Market Trends of NA Pharmaceutical CMO Industry
This section covers the major market trends shaping the North America Pharmaceutical Contract Manufacturing Market according to our research experts:
Finished Dosage Formulation (FDF) Development and Manufacturing is Expected to Witness Significant Growth
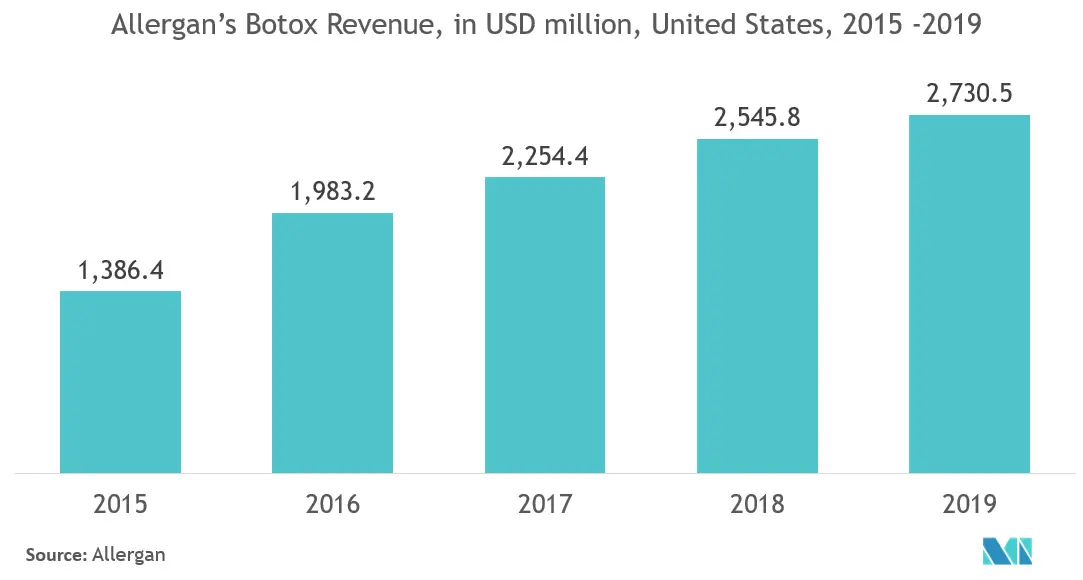
- The International Society of Aesthetic Plastic Surgery reported that with 4,361,867 cases, the United States ranked first in the total number of cosmetic procedures, in 2018. As of 2019, of those surveyed by the American Society for Dermatologic Surgery (ASDS), 70% were somewhat to extremely bothered by the lines and wrinkles around the eyes and led them to cosmetic procedures. The ASDS also stated that the demand for cosmetic surgery products is expected to rise in the United States, with injectables as the most significant and fastest-growing segment in the market.
- The US FDA later approved the use of injectable dermal fillers based on the review of data collected from controlled clinical studies that evaluated the safe and effective use of wrinkle fillers when injected into particular areas of facial tissue. Additionally, botox injections have increased in the country over the years. Moreover, according to the American Academy of Cosmetic Dentistry's Cosmetic Dentistry State of the Industry Survey, there was an increase in the number of patients seeking cosmetic dentistry and a rise in the average production per cosmetic patient per visit. This has also driven the demand for facial dentistry in the country.
- Baxter also acquired Claris injectables Ltd and gained complete access to the Claris Injectables product portfolio. Meitheal Pharmaceuticals Inc., a generic injectables company, got the US FDA approval for Enoxaparin Sodium Injection, USP, the generic equivalent of Lovenox. The company licensed Enoxaparin Sodium Injection exclusively through its partnership with Nanjing King-friend Biochemical Pharmaceutical Co. Ltd. Additionally, according to ICRA Limited, injectables drugs worth USD 16 billion went off-patent in the United States alone. The United States is witnessing a rise in demand for generic injectables, on account of increasing investment by the US Government for the development of generic drugs.
- Furthermore, the increase in the number of chronic illnesses, like diabetes and cancer (with roughly 1.8 million people diagnosed with cancer in 2019 and 34 million diabetic people in the United States), has increased the requirement for injectable drug delivery. Moreover, in the United States, drug shortages are a prime concern for consumers and the FDA. Though the number of new drug shortages has declined since 2011 due to the work of the FDA, industry, and other stakeholders, there is still a shortage of medically necessary products. In 2018, 63.7% of all drugs in shortage were sterile injectables, reported the US FDA. Quality problems at manufacturing sites can cause supply disruptions.
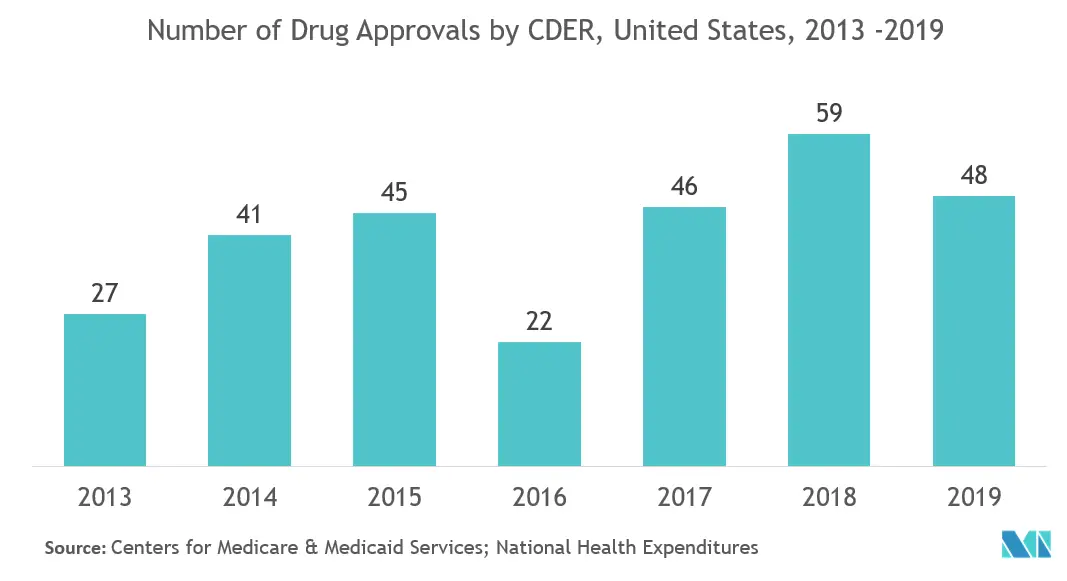
United States is Expected to Hold Major Share
- The United States has emerged as the largest market for drugs and accounts for almost half of the R&D spending in pharmaceutical and biotechnology markets. Hence, CMOs play a critical role in this market and have invested in new facilities and technologies to cater to a wide range of outsourcers. The country is experiencing a shortage of manufacturing capability for specific sectors, like cell and gene therapy. The CMOs have increased their manufacturing bases over the past two years. 2018 has been widely considered as a ‘breakthrough year’ for the United States in terms of approval of new molecular entities and biologics applications. In 2018, the Center for Drug Evaluation and Research approved a record 59 novel drugs in nearly two decades.
- The United States health spending is expected to have a 0.8% faster growth than the gross domestic product (GDP) per year over the 2018-27 period. The health share of GDP would rise from 17.9% in 2017 to 19.4% by 2027, as per estimates from the Centers for Medicare & Medicaid Services. The stricter regulation in the country ensures superior quality of the manufacturing and the end-product that is being adhered to by the CMOs. For instance, manufacturing autologous or allogeneic therapies is complex, and the manufacturing facilities are required to attain a GMP accreditation.
- In February 2020, Alchem Laboratories Corporation, a contract manufacturing organization based in Alachua, Florida, focused on custom synthesis, announced an expansion of its screening and clinical trial manufacturing capabilities. The development was made through construction of Building 3, a 12,000 square foot multi-use facility dedicated to high-throughput screening, biologic and drug product manufacturing clean-room areas, and analytical support. Pfizer, in mid-2019, announced an additional investment of USD 500 with the strategy is in line to increase manufacturing capability at its gene therapy plant, 230-acre campus in Sanford, North Carolina.