Market Trends of Multiple Myeloma Industry
This section covers the major market trends shaping the Multiple Myeloma Market according to our research experts:
Immunomodulators Segment Expects to Register a Significant CAGR Over the Forecast Period
The immunomodulators segment is expected to hold a significant share in the market over the forecast period owing to the factors such as the rising burden of multiple myeloma among the population as well as rising demand for immunomodulators drugs.
Immunomodulators alter or delay the progression of numerous autoimmune, inflammatory, and cancerous disorders. Some of the drugs that are used for treating multiple myeloma are Thalomid and its derivatives, Revlimid, and pomalyst.
The rising prevalence and incidence of multiple myeloma among the population is the key factor driving the demand for immunomodulator drugs over the forecast period. For instance, according to an article published in Blood Advances, in May 2021, it has been observed that lenalidomide, an immunomodulatory drug licensed for maintenance therapy in newly diagnosed multiple myeloma patients, has demonstrated improvements in progression-free survival, the depth and quality of the response, and, ultimately, improves the survival outcomes. Thus, such outcomes and increased survival rates of lenalidomide drug are expected to increase its adoption among patients, hence propelling the market growth.
Furthermore, the increasing company activities in developing drugs and rising drug approvals are also contributing to the market growth. For instance, in February 2022, Sandoz launched the generic oncology drug lenalidomide, which is indicated for several haemato-oncology conditions including multiple myeloma. Moreover, in March 2022, Teva Pharmaceuticals launched the first generic version of Revlimid (lenalidomide capsules), in 5mg, 10mg, 15mg, and 25mg strengths, in the United States.
Therefore, due to the above-mentioned factors, such as the rising adoption of lenalidomide for treating multiple myeloma and increasing product launches and approvals, the studied market is expected to witness significant growth over the forecast period.
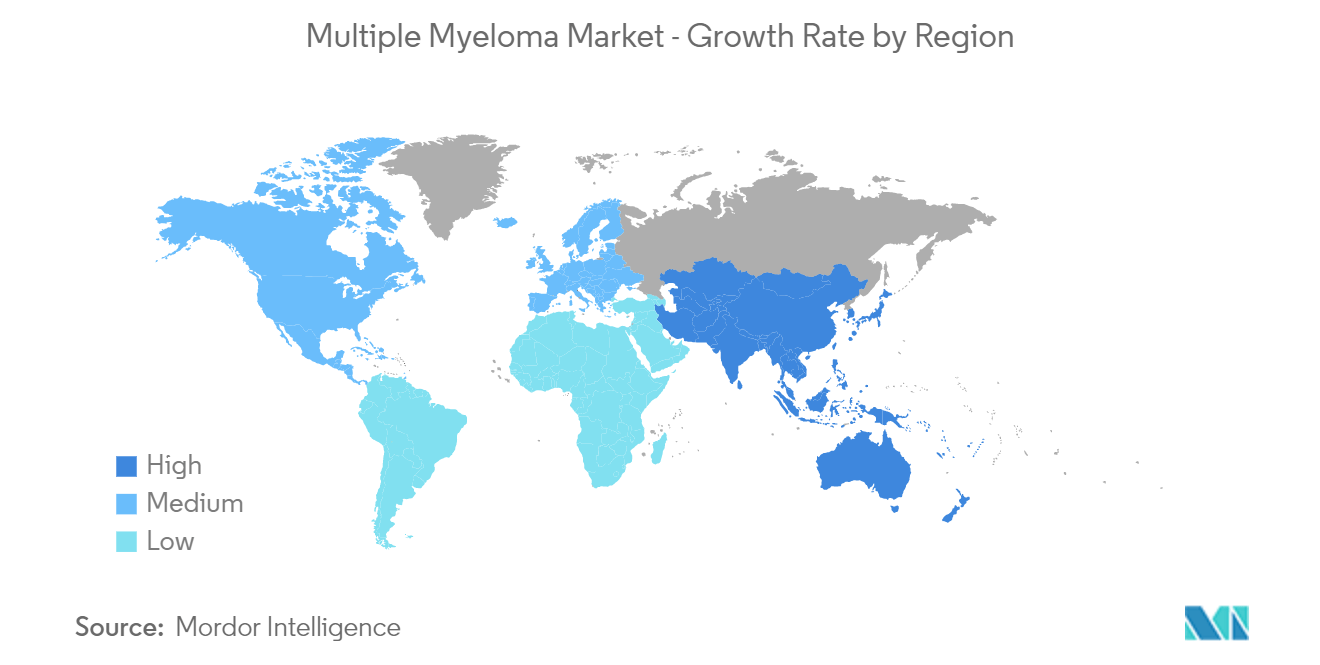
North America is Expected to Hold a Significant Share in the Market and Expected to do Same Over the Forecast Period
North America is expected to witness a significant share in the multiple myeloma market over the forecast period owing to the factors such as the increasing burden of multiple myeloma, growing company activities in developing effective treatment drugs, rising healthcare expenditure along with high healthcare spending, and the presence of key market players in the region.
The increasing prevalence of multiple myeloma is the key factor driving the market growth. For instance, according to the 2022 statistics published by ACS, about 34,470 new cases (19,100 in men and 15,370 in women) of multiple myeloma are expected to be diagnosed in the United States in 2022. In addition, as per the same source, multiple myeloma is a relatively uncommon cancer, and in the United States, the lifetime risk of getting multiple myeloma is 1 in 132 (0.76%). Additionally, as per the 2022 statistics published by the Canadian Cancer Society, about 4,000 Canadians are expected to be diagnosed with multiple myeloma (MM) in 2022. Thus, the high number of MM cases increases the demand for effective drugs for treating the disease which in turn is anticipated to augment the market growth over the forecast period.
Furthermore, the rising initiatives in launching several awareness programs and campaigns for increasing public awareness regarding the MM condition are also contributing to the market growth. For instance, as per a February 2022 update, Myeloma Action Month is organized with the International Myeloma Foundation (IMF), every year during the entire month of March, to inspire people and organizations to adopt initiatives that will improve the myeloma community in the United States. This year, the IMT invited participants to Myeloma Action Month to take action against the disease.
Moreover, the increasing company focus on adopting several strategic initiatives, such as collaboration, partnership, product launches, and approvals, to withhold their market position is also contributing to the market growth. For instance, in February 2022, the United States FDA approved Janssen's first cell therapy Carvykti (ciltacabtagene autoleucel), a B-cell maturation antigen (BCMA)-directed, genetically modified autologous T-cell immunotherapy, for the treatment of relapsed or refractory multiple myeloma in adults. Also, in May 2021, Health Canada granted conditional approval to Bristol Myers Squibb's ABECMA (idecabtagene vicleucel; ide-cel) as the first and only B-cell maturation antigen (BCMA)-directed chimeric antigen receptor (CAR) T cell immunotherapy for adults with multiple myeloma. The drug is indicated for MM patients who have received at least three prior therapies, including an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 antibody, and who are refractory to their last treatment.
Therefore, owing to the aforementioned factors, such as the rising burden of multiple myeloma, increasing awareness programs as well as growing product launches and approvals, the studied market is expected to grow over the forecast period in the North America region.