Market Size of Global Real-World Evidence Solutions Industry
| Study Period | 2019 - 2029 |
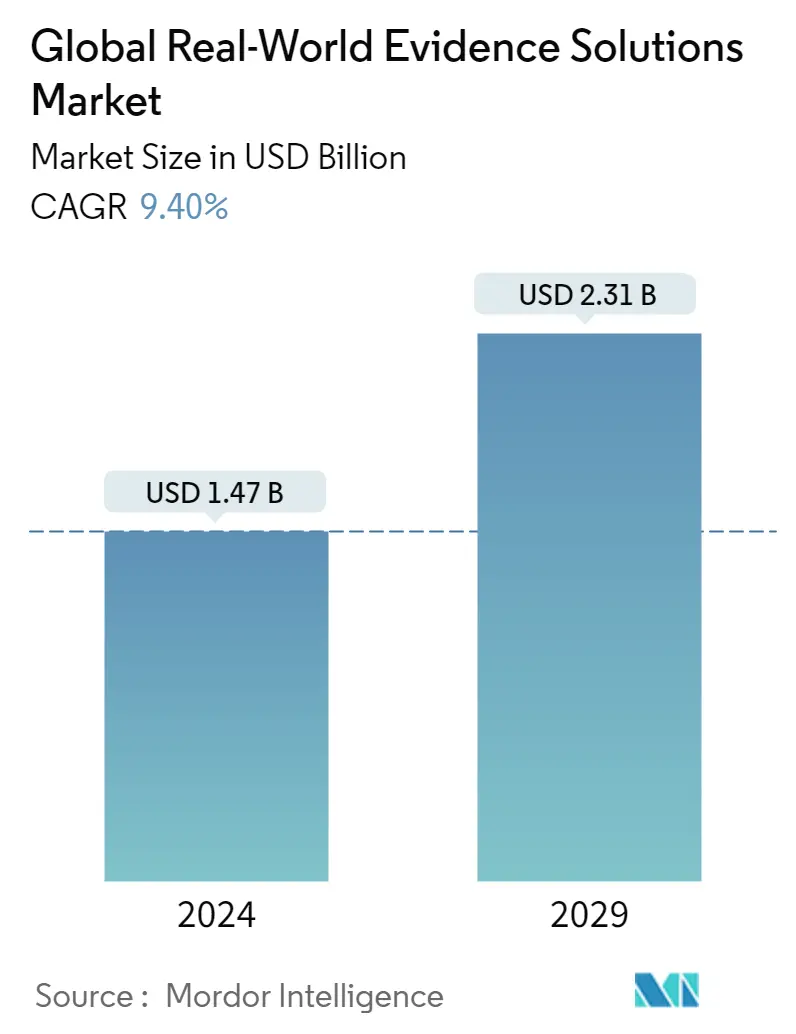
| Market Size (2024) | USD 1.47 Billion |
| Market Size (2029) | USD 2.31 Billion |
| CAGR (2024 - 2029) | 9.40 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Real-World Evidence Solutions Market Analysis
The Global Real-World Evidence Solutions Market size is estimated at USD 1.47 billion in 2024, and is expected to reach USD 2.31 billion by 2029, growing at a CAGR of 9.40% during the forecast period (2024-2029).
The contagious coronavirus has a potential economic impact and implications on most sectors, including the pharmaceutical industry. Governments worldwide are now responding to the threat of COVID-19 with all the essential measures, such as social distancing, nationwide lockdown, travel restrictions, and large-scale quarantines that are anticipated to negatively impact businesses and consumer spending.
In the context of the COVID-19, government organizations in various countries, research institutes, and many biotech and pharmaceutical firms are focusing on the development of effective and rapid diagnostic technologies. Moreover, many healthcare providers and drug developers have been stretching their available resources to curb the spread and develop effective medicines/vaccines for the COVID-19 pandemic. The market is, thus, found witnessing a trend that is shifting toward digitalization and patient-centered virtual care. This emphasizes the need for effective real-world solutions.
In April 2020, two healthcare data science companies, Aetion and HealthVerity, were found collaborating, extending their current relationship, to extract real-world medical evidence from electronic health records and advise drug makers and regulators on COVID-19 treatments.
The real-world evidence solutions market will show rapid growth due to the shift from volume to value-based care, increasing aging population and prevalence of chronic diseases, delays in drug development, and subsequent increase in development costs. According to the World Ageing Population report, in 2020 highlights, approximately 727 million people living worldwide were aged 65 years and above, and the number is expected to reach 1.5 billion by 2050. It has been calculated that almost half of the total chronic disease deaths are attributable to cardiovascular diseases, cancer, obesity, and diabetes which are showing increasing trends.
Pharmaceutical companies take several years to introduce a new drug molecule to market, and they invest billions in research and development with a low probability of success. To shorten the drug development cycles, pharma research centers need a clear insight on treatment options that work in a wide population compared to few patient pools in clinical trials. The use of real-world evidence (RWE) insights is thus found to increase as pharma and biotech companies realize that real-world evidence (RWE) can be important in drug development and life-cycle management and increase the innovation efficiency cost of clinical trials.
The big pharmaceutical companies are found using a clinico-genomic database with tumor sequencing information for cancer discovery studies and targeted drug development. Companies are thus able to broaden their indication range and conduct a small number of clinical trials for the rare biomarker-defined population. Thus it indicates the growth of the real-world evidence solutions market during the forecast period.
In addition, the real-world data sets are widely adopted by end users for real-world evidence (RWE) for various applications such as drug development and approvals, market access and reimbursement/coverage decisions, clinical decision-making, and medical device development and approvals, among others, which helps in the growth of the overall market.
