Market Size of Global Pediatric Vaccines Industry
| Study Period | 2019 - 2029 |
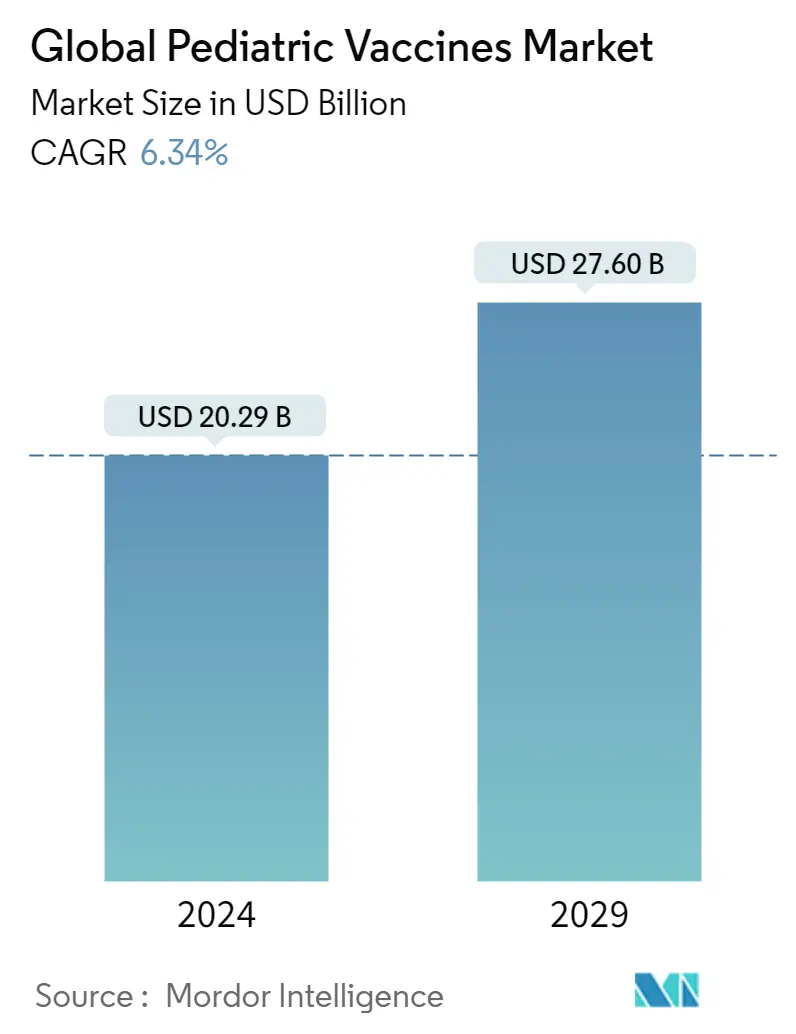
| Market Size (2024) | USD 20.29 Billion |
| Market Size (2029) | USD 27.60 Billion |
| CAGR (2024 - 2029) | 6.34 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Pediatric Vaccines Market Analysis
The Global Pediatric Vaccines Market size is estimated at USD 20.29 billion in 2024, and is expected to reach USD 27.60 billion by 2029, growing at a CAGR of 6.34% during the forecast period (2024-2029).
The pediatric vaccine market experienced a severe setback in terms of growth as a result of the abrupt outbreak of COVID-19. As the number of COVID-19 cases surged, healthcare systems redirected their focus to managing the disease, delaying the prevention, diagnosis, and treatment of other chronic illnesses in the pediatric population. According to World Health Organization(WHO) data in 2021, the number of entirely unvaccinated children rose by 3.4 million in 2020. In 2020, only 19 vaccines were introduced, which is less than half of any year in the previous two decades. Moreover, in 2020, 1.6 million female children were not fully immunized against the human papillomavirus (HPV) as the year before. Thus, the impact of the pandemic on the market in the initial phase was adverse. However, due to the relaxation of restrictions and the resumption of R&D activities and clinical trials for pediatric vaccinations, the market is likely to gain traction in the coming years. For instance, the United States Food and Drug Administration (FDA) has approved an emergency use authorization for a Pfizer-BioNTech COVID-19 vaccination for children aged 5 to 11. For this vaccine, two doses are given three weeks apart.
Furthermore, the rising burden of chronic diseases, the necessity and awareness of vaccination, developments in the route of vaccine administration, and an increase in government and non-government financing for research and development are some of the significant drivers propelling the market's growth.
For instance, as per the article titled "Clinical Characteristics, Antimicrobial Resistance, and Risk Factors for Mortality in Pediatric Invasive Pneumococcal Disease in Beijing" published in BMC Infectious Diseases in April 2022, researchers looked at clinical features, antibiotic susceptibility, and risk factors for mortality in children with invasive pneumococcal disease (IPD). The research study concluded that meningitis was the most common clinical manifestation of IPD, and it was commonly linked to death. Since the strains in the PCV13 vaccination would cover the majority of cases, PCV13 should be used more widely. Furthermore, according to World Health Organization (WHO) estimates from March 2022, the measles virus is one of the world's most contagious human diseases, affecting over 100,000 children each year, and rubella is a leading vaccine-preventable cause of birth abnormalities. Only two doses of a safe and efficient vaccine can prevent measles and rubella. Additionally, as per an article titled "Measles" published in January 2022, young children and pregnant women who have not been vaccinated are at significant risk of developing measles. Due to growing levels of vaccination coverage and changes in population immunity at various ages, there has been a recent shift to older children and teenagers. Passive antibody transfer protects young infants who have gained immunity from measles, but as these antibodies diminish, they become more susceptible.
As immunization plays a significant role in disease prevention, there are several international programs for children, which may vary based on the disease. According to World Health Organization (WHO) data published in March 2022, India initiated the Intensified Mission Indradhanush (IMI) 4.0 in phases to meet its objective of providing 90 percent vaccination coverage and beyond to expand the scope of the universal immunization program, which vaccinates nearly 3 crore pregnant women and 2.6 crore children each year. The National Strategic Plan for Achieving and Sustaining Measles and Rubella Elimination in India was adopted , and the MR vaccine campaign immunized nearly 324 million children till 2020. For the first time, India met the global guideline of 2 per 100,000 population for non-measles, non-rubella discard rates in the last six months of 2021. Thus, the aforementioned factors are anticipated to drive the market's growth over the forecast period.
The high cost of immunization and limited access to healthcare in low- and middle-income countries, on the other hand, are expected to hamper the market's growth over the forecast period.
