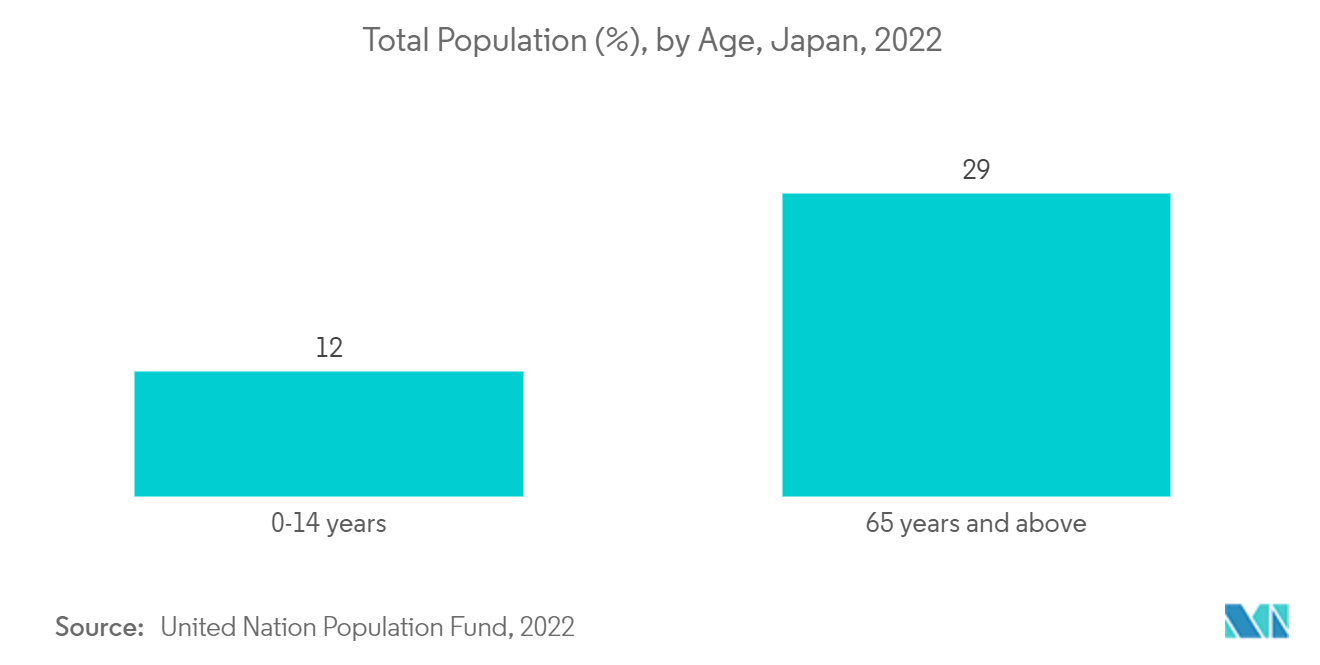Market Trends of Japan Pharmaceutical Industry
This section covers the major market trends shaping the Japan Pharmaceutical Market according to our research experts:
Prescription Drugs Segment is Expected to Hold a Significant Share in the Market Over Forecast Period
The prescription drugs segment is expected to witness significant growth in the pharmaceutical market over the forecast period due to factors such as the rising prevalence of chronic diseases, the increasing geriatric population, the growing demand and adoption of prescription drugs, and increasing product launches in the country.
The rising number of people suffering chronic diseases in Japan is increasing the demand for the development of pharmaceutical products such as vaccines, biologics, or other therapeutic drugs, hence boosting the market's growth. According to the WHO, as of August 9, 2022, 14,421,539 confirmed cases of COVID-19 and 33,825 deaths were reported in Japan. Thus, the increasing number of COVID-19 cases among the population is expected to increase the demand for COVID-19 vaccines in the country, thereby propelling the market's growth.
The rising company focus on developing pharmaceutical products and adopting various business strategies such as collaboration, partnerships, and product launches are also contributing to the growth of the market studied. For instance, in March 2022, the Japanese Ministry of Health, Labor, and Welfare (MHLW) granted marketing authorization for Xenpozyme (olipudase alfa) for the treatment of adult and pediatric patients with non-central nervous system (non-CNS) manifestations of acid sphingomyelinase deficiency (ASMD). In January 2022, Chugai's anti-IL-6 receptor monoclonal antibody Actemra was approved for the additional indication of the treatment of SARS-CoV-2 pneumonia (limited to patients requiring oxygen intervention) in Japan.
Therefore, due to the above-mentioned factors, the prescription drugs segment is expected to hold a significant market share in the Japanese pharmaceutical market over the forecast period.
Respiratory System Segment is Expected to Have a Significant Market Share Over the Forecast Period
The respiratory segment is expected to grow over the forecast period due to the increasing prevalence of respiratory diseases, such as asthma, chronic obstructive respiratory diseases, and others. The rising pediatric and aging population is more susceptible to developing respiratory diseases due to weak immunity, which is also contributing to the market's growth. According to the GLOBOCAN 2020 report, 138,532 new lung cancer cases were reported in Japan in 2020, and this number is projected to double by 2040. According to an article published in the Journal of Thoracic Disease in June 2021, the prevalence of chronic obstructive pulmonary disease (COPD) was higher among the population aged 40 years and above. Thus, the high burden of respiratory diseases among the population is anticipated to increase the demand for asthma and COPD drugs, propelling the market's growth.
The rising company activities in developing drugs and increasing drug approvals are expected to increase the market's growth. For instance, in September 2022, the Japanese MHLW approved AstraZeneca's Tezspire (tezepelumab) for the treatment of bronchial asthma in patients with severe or refractory disease in whom asthma symptoms cannot be controlled with mid- or high-dose inhaled corticosteroids and other long-term maintenance therapies, in Japan. In August 2022, the Japanese MHLW approved AstraZeneca's Tagrisso (osimertinib) for the adjuvant treatment of patients with epidermal growth factor receptor mutated (EGFRm) non-small cell lung cancer (NSCLC) after surgery in Japan.
In May 2022, the Japanese MHLW approved Chugai Pharmaceutical's Tecentriq for an additional indication of the anti-cancer agent/humanized anti-PD-L1 monoclonal antibody for the adjuvant treatment of PD-L1-positive non-small cell lung cancer (NSCLC). In January 2022, the Japanese MHLW approved Amgen's Lumakras for the treatment of KRAS G12C-mutated positive, unresectable, advanced, and recurrent non-small cell lung cancer (NSCLC) that has progressed after systemic anti-cancer therapy in Japan.
Therefore, due to the above-mentioned factors, the respiratory system segment is expected to grow during the forecast period.