Market Size of Track and Trace Solutions Industry
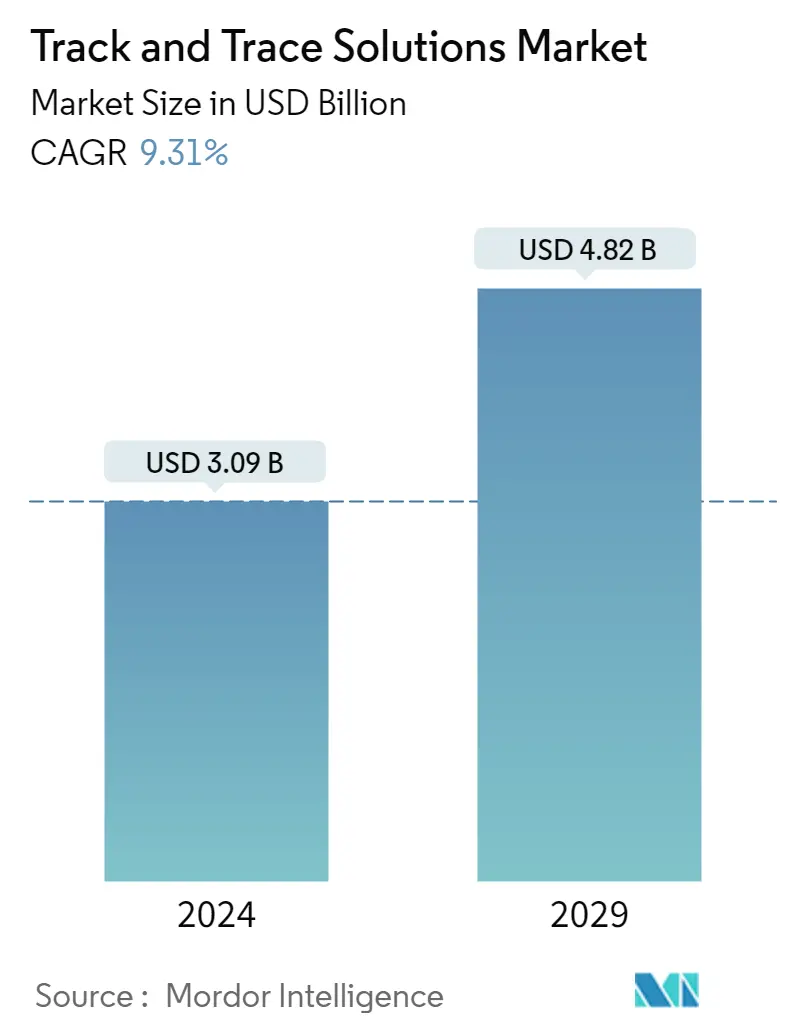
| Study Period | 2019 - 2029 |
| Market Size (2024) | USD 3.09 Billion |
| Market Size (2029) | USD 4.82 Billion |
| CAGR (2024 - 2029) | 9.31 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Track and Trace Solutions Market Analysis
The Track and Trace Solutions Market size is estimated at USD 3.09 billion in 2024, and is expected to reach USD 4.82 billion by 2029, growing at a CAGR of 9.31% during the forecast period (2024-2029).
With the development in the COVID-19 vaccine and the various treatment and diagnostic devices and instruments to curb the pandemic, the risk of counterfeit drugs and diagnostic devices increased globally, which impacted the pharmacy automation market. For example, in March and April 2020, Homeland Security in the United States also notified 19,000 suspects of COVID-19-related domain names. As a result, it seized over USD 3.2 million linked to 494 shipments of mislabeled, fraudulent, unauthorized, or prohibited COVID-19 test kits, treatment kits, homeopathic remedies, purported antiviral products, and PPE kits, which further led to 11 arrests. Such instances are expected to drive the adoption of track and trace solutions.
The major factors propelling the growth of the track and trace solutions market include the growth of the medical devices and pharmaceutical industries, an increase in implementation of serialization, a rise in the number of packaging-related product recalls, and an increasing number of counterfeit drugs.
Drug counterfeiting is one of the significant problems in large pharmaceutical and biopharmaceutical companies. As per the article published by the World Health Organization in 2019, about 1 out of 10 medical products that circulate in low- and middle-income countries are found to be substandard or falsified. This factor, consequently, is driving the adoption of track and trace solutions among drug manufacturers and other end users. Furthermore, in December 2020, Janssen Pharmaceutical, a subsidiary of Johnson & Johnson, notified that counterfeit SYMTUZA (darunavir/cobicistat/emtricitabine/tenofovir alafenamide) had been distributed to over three pharmacies in the United States. Janssen is currently working closely with the US Food and Drug Administration (FDA) to prevent further distribution and support the agency's investigation into the reported instances. Hence, these factors are expected to fuel market growth.
However, factors such as high costs associated with serialization and aggregation and lack of common standards for serialization and aggregation may hinder the market growth.
