Market Trends of Global Pediatric Medical Devices Industry
This section covers the major market trends shaping the Pediatric Medical Devices Market according to our research experts:
Neonatal ICU Devices Segment is Expected to Show Significant Growth Over the Forecast Period
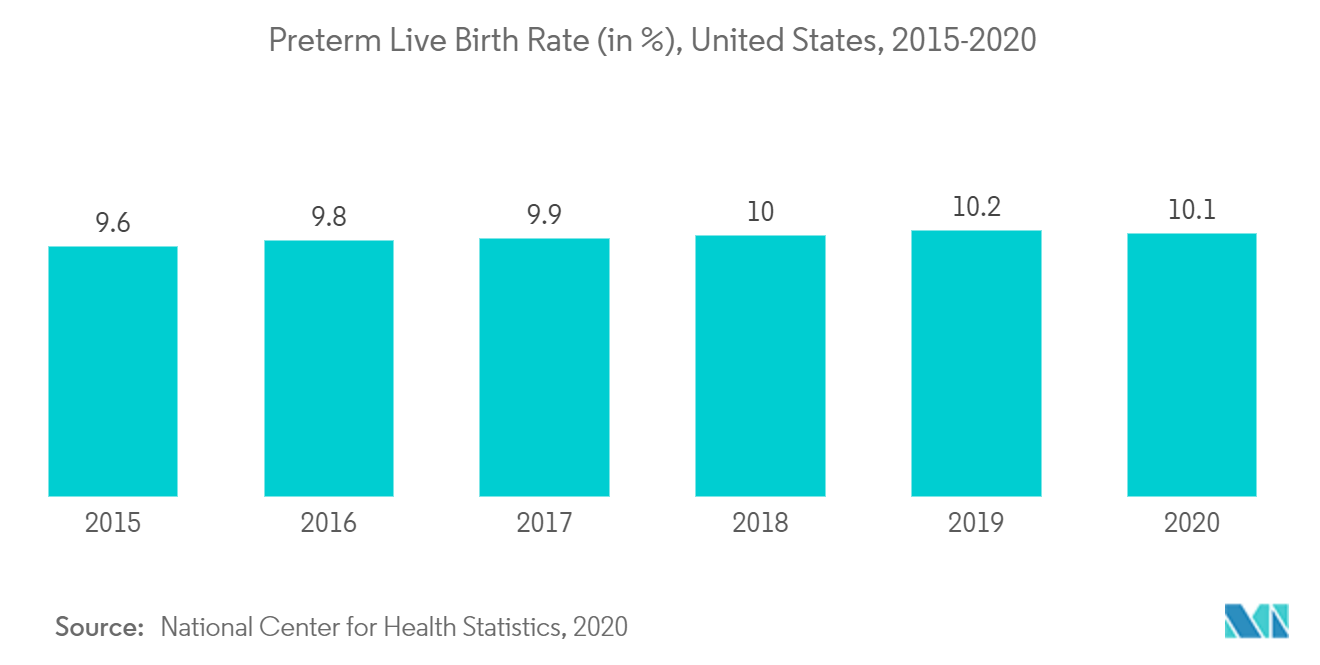
Due to challenges during delivery, early birth, or health issues after birth, children who require NICU care are frequently moved there within 24 hours of birth. A baby is considered preterm if it is born before 37 weeks of pregnancy. One out of every ten babies born in the United States in 2020 suffered from preterm birth, according to the Centers for Disease Control and Prevention's November 2021 update. From 10.2 percent in 2019 to 10.1 percent in 2020, the preterm birth rate decreased by one percent. Babies born prematurely are more prone to illnesses like sepsis, pneumonia, etc. According to the previously stated report, preterm birth rates among African-American women (14.4%) in 2020 were approximately 50% higher than those among white or Hispanic women (9.1 percent and 9.8 percent respectively). This is likely to boost the growth of the segment in upcoming years.
A lucrative expansion of the market will also result from the introduction of new items in the area. For instance, SAANS, one of the first portable, neonatal CPAP devices in the world, was introduced by InnAccel in August 2019. It is designed to give severely ill newborns with Respiratory Distress Syndrome (RDS) breathing support both during transportation and in situations with limited resources. In addition, Medtronic purchased SonarMed Inc. in December 2020 in order to introduce SonarMed Airway Monitoring System for NICUs. All newborns who need help breathing are likely to receive better treatment through this technology. As a result, such strategic moves by major market competitors would increase the range of products available in the neonatal ICU segment, propelling the market. As a result, it is anticipated that the aforementioned reasons will significantly increase the segment's growth, propelling the market.
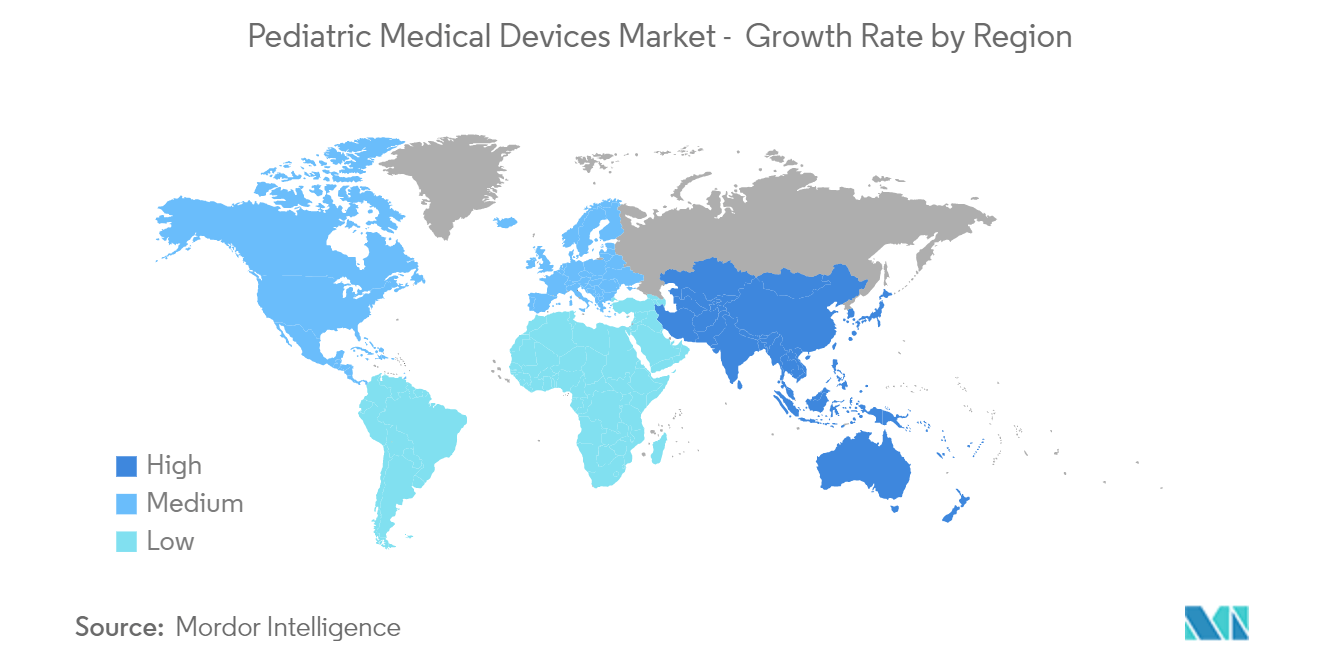
North America Dominates the Market and Expected to do Same in the Forecast Period
North America is expected to dominate the overall market, throughout the forecast period. The market growth is due to the factors such as the presence of key players, high prevalence of pediatric chronic diseases in the region, established healthcare infrastructure, and launch of new products. In the United States, more than 40% of school-aged children and adolescents have at least one chronic health condition, such as asthma, obesity, other physical diseases, and behavior/learning issues, according to the Centers for Disease Control and Prevention's October 2021 report. Children with chronic illnesses can have complex, ongoing healthcare needs that require both routine management and preparation for potential emergencies. As a result, pediatric medical devices are created with the idea of providing enhanced care, which will greatly influence the market.
The country's studied market will benefit from the product debuts there as well. Preceptis Medical, Inc., for instance, announced the release of its next-generation Hummingbird Tympanostomy Tube System (TTS) for office-based pediatric ear tube surgeries in June 2021. The new Hummingbird device has an improved ergonomic layout that makes it possible to administer ear tubes to toddlers more successfully. The Harmony Transcatheter Pulmonary Valve (TPV) System, which treats pediatric patients with a native or surgically repaired right ventricular outflow tract (RVOT), the portion of the heart that carries blood from the right ventricle to the lungs, received approval from the U.S. Food and Drug Administration in March 2021. The device is intended for patients with severe pulmonary valve regurgitation, which is characterised by blood leakage into the right lower chamber of the heart. Congenital heart disease frequently causes this illness. In addition, encouraging government initiatives and a rise in research collaboration numbers are among the factors anticipated to boost market expansion. Due to favourable healthcare policies, a large patient population, and a developed healthcare market, the region is expected to grow in the future.