Market Size of Global Regulatory Reporting and Compliance Industry
| Study Period | 2019 - 2029 |
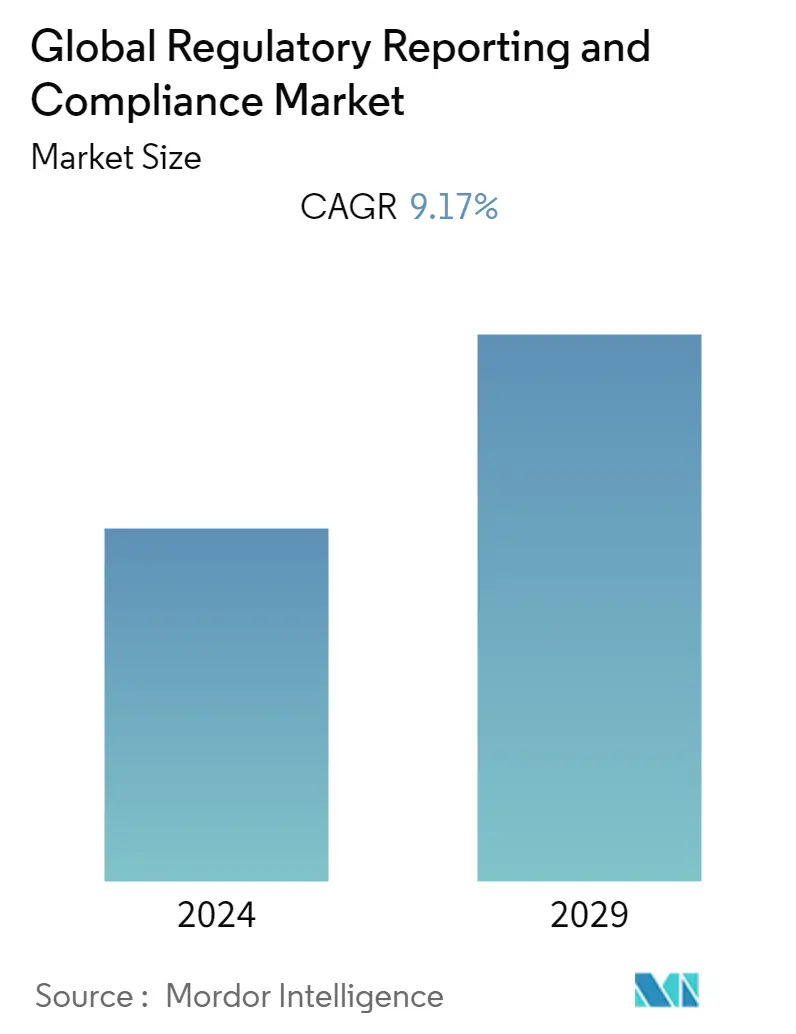
| Base Year For Estimation | 2023 |
| CAGR | 9.17 % |
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Regulatory Reporting & Compliance Market Analysis
The regulatory reporting and compliance market studied is expected to register a CAGR of 9.17% over the forecast period.
The regulatory bodies remain strict and stick to their regulatory processes. The outbreak of the COVID-19 pandemic enabled flexibility in regulations to make treatment and cure rapidly accessible and developed in challenging times. The regulatory bodies, such as the Medicines and Healthcare products Regulatory Agency (MHRA), the United States Food and Drug Administration (USFDA), and European Medicines Agency(EMA), took proactive steps to support the industry. To accelerate the approval procedure, regularly updated guidelines on flexibility were issued to meet compliance, the routine inspection was postponed, and reporting was shifted to electronic mode. In April 2020, the United States Food and Drug Administration (USFDA) announced Coronavirus Treatment Acceleration Program(CTAP), under which till May 2021, 460+trials are reviewed by the FDA, and 620+ are in the planning stage of the drug development program. Thus, the COVID-19 pandemic is expected to show a positive impact on the regulatory reporting and compliance market, and it could be considered a catalyst for the growth of the market.
The studied market growth is attributed to the growing demand for a faster approval process, the continuously changing regulatory landscape, and the digitalization of the regulatory process. With the emergence of new diseases and new treatment fields, like precision medicine, targeted gene therapy, and specialty medicines, the need for a faster approval process is increasing to benefit the patients at the earliest. In January 2021, the United States Food and Drug Administration (USFDA) reported that in 2020, many innovative and advanced therapy drugs, like the first-ever therapy for Chaga's disease for pediatric patients of less than two years, a new therapy for Parkinson's disease for curing off episodes, was approved. The continuous change in the regulatory landscape, like different analytical criteria and inspection requirements across different regions, is a driver for the regulatory reporting and compliance market. Moreover, the difference in Good Manufacturing Practices (GMP) by different health authorities, like European Medicines Agency(EMA) and the United States Food and Drug Administration (USFDA), makes it difficult for global players to compliance with them. Most regulatory affairs functions rely on a manual process, which becomes unsustainable in the digitally transforming world. The digitalization and involvement of automated technological solutions will provide a significant reduction in time, cost, and efforts to achieve business goals. The unlock to intelligent automation technology will enable publishing, submissions, labeling, and approval notifications to configure to faster design improvement and governance of workflow in a transparent and trackable manner. Thus, the regulatory reporting and compliance market is expected to witness huge growth over the forecast period.
However, regulatory reporting and compliance are mandatory procedures for the healthcare industry. The lack of technologically advanced infrastructure in developing countries and complicated local and global regulatory norms may obstruct the growth of the market.

