Market Size of Global Migraine Therapeutics Industry
| Study Period | 2019 - 2029 |
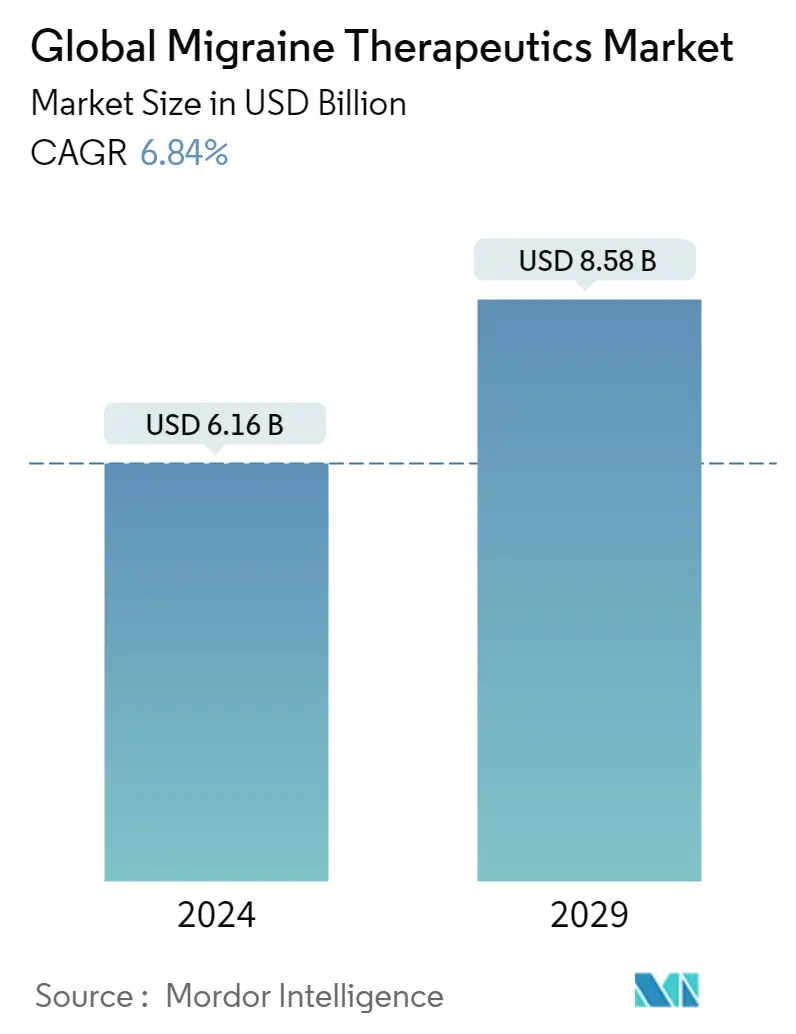
| Market Size (2024) | USD 6.16 Billion |
| Market Size (2029) | USD 8.58 Billion |
| CAGR (2024 - 2029) | 6.84 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Migraine Therapeutics Market Analysis
The Global Migraine Therapeutics Market size is estimated at USD 6.16 billion in 2024, and is expected to reach USD 8.58 billion by 2029, growing at a CAGR of 6.84% during the forecast period (2024-2029).
COVID-19 was a global public health crisis, and it impacted almost every industry, and its long-term effects were expected to affect industry growth over the forecast period. According to the study titled "Impact of COVID-19 pandemic on migraine management in the United States: insights from migraine tracking app users" published in BMC neurology in September 2021, although the number of migraine attacks decreased, the proportion of stress-related migraine attacks peaked at 53% during COVID-19. Also, the study titled "COVID-19 and Headache Medicine: A Narrative Review of NonSteroidal AntiInflammatory Drug (NSAID) and Corticosteroid Use" published in July 2021, reported that there may be an increase in the number of patients with headaches as their primary complaint during the COVID19 pandemic. Hence, COVID-19 had a significant impact on the migraine therapeutics market.
Growing awareness about migraine and treatment options, rising R&D spending leading to massive pipeline products, and an increase in the prevalence of migraines with high unmet needs are all contributing to the global growth of the migraine therapeutics market.
The various strategic activities by the key market players, such as product launches and product approvals, as well as mergers and acquisitions, boost the market over the forecast period. For instance, in March 2022, AbbVie reported a positive phase 3 progress trial evaluating atogepant (QULIPTA in the United States) for the preventive treatment of chronic migraine in adults. It is an oral calcitonin gene-related peptide (CGRP) receptor antagonist (gepant). In the trial, the drug met its primary endpoint of a statistically significant reduction from baseline in mean monthly migraine days compared to placebo, for both the 60 mg once daily (QD) and 30 mg twice daily (BID) doses, across the 12-week treatment period.
Additionally, in September 2021, the United States Food and Drug Administration (FDA) approved QULIPTA (atogepant) to AbbVie for the prevention of episodic migraine in adults. QULIPTA is one of the world's first and only oral calcitonin gene-related peptide (CGRP) receptor antagonists (gepant) developed specifically for migraine prevention.
Thus, the market is expected to project growth over the forecast period. However, the growth of the migraine therapeutics market may be hampered due to a lack of proper diagnosis, undiagnosed cases, and drug side effects over the forecast period.
