Market Size of Kidney Cancer Therapeutics and Diagnostics Industry
| Study Period | 2021 - 2029 |
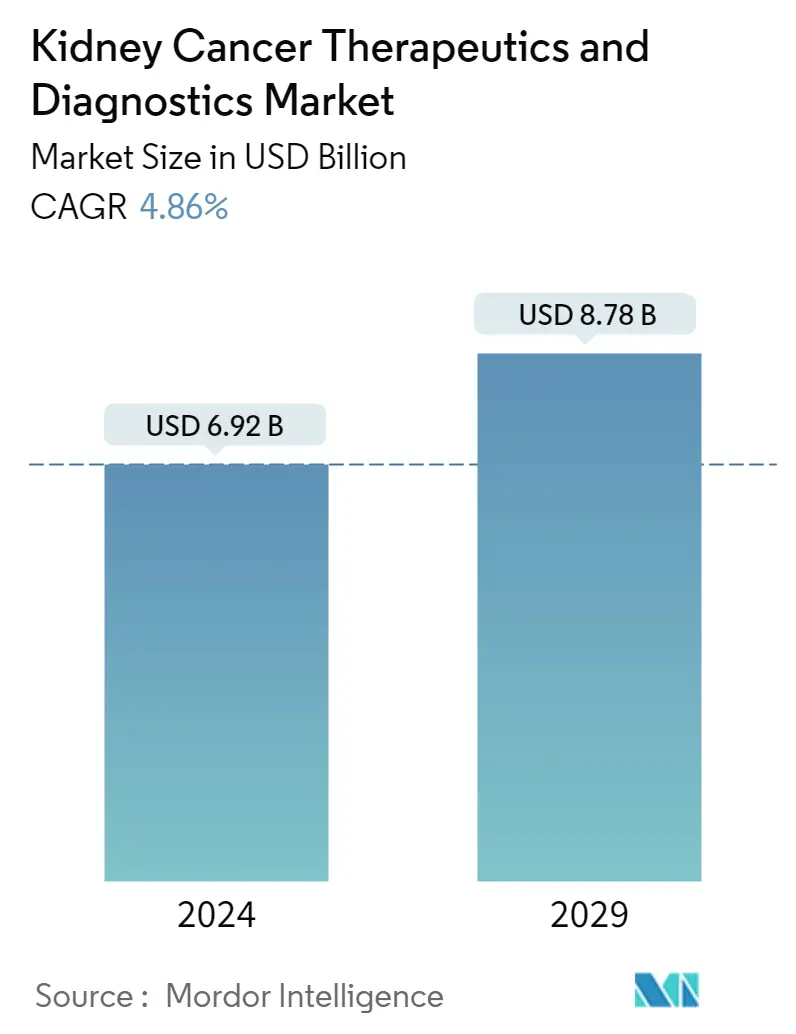
| Market Size (2024) | USD 6.92 Billion |
| Market Size (2029) | USD 8.78 Billion |
| CAGR (2024 - 2029) | 4.86 % |
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Kidney Cancer Therapeutics & Diagnostics Market Analysis
The Kidney Cancer Therapeutics and Diagnostics Market size is estimated at USD 6.92 billion in 2024, and is expected to reach USD 8.78 billion by 2029, growing at a CAGR of 4.86% during the forecast period (2024-2029).
The COVID-19 pandemic affected healthcare systems and resulted in the interruption of usual care in many healthcare facilities, exposing vulnerable patients with cancer, including kidney cancer, to significant risks. For instance, as per the article published in November 2022, during the first COVID wave (weeks 9-22), the number of renal cell carcinoma (RCC) diagnoses decreased by 15%. The decline was most pronounced in weeks 9-40 in patients >70 years of age with stage 1 disease. Thus, during the COVID-19 pandemic, the market for kidney cancer therapeutics and diagnostics was severely impacted. However, as per the study published in the BMC Journal in December 2022, once COVID-19 had infected cancer patients, a number of genetic changes brought on by COVID-19 may have rendered the first treatment ineffective, increasing the challenge of treating cancer clinically. In accordance with the study's findings, the GINS1 gene is very sensitive to COVID-19 in all 11 forms of cancer, including those of the lung, stomach, liver, esophagus, thyroid, ovary, cervix, breast, colorectum, prostate, and kidney. Furthermore, the study detailed that there is a rational explanation regarding cancer patients infected with COVID-19 could develop a more severe condition since GINS1 is expressed more strongly in cancer tissues than in normal tissues. Thus, as per the analysis, such findings are anticipated to bolster the demand for diagnosis and therapeutics in kidney cancer patients along with new emerging strains of the SARS-CoV-2 virus globally, in turn boosting the market growth over the post-pandemic phase.
The market is primarily driven by the increase in the prevalence of kidney cancer and increased research and development expenditures from pharmaceutical companies. According to a report by the International Kidney Cancer Coalition in July 2021, each year, around 431,000 people worldwide are diagnosed with kidney cancer. Thus, with the growing prevalence of kidney cancer, it is expected that the demand for kidney cancer therapeutics and diagnostics is likely to increase over the forecast period. Additionally, product launches by the key market players will support the market expansion over the forecast period. For instance, in February 2021, Glenmark Pharma introduced SUTIB, a generic kidney cancer treatment, in India. Sunitinib, an oral multikinase inhibitor (MKI), is effective in treating individuals with gastrointestinal stromal tumors and advanced renal cell carcinoma. Similarly, in September 2021, MSN Labs launched Cabolong, a branded generic of Cabozantinib, for the treatment of renal cell carcinoma.
Moreover, funding for kidney cancer research increases the opportunities to develop a therapy for kidney cancer, which is anticipated to boost market growth over the forecast period. For instance, in September 2022, Weill Cornell Medicine was awarded a USD 1 million, three-year grant from the Department of Defense's Kidney Cancer Research Program to fund research on the role of the protein ATF4 in clear cell renal cell carcinoma (ccRCC), a form of kidney cancer. Similarly, in July 2022, Bristol-Myers Squibb announced the results of its Phase III trial of the company's Opdivo and Yervoy combination in renal cell carcinoma (RCC).
Thus, due to the rise in kidney diseases, increase in the funding of research in kidney disease, and surge in health care expenditure, the kidney cancer therapeutics and diagnostics market is anticipated to witness growth in the market over the forecast period. However, the high cost associated with the treatment and low success rate in clinical trials for cancer drugs may restrain market growth over the forecast period.
