Market Size of Europe Companion Diagnostics Industry
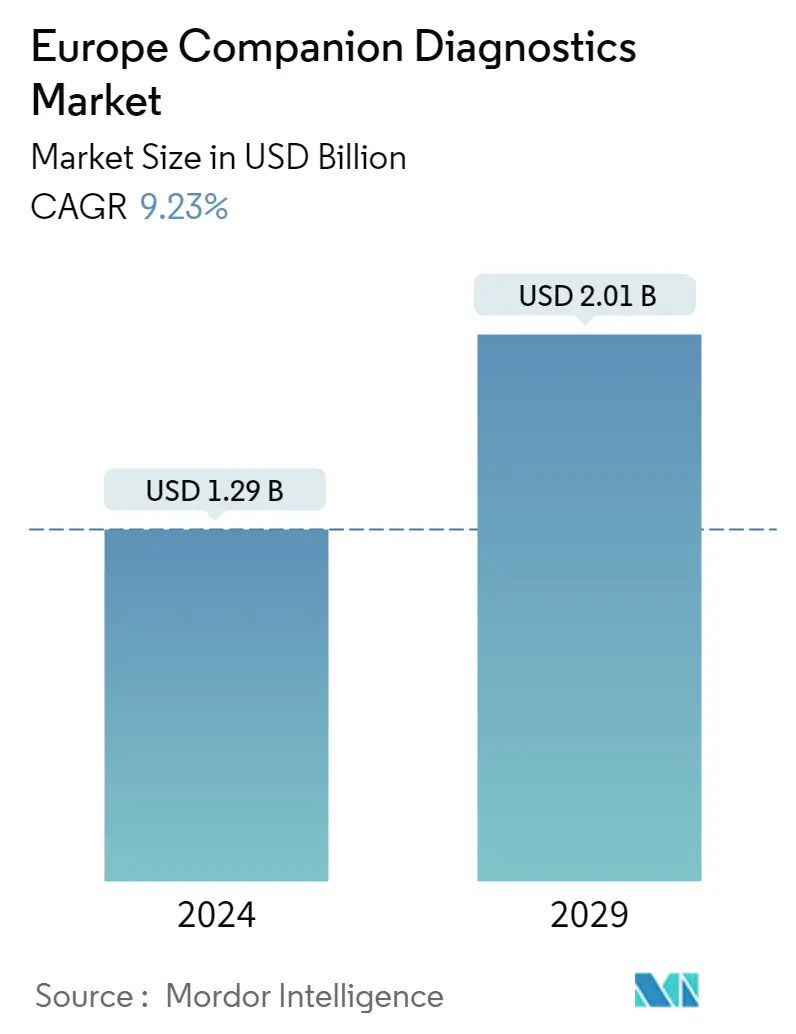
| Study Period | 2021 - 2029 |
| Base Year For Estimation | 2023 |
| Market Size (2024) | USD 1.29 Billion |
| Market Size (2029) | USD 2.01 Billion |
| CAGR (2024 - 2029) | 9.23 % |
| Market Concentration | Low |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Europe Companion Diagnostics Market Analysis
The Europe Companion Diagnostics Market size is estimated at USD 1.29 billion in 2024, and is expected to reach USD 2.01 billion by 2029, growing at a CAGR of 9.23% during the forecast period (2024-2029).
The COVID-19 pandemic initially had a substantial impact on the companion diagnostics market. The usage of companion diagnostic products showed a significant rise during the pandemic as research for various COVID-19 products increased during that period. For instance, an article published by Bioanalysis Zone in May 2021 stated that companion diagnostics played a major role in guiding, understanding, and managing COVID-19 products. It further stated that companion diagnostics played an immense role as it helped in understanding the COVID-19 disease and its short and long-term impact on health. Currently, the usage of companion diagnostics has become normal as the pandemic has almost subsided. Thus, the COVID-19 outbreak affected the market's growth favorably in its preliminary phase; however, the market is expected to have stable growth during the forecast period of the study.
The growth of the Europe companion diagnostics market can be attributed to the increasing focus on personalized medicine and the co-development of drug and diagnostic technologies. In addition, rising cases of adverse drug reactions related to drugs drive the need for companion diagnostics. The other factors that drive the market include the rising prevalence of chronic diseases, rise in biomarker discovery, reduced drug development costs, and increasing partnerships between market players and government organizations.
For instance, according to an article published by Future Medicine in April 2021, there are joint funding schemes present in Europe that are cooperating with European Commission (EC) to provide funds for basic and translational research in developing personalized medicines. Investments in personalized and genomic medicine initiatives were also being made in Europe, and the implementation of regional strategies is being carried out with personalized medicine at its core. Furthermore, in December 2021, QIAGEN and Denovo Biopharma LLC announced a collaboration to develop a blood-based companion diagnostic (CDx) test to identify patients expressing Denovo Genomic Marker 1 (DGM1) who are likely to respond to Denovo's investigational cancer drug DB102 for treatment of diffuse large B-cell lymphoma (DLBCL), which is one of the most common lymphoid cancers. Moreover, in July 2021, Labcorp announced the availability of therascreen KRAS PCR Mutation Analysis, a companion diagnostic for identifying patients with non-small cell lung cancer (NSCLC) who are eligible for treatment with LUMAKRAS (sotorasib), a new treatment option developed by Amgen. Hence, the increasing focus on personalized medicines and other emerging healthcare technologies like biomarkers are poised to enhance the growth of the studied market during the forecast period.
Oncology is one of the primary areas that has significantly benefitted due to the advent of companion diagnostics. Companion diagnostics have been highly beneficial in identifying patients who need specific biomarkers for proper responses. Companion diagnostics have the power to predict the response of any particular therapy targeted for a specific person, which, in turn, results in better patient treatments and the reduction of healthcare expenditure. For instance, in November 2022, Akoya Biosciences, Inc. launched the PhenoCode Signature Panels for high-throughput spatial biomarker discovery for cancer immunotherapy.
Therefore, owing to the aforementioned factors, the studied market is anticipated to witness growth over the analysis period. However, the high cost of drug development and associated clinical trials and reimbursement issues are likely to impede the market's growth.
