infections
Chlamydia case notifications have climbed steeply, with 216,508 confirmed cases reported across 27 EU/EEA countries in 2022, a 16% rise from 2021.[1]European Centre for Disease Prevention and Control, “Chlamydia Infections Annual Epidemiological Report for 2023,” ecdc.europa.eu Young adults aged 20-24 bear a disproportionate share, while infections among men who have sex with men have surged 72% over five years, demonstrating shifting transmission patterns. Because over 80% of infections present without symptoms, disease prevalence is substantially under-reported, pushing health systems toward routine, population-level screening. These epidemiological realities expand the Chlamydia infection diagnostics market by broadening the pool of individuals who require testing in both clinical and community settings.
Rising Government Funding for STI Screening Programs
Government budgets earmarked for STI control have risen across major markets. In the United States, the CDC’s Prevention and Control program channels multi-year grants to state health departments, while its 2024 awards provided USD 9 million to scale integrated HIV/STI screening services.[2]Centers for Disease Control and Prevention, “STD Prevention and Control for Health Departments Cooperative Agreement,” cdc.gov A parallel five-year laboratory-capacity initiative (NOFO CD-25-0019) kicks off in July 2025, ensuring reagent supply, staff training, and quality assurance for nucleic acid amplification testing. Similar policy alignment is taking shape internationally as the WHO positions NAAT as the gold-standard test, standardizing procurement criteria and reinforcing the demand curve for the Chlamydia infection diagnostics market.
Technological Advancements in Molecular Diagnostic Platforms
Rapid molecular platforms are compressing result times from days to minutes. Roche’s cobas liat delivers on-site PCR results for chlamydia in 20 minutes and received FDA clearance with a CLIA waiver in January 2025. Abbott’s high-throughput Alinity m system simultaneously detects four pathogens at scale, while integrated internal controls bolster accuracy. Automation not only lifts throughput but also cuts labor requirements, enabling point-of-care deployment in urgent care centers, retail clinics, and mobile vans. Consequently, healthcare providers can screen more individuals, treat positives immediately, and lower onward transmission—drivers that sharpen the competitive edge of the Chlamydia infection diagnostics market.
Growing Acceptance of Home-Based Self-Testing Solutions
The FDA’s March 2025 authorization of an over-the-counter chlamydia home test achieved 97.2% sensitivity and 98.8% negative predictive value. Comparable moves by Australia’s Therapeutic Goods Administration extend the trend globally. [3]Therapeutic Goods Administration, “Australian Register of Therapeutic Goods Public Summary for Touch Biotechnology Self-Test,” tga.gov.au Clinical data show that self-collected pharyngeal and rectal swabs are non-inferior to clinician-collected samples, even identifying infections missed in clinics. Integration with telehealth apps delivers private counseling and prescription services, removing privacy-related barriers and enlarging the Chlamydia infection diagnostics market, particularly among tech-savvy younger adults.
Restraints Impact Analysis
| Restraints Impact Analysis | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Persistent social stigma around STI testing | -1.4% | Global; strongest in conservative societies & rural areas | Long term (≥ 4 years) |
| Inadequate laboratory infrastructure in emerging economies | -1.1% | Sub-Saharan Africa, parts of Asia-Pacific & Latin America | Long term (≥ 4 years) |
| High cost of advanced molecular diagnostic tests | -0.8% | Low- and middle-income countries globally | Medium term (2-4 years) |
| Regulatory and reimbursement uncertainties across regions | -0.6% | Emerging markets and countries with evolving healthcare policies | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Persistent Social Stigma Around STI Testing
Stigma continues to deter individuals from seeking diagnosis. Surveys show high hesitancy among adolescents and rural residents who fear judgment or breaches of confidentiality, suppressing clinical visitation rates even when symptoms are present. While home tests offer anonymity, social norms in many regions still discourage purchase and use, limiting uptake and tempering growth in the Chlamydia infection diagnostics market.
Inadequate Laboratory Infrastructure in Emerging Economies
Many low-income regions lack reliable power, cold chain capacity, or trained staff, which curtails test availability and accuracy. A Northern Ghana survey found health centers performed only 2 of 20 WHO-essential tests, underscoring systemic gaps that restrict test volumes. Supply-chain fragmentation further constrains reagent availability, delaying diagnosis and treatment. These deficiencies cap market penetration despite clear public-health need.
Segment Analysis
By Test Type: NAAT Maintain Leadership as Rapid Assays Gain Ground
The Chlamydia infection diagnostics market size for NAAT reached USD 0.95 billion in 2024 and is growing at a stable 7.1% CAGR. Culture tests, while still essential for antimicrobial-resistance surveillance, ceded routine detection to NAAT because of longer incubation times. Rapid lateral-flow assays, projected to post the segment’s quickest 4.67% CAGR, leverage low-cost cartridges and minimal hardware to serve pharmacies, outreach vans, and sexual-health clinics. Regulatory momentum favors hybrid platforms that fuse NAAT accuracy with rapid assay turnaround, suggesting portfolio diversification will decide future competitive standings.
Demand for multiplex panels that detect chlamydia, gonorrhea, and trichomonas in one run is increasing. Abbott, Roche, and Hologic are upgrading firmware to integrate new targets without hardware changeovers, preserving capital investments for laboratories and sustaining brand lock-in. High throughput also meets bulk-testing needs driven by government-funded screening, reaffirming NAAT’s dominance within the broader Chlamydia infection diagnostics market.

By Specimen Type: Urine Collection Challenges Swab Tradition
Vaginal swabs generated the largest Chlamydia infection diagnostics market size among specimens at USD 0.54 billion in 2024. Nevertheless, urine samples are posting the highest growth thanks to non-invasive self-collection, no need for speculum examination, and high patient acceptance. Recent meta-analyses confirm equivalent sensitivity between urine NAAT and swab NAAT, encouraging physicians to liberalize specimen policies.
Extracervical sites are under-tested but epidemiologically important; rectal and pharyngeal sampling uncovers occult infections in men who have sex with men and women practicing oral sex. Home-collection kits now include color-coded swabs and pictorial instructions to reduce user error, broadening specimen diversity. As telemedicine consultations grow, flexible specimen options will remain pivotal to maximizing reach for the Chlamydia infection diagnostics market.
By End User: Reference Labs Defend Share While Home Testing Scales
Reference laboratories processed the majority of chlamydia tests by leveraging economies of scale, with automation lines running 24/7 to support hospital and physician-office demand. However, the Chlamydia infection diagnostics market is witnessing the fastest revenue expansion in home-care/direct-to-consumer channels. Tablets, mail-back envelopes, and app-delivered results combine to deliver discreet, end-to-end service within three days. Clinical governance is maintained through tele-consultation networks that prescribe doxycycline within hours of result release.
Hospitals and STI clinics continue to account for a sizeable share, especially for complicated cases or co-infection management. Yet even these providers contract out routine NAAT to centralized labs or authorize bedside rapid assays, reflecting a blurring of traditional end-user demarcations. Ultimately, firms that align product formats with each venue’s workflow will win share in the evolving Chlamydia infection diagnostics market.

Geography Analysis
North America held 38.88% of global revenue in 2024, buoyed by mandatory annual screening guidelines for sexually active women under 25 and comprehensive third-party reimbursement that removes patient cost hurdles. Medicare’s preventive-services provisions waive copays and deductibles for STI testing, preserving test volumes even when insurance deductibles trend upward. FDA fast-track pathways also shorten time-to-market for innovators, making the region the premier launchpad for new assays.
Asia-Pacific is the fastest-growing arena, advancing at 4.67% CAGR as public-private initiatives extend laboratory grids and telehealth apps penetrate smartphone-dense populations. Australia’s pharmacy-based self-testing rollout, endorsed by the Therapeutic Goods Administration, illustrates policy openness to decentralized screening. Pilot programs in rural indigenous clinics cut the median time from sample collection to treatment from eight days to two, highlighting how rapid diagnostics can curb transmission.
Europe remains a mature but expanding market. The 2025 European guideline solidifies NAAT as first-line diagnosis and recommends doxycycline therapy for seven days, standardizing clinical practice across member states. Surveillance data reveal the region’s steady infection climb, thus keeping screening budgets intact. Investments such as bioMérieux’s EUR 25 million facility upgrade reinforce Europe’s commitment to in-house R&D and advanced microbiology platforms, sustaining its strategic weight in the Chlamydia infection diagnostics market.
Competitive Landscape
The Chlamydia infection diagnostics market is moderately concentrated. Hologic, Abbott, Roche, and bioMérieux command core molecular franchises supported by wide installed bases and extensive regulatory clearances. Innovation cycles focus on greater multiplexing and reduced sample-to-answer times; Roche’s 20-minute cobas liat and Hologic’s Panther Fusion upgrades exemplify this arms race. Acquisitions are a primary route to new technology: bioMérieux purchased SpinChip Diagnostics for EUR 138 million to obtain 10-minute whole-blood immunoassays, while OraSure’s acquisition of Sherlock Biosciences secures CRISPR-based lateral-flow assets.
Start-ups are exploiting funding from the Bill & Melinda Gates Foundation, NIH, and European Innovation Council to commercialize isothermal and digital PCR formats that shed expensive thermal-cycling components. Linear Diagnostics’ EXPAR technology aims for 20-minute detection with single-use cartridges, and Aptitude Medical’s Metrix seeks both point-of-care and OTC approvals. Market entry barriers remain high because CLIA waiver and CE-IVDR conformity demand robust clinical evidence. Nonetheless, regulatory shifts—such as the FDA’s De Novo classifications for home tests—are lowering hurdles for consumer-facing devices, widening competitive participation in the Chlamydia infection diagnostics market.
Chlamydia Infection Diagnostics Industry Leaders
Hologic Inc.
Danaher
Abbott Laboratories
F. Hoffmann-La Roche Ltd.
Becton, Dickinson & Co.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- June 2025: Linear Diagnostics secured £1 million from the UK NIHR to develop EXPAR-based 20-minute chlamydia tests, with clinical trials slated for 2026.
- May 2025: Umeå University and Michigan State University researchers identified a molecule that selectively kills chlamydia bacteria while sparing normal flora, opening a pathway for adjunctive therapeutics.
- March 2025: Sanofi received FDA fast-track designation for its mRNA chlamydia vaccine, launching Phase 1/2 trials in adults aged 18-29.
- March 2025: The FDA authorized Visby Medical’s over-the-counter Women’s Sexual Health Test, the first home test for chlamydia, gonorrhea, and trichomoniasis.
Global Chlamydia Infection Diagnostics Market Report Scope
As per the scope of the report, chlamydia is a curable and common sexually transmitted infection that may not cause symptoms. It spreads through vaginal, anal, and oral sex. The infection is carried in semen (cum), pre-cum, and vaginal fluids. Chlamydia can infect the penis, vagina, cervix, anus, urethra, eyes, and throat. It affects people of all ages but is most common in young women.
The Chlamydia Infection Diagnostics Market is Segmented by Type (Culture Tests, Nucleic Acid Amplification Test (NAAT), Direct Fluorescent Antibody Test, Serology Tests, and Others), End-User (Hospitals, Diagnostic Centers and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD million) for the above segments.
| Culture Tests |
| Nucleic Acid Amplification Tests (NAAT) |
| Serology / Rapid Lateral-Flow Assays |
| Direct Fluorescent Antibody Tests |
| Other Test Types |
| Vaginal Swabs |
| Urine Samples |
| Endocervical Swabs |
| Rectal & Pharyngeal Swabs |
| Blood / Serum |
| Hospitals |
| Diagnostic Reference Laboratories |
| Sexual-Health / STI Clinics |
| Physician Offices |
| Home-care / Direct-to-Consumer |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Test Type | Culture Tests | |
| Nucleic Acid Amplification Tests (NAAT) | ||
| Serology / Rapid Lateral-Flow Assays | ||
| Direct Fluorescent Antibody Tests | ||
| Other Test Types | ||
| By Specimen Type | Vaginal Swabs | |
| Urine Samples | ||
| Endocervical Swabs | ||
| Rectal & Pharyngeal Swabs | ||
| Blood / Serum | ||
| By End User | Hospitals | |
| Diagnostic Reference Laboratories | ||
| Sexual-Health / STI Clinics | ||
| Physician Offices | ||
| Home-care / Direct-to-Consumer | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the current size of the Chlamydia infection diagnostics market?
The market generated USD 1.56 billion in 2025 and is forecast to reach USD 2.24 billion by 2030 at a 7.53% CAGR.
Which test type holds the largest share in the Chlamydia infection diagnostics market?
Nucleic Acid Amplification Tests led with 61.23% market share in 2024 owing to high sensitivity and widespread clinical adoption.
Why is Asia-Pacific the fastest-growing regional market?
Governments are investing in laboratory networks, mobile clinics, and home-testing approvals, driving a 4.67% CAGR through 2030.
How is home testing changing the competitive landscape?
Social stigma deters testing in many communities, and inadequate laboratory infrastructure in low-income regions limits access despite rising disease burden.
Which companies made notable strategic moves recently?
BioMérieux bought SpinChip for point-of-care technology, while OraSure’s purchase of Sherlock Biosciences secured CRISPR-based self-testing assets.
Page last updated on: